Abstract
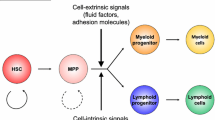
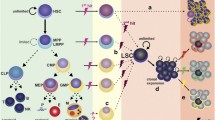
Hematopoiesis is hierarchically orchestrated by a very small population of hematopoietic stem cells (HSCs) that reside in the bone-marrow niche and are tightly regulated to maintain homeostatic blood production. HSCs are predominantly quiescent, but they enter the cell cycle in response to inflammatory signals evoked by severe systemic infection or injury. Thus, hematopoietic stem and progenitor cells (HSPCs) can be activated by pathogen recognition receptors and proinflammatory cytokines to induce emergency myelopoiesis during infection. This emergency myelopoiesis counterbalances the loss of cells and generates lineage-restricted hematopoietic progenitors, eventually replenishing mature myeloid cells to control the infection. Controlled generation of such signals effectively augments host defense, but dysregulated stimulation by these signals is harmful to HSPCs. Such hematopoietic failure often results in blood disorders including chronic inflammatory diseases and hematological malignancies. Recently, we found that interleukin (IL)-27, one of the IL-6/IL-12 family cytokines, has a unique ability to directly act on HSCs and promote their expansion and differentiation into myeloid progenitors. This process resulted in enhanced production of neutrophils by emergency myelopoiesis during the blood-stage mouse malaria infection. In this review, we summarize recent advances in the regulation of myelopoiesis by proinflammatory cytokines including type I and II interferons, IL-6, IL-27, granulocyte colony-stimulating factor, macrophage colony-stimulating factor, and IL-1 in infectious diseases.




Similar content being viewed by others
Abbreviations
- BM:
-
Bone marrow
- CAR:
-
C-X-C motif ligand 12-abundant reticular
- CTL:
-
Cytotoxic CD8+ T cell
- CXCL:
-
C-X-C motif ligand
- DC:
-
Dendritic cell
- EC:
-
Endothelial cell
- G-CSF:
-
Granulocyte colony-stimulating factor
- gp130:
-
β-Receptor glycoprotein 130
- GM-CSF:
-
Granulocyte macrophage colony-stimulating factor
- HSC:
-
Hematopoietic stem cell
- HSPC:
-
Hematopoietic stem and progenitor cell
- IFN:
-
Interferon
- IFNAR:
-
IFN-α receptor
- IL:
-
Interleukin
- JAK:
-
Janus kinase
- LCMV:
-
Lymphocytic choriomeningitis virus
- LSK:
-
Lineage−Sca-1+c-Kit+
- LT-HSC:
-
Long-term repopulating hematopoietic stem cell
- M-CSF:
-
Macrophage colony-stimulating factor
- MDSC:
-
Myeloid-derived suppressor cell
- MPP:
-
Multipotent progenitor
- MYD88:
-
Myeloid differentiation primary response gene 88
- MyRP:
-
Myeloid-restricted progenitor cell
- MSC:
-
Mesenchymal stem/stromal cell
- NK:
-
Natural killer
- P. :
-
Plasmodium
- R:
-
Receptor
- RBC:
-
Red blood cell
- Sca-1:
-
Stem cell antigen-1
- SCF:
-
Stem cell factor
- STAT:
-
Signal transducer and activator of transcription
- Th:
-
Helper T
- TLR:
-
Toll-like receptor
- Treg:
-
Regulatory T
References
Orkin SH, Zon LI (2008) Hematopoiesis: an evolving paradigm for stem cell biology. Cell 132(4):631–644. https://doi.org/10.1016/j.cell.2008.01.025
Oguro H, Ding L, Morrison SJ (2013) SLAM family markers resolve functionally distinct subpopulations of hematopoietic stem cells and multipotent progenitors. Cell Stem Cell 13(1):102–116. https://doi.org/10.1016/j.stem.2013.05.014
Pang WW, Price EA, Sahoo D, Beerman I, Maloney WJ, Rossi DJ, Schrier SL, Weissman IL (2011) Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proc Natl Acad Sci USA 108(50):20012–20017. https://doi.org/10.1073/pnas.1116110108
Mohrin M, Bourke E, Alexander D, Warr MR, Barry-Holson K, Le Beau MM, Morrison CG, Passegue E (2010) Hematopoietic stem cell quiescence promotes error-prone DNA repair and mutagenesis. Cell Stem Cell 7(2):174–185. https://doi.org/10.1016/j.stem.2010.06.014
