Summary
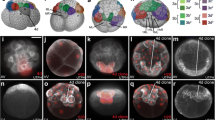
A detailed fate map of all the progeny derived from each of the blastomeres of the 4- and 8-cell stage South African clawed frog (Xenopus laevis) embryo is presented. Each “identified” blastomere that results from stereotypic cleavages has a characteristic set of progeny that distinguishes it from the other blastomeres of the embryo. The 4-cell dorsal (D) blastomere is the major progenitor of the stomodeum, cement gland, retina, notochord, head somite, pharynx and liver. The 4-cell ventral (V) blastomere is the major progenitor of the trunk and fin epidermis, ventral somite, nephrotome, lateral plate mesoderm and proctodeum. The other organs are derived from both blastomeres. At the next cell division, the animal hemisphere daughters of both blastomeres (D1 and V1, respectively) become the major progenitors for head ectodermal and mesodermal structures, and the vegetal hemisphere daughters become the major progenitors for trunk mesodermal (D2) or trunk endodermal (V2) structures. Semiquantitative lineage diagrams, using data from this and from previous studies demonstrate that as cleavage proceeds from the 2- to the 32-cell stage, the progenitors for particular organs or for specific regions of organs segregate into defined regions of the blastula. To determine whether this segregation is related to the position of the blastomere or to its geneological lineage, we compared the fates of radial 8-cell blastomeres to those of stereotypic 8-cell blastomeres. Radial blastomeres have fates nearly equivalent to the sum of the two 16-cell blastomeres that occupy the same position in the embryo, demonstrating that fate depends upon blastomere position rather than lineage.
Similar content being viewed by others
References
Heasman J, Wylie CC, Hausen P, Smith JC (1984) Fates and states of determination of single vegetal pole blastomeres ofXenopus laevis. Cell 37:185–194
Hirose G, Jacobson M (1979) Clonal organization of the central nervous system of the frog. I. Clones stemming from individual blastomeres of the 16-cell and earlier stages. Dev Biol 71:191–202
Jacobson M (1985) Clonal analysis and cell lineages of the vertebrate central nervous system. Annu Rev Neurosci 8:71–102
Jacobson M, Hirose G (1981) Clonal organization of the central nervous system of the frog. II. Clones stemming from individual blastomeres of the 32- and 64-cell stages. J Neurosci 1:271–284
Jacobson M, Wei-long X (1989) States of determination of single cells transplanted between 512-cellXenopus embryos. Dev Biol 131:119–125
Jones EA, Woodland HR (1987) The development of animal cap cells inXenopus: a measure of the start of animal cap competence to form mesoderm. Development 101:557–563
Kageura H, Yamana K (1983) Pattern regulation in isolated halves and blastomeres of earlyXenopus laevis. J Embryol Exp Morphol 74:221–234
Kageura H, Yamana K (1984) Pattern regulation in defect embryos ofXenopus laevis. Dev Biol 101:410–415
Kimmel CB, Law RD (1985) Cell lineage of zebrafish blastomeres. III. Clonal analysis of the blastula and gastrula stages. Dev Biol 108:94–101
Klein SL (1987) The first cleavage furrow demarcates the dorsalventral axis inXenopus laevis embryos. Dev Biol 120:299–304.
Klein SL, Moody SA (1989) Lithium changes the ectodermal fate of individual frog blastomeres because it causes ectopic neural plate formation. Development 106:599–610
Kuhtreiber WM, Serras F, van den Biggelaar JAM (1987) Spreading of microinjected horseradish peroxidase to nondescendant cells in embryos ofPatella (Molusca, Gastropoda). Development 100:713–722
Masho R (1988) Fates of animal dorsal blastomeres of eight-cell stageXenopus embryos vary according to the specific patterns of the third cleavage plane. Dev Growth Differ 30:347–359
Masho R (1990) Close correlation between the first cleavage plane and the body axis in earlyXenopus embryos. Dev Growth Differ 32:57–64
Masho R, Kubota HY (1986) Developmental fates of blastomeres of eight-cell stageXenopus laevis embryos. Dev Growth Differ 28:113–123
Moody SA (1987a) Fates of the blastomeres of the 16-cellXenopus embryo. Dev Biol 119:560–578
Moody SA (1987b) Fates of the blastomeres of the 32-cellXenopus embryo. Dev Biol 122:300–319
Moody SA (1989) Quantitative lineage analysis of the origin of frog primary motor and sensory neurons from cleavage stage blastomeres. J Neurosci 9:2919–2930
Newport J, Kirschner M (1982) A major developmental transition in earlyXenopus embryos. I. Characterization and time of cellular changes at the midblastula stage. Cell 30:675–686
Nieuwkoop PD, Faber J (1964) Normal Table ofXenopus laevis (Daudin). North-Holland, Amsterdam
Nishida H, Satoh N (1983) Cell lineage analysis in Ascidian embryos by intracellular injection of a tracer enzyme. I. Up to the eight-cell stage. Dev Biol 99:382–394
Slack JMW (1983) From Egg to Embryo, Cambridge Univ Press, London
Stent GS (1985) The role of cell lineage in development. Philos Trans R Soc Lond [Biol] 312:3–19
Stent GS, Weisblat DA (1985) Cell lineage in the development of invertebrate nervous systems. Annu Rev Neurosci 8:45–70
Sulston JE, Schierenberg E, White JG, Thomson JN (1983) The embryonic cell lineage of the nematodeCaenorhabditis elegans. Dev Biol 100:64–119
Weisblat DA, Harper G, Stent GS, Sawyer RT (1980) Embryonic cell lineages in the nervous system of the glossiphoniid leechHelobdella triserialis. Dev Biol 76:58–78
Weisblat DA, Kim SY, Stent GS (1984) Embryonic origins of cells in the leechHelobdella triserialis. Dev Biol 104:64–85
Ziomek CA, Johnson MH, Handyside AH (1982) The developmental potential of mouse 16-cell blastomeres. J Exp Zool 221:345–355