Ding L, Morrison SJ (2013) Haematopoietic stem cells and early lymphoid progenitors occupy distinct bone marrow niches. Nature 495(7440):231–235. https://doi.org/10.1038/nature11885
Ding L, Saunders TL, Enikolopov G, Morrison SJ (2012) Endothelial and perivascular cells maintain haematopoietic stem cells. Nature 481(7382):457–462. https://doi.org/10.1038/nature10783
Mendez-Ferrer S, Michurina TV, Ferraro F, Mazloom AR, Macarthur BD, Lira SA, Scadden DT, Ma’ayan A, Enikolopov GN, Frenette PS (2010) Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 466(7308):829–834. https://doi.org/10.1038/nature09262
Monteiro JP, Benjamin A, Costa ES, Barcinski MA, Bonomo A (2005) Normal hematopoiesis is maintained by activated bone marrow CD4+ T cells. Blood 105(4):1484–1491. https://doi.org/10.1182/blood-2004-07-2856
Yamazaki S, Ema H, Karlsson G, Yamaguchi T, Miyoshi H, Shioda S, Taketo MM, Karlsson S, Iwama A, Nakauchi H (2011) Nonmyelinating Schwann cells maintain hematopoietic stem cell hibernation in the bone marrow niche. Cell 147(5):1146–1158. https://doi.org/10.1016/j.cell.2011.09.053
Takizawa H, Boettcher S, Manz MG (2012) Demand-adapted regulation of early hematopoiesis in infection and inflammation. Blood 119(13):2991–3002. https://doi.org/10.1182/blood-2011-12-380113
Sato T, Onai N, Yoshihara H, Arai F, Suda T, Ohteki T (2009) Interferon regulatory factor-2 protects quiescent hematopoietic stem cells from type I interferon-dependent exhaustion. Nat Med 15(6):696–700. https://doi.org/10.1038/nm.1973
Essers MA, Offner S, Blanco-Bose WE, Waibler Z, Kalinke U, Duchosal MA, Trumpp A (2009) IFNalpha activates dormant haematopoietic stem cells in vivo. Nature 458(7240):904–908. https://doi.org/10.1038/nature07815
Baldridge MT, King KY, Boles NC, Weksberg DC, Goodell MA (2010) Quiescent haematopoietic stem cells are activated by IFN-gamma in response to chronic infection. Nature 465(7299):793–797. https://doi.org/10.1038/nature09135
Boettcher S, Ziegler P, Schmid MA, Takizawa H, van Rooijen N, Kopf M, Heikenwalder M, Manz MG (2012) Cutting edge: LPS-induced emergency myelopoiesis depends on TLR4-expressing nonhematopoietic cells. J Immunol 188(12):5824–5828. https://doi.org/10.4049/jimmunol.1103253
Boettcher S, Manz MG (2016) Sensing and translation of pathogen signals into demand-adapted myelopoiesis. Curr Opin Hematol 23(1):5–10. https://doi.org/10.1097/MOH.0000000000000201
Mirantes C, Passegue E, Pietras EM (2014) Pro-inflammatory cytokines: emerging players regulating HSC function in normal and diseased hematopoiesis. Exp Cell Res 329(2):248–254. https://doi.org/10.1016/j.yexcr.2014.08.017
Clapes T, Lefkopoulos S, Trompouki E (2016) Stress and non-stress roles of inflammatory signals during HSC emergence and maintenance. Front Immunol 7:487. https://doi.org/10.3389/fimmu.2016.00487
Boiko JR, Borghesi L (2012) Hematopoiesis sculpted by pathogens: toll-like receptors and inflammatory mediators directly activate stem cells. Cytokine 57(1):1–8. https://doi.org/10.1016/j.cyto.2011.10.005
Monlish DA, Bhatt ST, Schuettpelz LG (2016) The role of toll-like receptors in hematopoietic malignancies. Front Immunol 7:390. https://doi.org/10.3389/fimmu.2016.00390
Seita J, Asakawa M, Ooehara J, Takayanagi S, Morita Y, Watanabe N, Fujita K, Kudo M, Mizuguchi J, Ema H, Nakauchi H, Yoshimoto T (2008) Interleukin-27 directly induces differentiation in hematopoietic stem cells. Blood 111(4):1903–1912. https://doi.org/10.1182/blood-2007-06-093328
Furusawa J, Mizoguchi I, Chiba Y, Hisada M, Kobayashi F, Yoshida H, Nakae S, Tsuchida A, Matsumoto T, Ema H, Mizuguchi J, Yoshimoto T (2016) Promotion of expansion and differentiation of hematopoietic stem cells by interleukin-27 into myeloid progenitors to control infection in emergency myelopoiesis. PLoS Pathog 12(3):e1005507. https://doi.org/10.1371/journal.ppat.1005507
Isaacs A, Lindenmann J (1957) Virus interference. I. The interferon. Proc R Soc Lond B Biol Sci 147(927):258–267
Binder D, Fehr J, Hengartner H, Zinkernagel RM (1997) Virus-induced transient bone marrow aplasia: major role of interferon-alpha/beta during acute infection with the noncytopathic lymphocytic choriomeningitis virus. J Exp Med 185(3):517–530
Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J, Minden M, Paterson B, Caligiuri MA, Dick JE (1994) A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 367(6464):645–648. https://doi.org/10.1038/367645a0
Riether C, Schurch CM, Ochsenbein AF (2015) Regulation of hematopoietic and leukemic stem cells by the immune system. Cell Death Differ 22(2):187–198. https://doi.org/10.1038/cdd.2014.89
Schepers K, Pietras EM, Reynaud D, Flach J, Binnewies M, Garg T, Wagers AJ, Hsiao EC, Passegue E (2013) Myeloproliferative neoplasia remodels the endosteal bone marrow niche into a self-reinforcing leukemic niche. Cell Stem Cell 13(3):285–299. https://doi.org/10.1016/j.stem.2013.06.009
Pietras EM, Lakshminarasimhan R, Techner JM, Fong S, Flach J, Binnewies M, Passegue E (2014) Re-entry into quiescence protects hematopoietic stem cells from the killing effect of chronic exposure to type I interferons. J Exp Med 211(2):245–262. https://doi.org/10.1084/jem.20131043
Haas S, Hansson J, Klimmeck D, Loeffler D, Velten L, Uckelmann H, Wurzer S, Prendergast AM, Schnell A, Hexel K, Santarella-Mellwig R, Blaszkiewicz S, Kuck A, Geiger H, Milsom MD, Steinmetz LM, Schroeder T, Trumpp A, Krijgsveld J, Essers MA (2015) Inflammation-induced emergency megakaryopoiesis driven by hematopoietic stem cell-like megakaryocyte progenitors. Cell Stem Cell 17(4):422–434. https://doi.org/10.1016/j.stem.2015.07.007
Billiau A, Matthys P (2009) Interferon-gamma: a historical perspective. Cytokine Growth Factor Rev 20(2):97–113. https://doi.org/10.1016/j.cytogfr.2009.02.004
Bach EA, Aguet M, Schreiber RD (1997) The IFN gamma receptor: a paradigm for cytokine receptor signaling. Annu Rev Immunol 15:563–591. https://doi.org/10.1146/annurev.immunol.15.1.563
Farrar MA, Schreiber RD (1993) The molecular cell biology of interferon-gamma and its receptor. Annu Rev Immunol 11:571–611. https://doi.org/10.1146/annurev.iy.11.040193.003035
Maciejewski J, Selleri C, Anderson S, Young NS (1995) Fas antigen expression on CD34+ human marrow cells is induced by interferon gamma and tumor necrosis factor alpha and potentiates cytokine-mediated hematopoietic suppression in vitro. Blood 85(11):3183–3190
Snoeck HW, Van Bockstaele DR, Nys G, Lenjou M, Lardon F, Haenen L, Rodrigus I, Peetermans ME, Berneman ZN (1994) Interferon gamma selectively inhibits very primitive CD342+ CD38− and not more mature CD34+ CD38+ human hematopoietic progenitor cells. J Exp Med 180(3):1177–1182
Kawano Y, Takaue Y, Hirao A, Abe T, Saito S, Matsunaga K, Watanabe T, Hirose M, Ninomiya T, Kuroda Y et al (1991) Synergistic effect of recombinant interferon-gamma and interleukin-3 on the growth of immature human hematopoietic progenitors. Blood 77(10):2118–2121
Brugger W, Mocklin W, Heimfeld S, Berenson RJ, Mertelsmann R, Kanz L (1993) Ex vivo expansion of enriched peripheral blood CD34+ progenitor cells by stem cell factor, interleukin-1 beta (IL-1 beta), IL-6, IL-3, interferon-gamma, and erythropoietin. Blood 81(10):2579–2584
de Bruin AM, Demirel O, Hooibrink B, Brandts CH, Nolte MA (2013) Interferon-gamma impairs proliferation of hematopoietic stem cells in mice. Blood 121(18):3578–3585. https://doi.org/10.1182/blood-2012-05-432906
Belyaev NN, Biro J, Langhorne J, Potocnik AJ (2013) Extramedullary myelopoiesis in malaria depends on mobilization of myeloid-restricted progenitors by IFN-gamma induced chemokines. PLoS Pathog 9(6):e1003406. https://doi.org/10.1371/journal.ppat.1003406
Schurch CM, Riether C, Ochsenbein AF (2014) Cytotoxic CD8+ T cells stimulate hematopoietic progenitors by promoting cytokine release from bone marrow mesenchymal stromal cells. Cell Stem Cell 14(4):460–472. https://doi.org/10.1016/j.stem.2014.01.002
Guermonprez P, Helft J, Claser C, Deroubaix S, Karanje H, Gazumyan A, Darasse-Jeze G, Telerman SB, Breton G, Schreiber HA, Frias-Staheli N, Billerbeck E, Dorner M, Rice CM, Ploss A, Klein F, Swiecki M, Colonna M, Kamphorst AO, Meredith M, Niec R, Takacs C, Mikhail F, Hari A, Bosque D, Eisenreich T, Merad M, Shi Y, Ginhoux F, Renia L, Urban BC, Nussenzweig MC (2013) Inflammatory Flt3l is essential to mobilize dendritic cells and for T cell responses during Plasmodium infection. Nat Med 19(6):730–738. https://doi.org/10.1038/nm.3197
Zhao X, Ren G, Liang L, Ai PZ, Zheng B, Tischfield JA, Shi Y, Shao C (2010) Brief report: interferon-gamma induces expansion of Lin(−)Sca-1(+)C-Kit(+) Cells. Stem Cells 28(1):122–126. https://doi.org/10.1002/stem.252
MacNamara KC, Jones M, Martin O, Winslow GM (2011) Transient activation of hematopoietic stem and progenitor cells by IFNgamma during acute bacterial infection. PLoS One 6(12):e28669. https://doi.org/10.1371/journal.pone.0028669
Umemoto T, Yamato M, Ishihara J, Shiratsuchi Y, Utsumi M, Morita Y, Tsukui H, Terasawa M, Shibata T, Nishida K, Kobayashi Y, Petrich BG, Nakauchi H, Eto K, Okano T (2012) Integrin-alphavbeta3 regulates thrombopoietin-mediated maintenance of hematopoietic stem cells. Blood 119(1):83–94. https://doi.org/10.1182/blood-2011-02-335430
Umemoto T, Matsuzaki Y, Shiratsuchi Y, Hashimoto M, Yoshimoto T, Nakamura-Ishizu A, Petrich B, Yamato M, Suda T (2017) Integrin alphavbeta3 enhances the suppressive effect of interferon-gamma on hematopoietic stem cells. EMBO J 36(16):2390–2403. https://doi.org/10.15252/embj.201796771
Belyaev NN, Brown DE, Diaz AI, Rae A, Jarra W, Thompson J, Langhorne J, Potocnik AJ (2010) Induction of an IL7-R(+)c-Kit(hi) myelolymphoid progenitor critically dependent on IFN-gamma signaling during acute malaria. Nat Immunol 11(6):477–485. https://doi.org/10.1038/ni.1869
Feng CG, Weksberg DC, Taylor GA, Sher A, Goodell MA (2008) The p47 GTPase Lrg-47 (Irgm1) links host defense and hematopoietic stem cell proliferation. Cell Stem Cell 2(1):83–89. https://doi.org/10.1016/j.stem.2007.10.007
King KY, Baldridge MT, Weksberg DC, Chambers SM, Lukov GL, Wu S, Boles NC, Jung SY, Qin J, Liu D, Songyang Z, Eissa NT, Taylor GA, Goodell MA (2011) Irgm1 protects hematopoietic stem cells by negative regulation of IFN signaling. Blood 118(6):1525–1533. https://doi.org/10.1182/blood-2011-01-328682
Ishihara K, Hirano T (2002) IL-6 in autoimmune disease and chronic inflammatory proliferative disease. Cytokine Growth Factor Rev 13(4–5):357–368
Schaper F, Rose-John S (2015) Interleukin-6: biology, signaling and strategies of blockade. Cytokine Growth Factor Rev 26(5):475–487. https://doi.org/10.1016/j.cytogfr.2015.07.004
Scheller J, Chalaris A, Schmidt-Arras D, Rose-John S (2011) The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim Biophys Acta 1813(5):878–888. https://doi.org/10.1016/j.bbamcr.2011.01.034
Maeda K, Baba Y, Nagai Y, Miyazaki K, Malykhin A, Nakamura K, Kincade PW, Sakaguchi N, Coggeshall KM (2005) IL-6 blocks a discrete early step in lymphopoiesis. Blood 106(3):879–885. https://doi.org/10.1182/blood-2005-02-0456
Reynaud D, Pietras E, Barry-Holson K, Mir A, Binnewies M, Jeanne M, Sala-Torra O, Radich JP, Passegue E (2011) IL-6 controls leukemic multipotent progenitor cell fate and contributes to chronic myelogenous leukemia development. Cancer Cell 20(5):661–673. https://doi.org/10.1016/j.ccr.2011.10.012
Zhao JL, Ma C, O’Connell RM, Mehta A, DiLoreto R, Heath JR, Baltimore D (2014) Conversion of danger signals into cytokine signals by hematopoietic stem and progenitor cells for regulation of stress-induced hematopoiesis. Cell Stem Cell 14(4):445–459. https://doi.org/10.1016/j.stem.2014.01.007
Pflanz S, Timans JC, Cheung J, Rosales R, Kanzler H, Gilbert J, Hibbert L, Churakova T, Travis M, Vaisberg E, Blumenschein WM, Mattson JD, Wagner JL, To W, Zurawski S, McClanahan TK, Gorman DM, Bazan JF, de Waal Malefyt R, Rennick D, Kastelein RA (2002) IL-27, a heterodimeric cytokine composed of EBI3 and p28 protein, induces proliferation of naive CD4 + T cells. Immunity 16(6):779–790. https://doi.org/10.1016/S1074-7613(02)00324-2
Hall AO, Silver JS, Hunter CA (2012) The Immunobiology of IL-27. Adv Immunol 115:1–44. https://doi.org/10.1016/b978-0-12-394299-9.00001-1
Mizoguchi I, Higuchi K, Mitobe K, Tsunoda R, Mizuguchi J, Yoshimoto T (2013) Interleukin-27: regulation of immune responses and disease development by a pleiotropic cytokine with pro- and anti-inflammatory properties. In: Yoshimoto T, Yoshimoto T (eds) Cytokine frontiers: regulation of immune responses in health and disease. Springer, Tokyo, pp 353–375
Yoshida H, Hunter CA (2015) The immunobiology of interleukin-27. Annu Rev Immunol 33:417–443. https://doi.org/10.1146/annurev-immunol-032414-112134
Morishima N, Owaki T, Asakawa M, Kamiya S, Mizuguchi J, Yoshimoto T (2005) Augmentation of effector CD8+ T cell generation with enhanced granzyme B expression by IL-27. J Immunol 175(3):1686–1693
Schneider R, Yaneva T, Beauseigle D, El-Khoury L, Arbour N (2011) IL-27 increases the proliferation and effector functions of human naive CD8+ T lymphocytes and promotes their development into Tc1 cells. Eur J Immunol 41(1):47–59. https://doi.org/10.1002/eji.201040804
Awasthi A, Carrier Y, Peron JP, Bettelli E, Kamanaka M, Flavell RA, Kuchroo VK, Oukka M, Weiner HL (2007) A dominant function for interleukin 27 in generating interleukin 10-producing anti-inflammatory T cells. Nat Immunol 8(12):1380–1389. https://doi.org/10.1038/ni1541
Fitzgerald DC, Zhang GX, El-Behi M, Fonseca-Kelly Z, Li H, Yu S, Saris CJ, Gran B, Ciric B, Rostami A (2007) Suppression of autoimmune inflammation of the central nervous system by interleukin 10 secreted by interleukin 27-stimulated T cells. Nat Immunol 8(12):1372–1379
Stumhofer JS, Silver JS, Laurence A, Porrett PM, Harris TH, Turka LA, Ernst M, Saris CJ, O’Shea JJ, Hunter CA (2007) Interleukins 27 and 6 induce STAT3-mediated T cell production of interleukin 10. Nat Immunol 8(12):1363–1371
Do J, Kim D, Kim S, Valentin-Torres A, Dvorina N, Jang E, Nagarajavel V, DeSilva TM, Li X, Ting AH, Vignali DAA, Stohlman SA, Baldwin WM 3rd, Min B (2017) Treg-specific IL-27Ralpha deletion uncovers a key role for IL-27 in Treg function to control autoimmunity. Proc Natl Acad Sci USA 114(38):10190–10195. https://doi.org/10.1073/pnas.1703100114
Yoshimoto T, Yoneto T, Waki S, Nariuchi H (1998) Interleukin-12-dependent mechanisms in the clearance of blood-stage murine malaria parasite Plasmodium berghei XAT, an attenuated variant of P. berghei NK65. J Infect Dis 177(6):1674–1681
Yoneto T, Waki S, Takai T, Tagawa Y, Iwakura Y, Mizuguchi J, Nariuchi H, Yoshimoto T (2001) A critical role of Fc receptor-mediated antibody-dependent phagocytosis in the host resistance to blood-stage Plasmodium berghei XAT infection. J Immunol 166(10):6236–6241
Hisada M, Kamiya S, Fujita K, Belladonna ML, Aoki T, Koyanagi Y, Mizuguchi J, Yoshimoto T (2004) Potent antitumor activity of interleukin-27. Cancer Res 64(3):1152–1156. https://doi.org/10.1158/0008-5472.CAN-03-2084
Yoshimoto T, Chiba Y, Furusawa JI, Xu M, Tsunoda R, Higuchi K, Mizoguchi I (2015) Potential clinical application of interleukin-27 as an antitumor agent. Cancer Sci. https://doi.org/10.1111/cas.12731
Mizoguchi I, Chiba Y, Furusawa JI, Xu M, Tsunoda R, Higuchi K, Yoshimoto T (2015) Therapeutic potential of interleukin-27 against cancers in preclinical mouse models. Oncoimmunology 4(10):e1042200. https://doi.org/10.1080/2162402X.2015.1042200
Chiba Y, Mizoguchi I, Furusawa J, Hasegawa H, Ohashi M, Xu M, Owaki T, Yoshimoto T (2017) Interleukin-27 exerts its antitumor effects by promoting differentiation of hematopoietic stem cells to M1 macrophages. Cancer Res. https://doi.org/10.1158/0008-5472.CAN-17-0960
Shimizu M, Shimamura M, Owaki T, Asakawa M, Fujita K, Kudo M, Iwakura Y, Takeda Y, Luster AD, Mizuguchi J, Yoshimoto T (2006) Antiangiogenic and antitumor activities of IL-27. J Immunol 176(12):7317–7324
Yoshimoto T, Morishima N, Mizoguchi I, Shimizu M, Nagai H, Oniki S, Oka M, Nishigori C, Mizuguchi J (2008) Antiproliferative activity of IL-27 on melanoma. J Immunol 180(10):6527–6535
Tormo AJ, Beaupre LA, Elson G, Crabe S, Gauchat JF (2013) A polyglutamic acid motif confers IL-27 hydroxyapatite and bone-binding properties. J Immunol 190(6):2931–2937. https://doi.org/10.4049/jimmunol.1201460
Larousserie F, Bsiri L, Dumaine V, Dietrich C, Audebourg A, Radenen-Bussiere B, Anract P, Vacher-Lavenu MC, Devergne O (2017) Frontline science: human bone cells as a source of IL-27 under inflammatory conditions: role of TLRs and cytokines. J Leukoc Biol 101(6):1289–1300. https://doi.org/10.1189/jlb.3HI0616-280R
Bronchud MH, Scarffe JH, Thatcher N, Crowther D, Souza LM, Alton NK, Testa NG, Dexter TM (1987) Phase I/II study of recombinant human granulocyte colony-stimulating factor in patients receiving intensive chemotherapy for small cell lung cancer. Br J Cancer 56(6):809–813
Gabrilove JL, Jakubowski A, Scher H, Sternberg C, Wong G, Grous J, Yagoda A, Fain K, Moore MA, Clarkson B et al (1988) Effect of granulocyte colony-stimulating factor on neutropenia and associated morbidity due to chemotherapy for transitional-cell carcinoma of the urothelium. N Engl J Med 318(22):1414–1422. https://doi.org/10.1056/NEJM198806023182202
Tamura M, Hattori K, Nomura H, Oheda M, Kubota N, Imazeki I, Ono M, Ueyama Y, Nagata S, Shirafuji N et al (1987) Induction of neutrophilic granulocytosis in mice by administration of purified human native granulocyte colony-stimulating factor (G-CSF). Biochem Biophys Res Commun 142(2):454–460
Duhrsen U, Villeval JL, Boyd J, Kannourakis G, Morstyn G, Metcalf D (1988) Effects of recombinant human granulocyte colony-stimulating factor on hematopoietic progenitor cells in cancer patients. Blood 72(6):2074–2081
Demetri GD, Griffin JD (1991) Granulocyte colony-stimulating factor and its receptor. Blood 78(11):2791–2808
Cebon J, Layton JE, Maher D, Morstyn G (1994) Endogenous haemopoietic growth factors in neutropenia and infection. Br J Haematol 86(2):265–274
Kawakami M, Tsutsumi H, Kumakawa T, Abe H, Hirai M, Kurosawa S, Mori M, Fukushima M (1990) Levels of serum granulocyte colony-stimulating factor in patients with infections. Blood 76(10):1962–1964
Sano E, Ohashi K, Sato Y, Kashiwagi M, Joguchi A, Naruse N (2007) A possible role of autogenous IFN-beta for cytokine productions in human fibroblasts. J Cell Biochem 100(6):1459–1476. https://doi.org/10.1002/jcb.21128
Fossiez F, Djossou O, Chomarat P, Flores-Romo L, Ait-Yahia S, Maat C, Pin JJ, Garrone P, Garcia E, Saeland S, Blanchard D, Gaillard C, Das Mahapatra B, Rouvier E, Golstein P, Banchereau J, Lebecque S (1996) T cell interleukin-17 induces stromal cells to produce proinflammatory and hematopoietic cytokines. J Exp Med 183(6):2593–2603
Liu F, Poursine-Laurent J, Link DC (2000) Expression of the G-CSF receptor on hematopoietic progenitor cells is not required for their mobilization by G-CSF. Blood 95(10):3025–3031
Levesque JP, Hendy J, Takamatsu Y, Simmons PJ, Bendall LJ (2003) Disruption of the CXCR4/CXCL12 chemotactic interaction during hematopoietic stem cell mobilization induced by GCSF or cyclophosphamide. J Clin Invest 111(2):187–196. https://doi.org/10.1172/JCI15994
Pelus LM, Bian H, King AG, Fukuda S (2004) Neutrophil-derived MMP-9 mediates synergistic mobilization of hematopoietic stem and progenitor cells by the combination of G-CSF and the chemokines GRObeta/CXCL2 and GRObetaT/CXCL2delta4. Blood 103(1):110–119. https://doi.org/10.1182/blood-2003-04-1115
Katayama Y, Battista M, Kao WM, Hidalgo A, Peired AJ, Thomas SA, Frenette PS (2006) Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell 124(2):407–421. https://doi.org/10.1016/j.cell.2005.10.041
Chow A, Lucas D, Hidalgo A, Mendez-Ferrer S, Hashimoto D, Scheiermann C, Battista M, Leboeuf M, Prophete C, van Rooijen N, Tanaka M, Merad M, Frenette PS (2011) Bone marrow CD169+ macrophages promote the retention of hematopoietic stem and progenitor cells in the mesenchymal stem cell niche. J Exp Med 208(2):261–271. https://doi.org/10.1084/jem.20101688
Semerad CL, Christopher MJ, Liu F, Short B, Simmons PJ, Winkler I, Levesque JP, Chappel J, Ross FP, Link DC (2005) G-CSF potently inhibits osteoblast activity and CXCL12 mRNA expression in the bone marrow. Blood 106(9):3020–3027. https://doi.org/10.1182/blood-2004-01-0272
Lieschke GJ, Grail D, Hodgson G, Metcalf D, Stanley E, Cheers C, Fowler KJ, Basu S, Zhan YF, Dunn AR (1994) Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 84(6):1737–1746
Liu F, Wu HY, Wesselschmidt R, Kornaga T, Link DC (1996) Impaired production and increased apoptosis of neutrophils in granulocyte colony-stimulating factor receptor-deficient mice. Immunity 5(5):491–501
Anasetti C, Logan BR, Lee SJ, Waller EK, Weisdorf DJ, Wingard JR, Cutler CS, Westervelt P, Woolfrey A, Couban S, Ehninger G, Johnston L, Maziarz RT, Pulsipher MA, Porter DL, Mineishi S, McCarty JM, Khan SP, Anderlini P, Bensinger WI, Leitman SF, Rowley SD, Bredeson C, Carter SL, Horowitz MM, Confer DL, Blood Marrow Transplant, Clinical Trials N (2012) Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med 367(16):1487–1496. https://doi.org/10.1056/NEJMoa1203517
Bernitz JM, Daniel MG, Fstkchyan YS, Moore K (2017) Granulocyte colony-stimulating factor mobilizes dormant hematopoietic stem cells without proliferation in mice. Blood 129(14):1901–1912. https://doi.org/10.1182/blood-2016-11-752923
Boettcher S, Gerosa RC, Radpour R, Bauer J, Ampenberger F, Heikenwalder M, Kopf M, Manz MG (2014) Endothelial cells translate pathogen signals into G-CSF-driven emergency granulopoiesis. Blood 124(9):1393–1403. https://doi.org/10.1182/blood-2014-04-570762
Ushach I, Zlotnik A (2016) Biological role of granulocyte macrophage colony-stimulating factor (GM-CSF) and macrophage colony-stimulating factor (M-CSF) on cells of the myeloid lineage. J Leukoc Biol 100(3):481–489. https://doi.org/10.1189/jlb.3RU0316-144R
Jones CV, Ricardo SD (2013) Macrophages and CSF-1: implications for development and beyond. Organogenesis 9(4):249–260. https://doi.org/10.4161/org.25676
Dai XM, Ryan GR, Hapel AJ, Dominguez MG, Russell RG, Kapp S, Sylvestre V, Stanley ER (2002) Targeted disruption of the mouse colony-stimulating factor 1 receptor gene results in osteopetrosis, mononuclear phagocyte deficiency, increased primitive progenitor cell frequencies, and reproductive defects. Blood 99(1):111–120
Sarrazin S, Mossadegh-Keller N, Fukao T, Aziz A, Mourcin F, Vanhille L, Kelly Modis L, Kastner P, Chan S, Duprez E, Otto C, Sieweke MH (2009) MafB restricts M-CSF-dependent myeloid commitment divisions of hematopoietic stem cells. Cell 138(2):300–313. https://doi.org/10.1016/j.cell.2009.04.057
Till JE, McCulloch EA, Siminovitch L (1964) A stochastic model of stem cell proliferation, based on the growth of spleen colony-forming cells. Proc Natl Acad Sci USA 51:29–36
Mossadegh-Keller N, Sarrazin S, Kandalla PK, Espinosa L, Stanley ER, Nutt SL, Moore J, Sieweke MH (2013) M-CSF instructs myeloid lineage fate in single haematopoietic stem cells. Nature 497(7448):239–243. https://doi.org/10.1038/nature12026
Kandalla PK, Sarrazin S, Molawi K, Berruyer C, Redelberger D, Favel A, Bordi C, de Bentzmann S, Sieweke MH (2016) M-CSF improves protection against bacterial and fungal infections after hematopoietic stem/progenitor cell transplantation. J Exp Med 213(11):2269–2279. https://doi.org/10.1084/jem.20151975
Dinarello CA, van der Meer JW (2013) Treating inflammation by blocking interleukin-1 in humans. Semin Immunol 25(6):469–484. https://doi.org/10.1016/j.smim.2013.10.008
Cullinan EB, Kwee L, Nunes P, Shuster DJ, Ju G, McIntyre KW, Chizzonite RA, Labow MA (1998) IL-1 receptor accessory protein is an essential component of the IL-1 receptor. J Immunol 161(10):5614–5620
Dinarello CA, Simon A, van der Meer JW (2012) Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov 11(8):633–652. https://doi.org/10.1038/nrd3800
Hestdal K, Ruscetti FW, Chizzonite R, Ortiz M, Gooya JM, Longo DL, Keller JR (1994) Interleukin-1 (IL-1) directly and indirectly promotes hematopoietic cell growth through type I IL-1 receptor. Blood 84(1):125–132
Morrissey P, Charrier K, Bressler L, Alpert A (1988) The influence of IL-1 treatment on the reconstitution of the hemopoietic and immune systems after sublethal radiation. J Immunol 140(12):4204–4210
Damia G, Komschlies KL, Futami H, Back T, Gruys ME, Longo DL, Keller JR, Ruscetti FW, Wiltrout RH (1992) Prevention of acute chemotherapy-induced death in mice by recombinant human interleukin 1: protection from hematological and nonhematological toxicities. Cancer Res 52(15):4082–4089
Smith MA, Knight SM, Maddison PJ, Smith JG (1992) Anaemia of chronic disease in rheumatoid arthritis: effect of the blunted response to erythropoietin and of interleukin 1 production by marrow macrophages. Ann Rheum Dis 51(6):753–757
Dinarello CA (2005) Blocking IL-1 in systemic inflammation. J Exp Med 201(9):1355–1359. https://doi.org/10.1084/jem.20050640
Cain D, Kondo M, Chen H, Kelsoe G (2009) Effects of acute and chronic inflammation on B-cell development and differentiation. J Invest Dermatol 129(2):266–277. https://doi.org/10.1038/jid.2008.286
Pietras EM, Mirantes-Barbeito C, Fong S, Loeffler D, Kovtonyuk LV, Zhang S, Lakshminarasimhan R, Chin CP, Techner JM, Will B, Nerlov C, Steidl U, Manz MG, Schroeder T, Passegue E (2016) Chronic interleukin-1 exposure drives haematopoietic stem cells towards precocious myeloid differentiation at the expense of self-renewal. Nat Cell Biol 18(6):607–618. https://doi.org/10.1038/ncb3346
Jovcic G, Ivanovic Z, Biljanovic-Paunovic L, Bugarski D, Stosic-Grujicic S, Milenkovic P (1996) The effect of IL-1 receptor antagonist on the proliferation of hematopoietic progenitor cells in regenerating bone marrow. Leukemia 10(3):564–569
Acknowledgements
This study was supported in part by a Grant-in-aid and the Private University Strategic Research Based Support Project from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chiba, Y., Mizoguchi, I., Hasegawa, H. et al. Regulation of myelopoiesis by proinflammatory cytokines in infectious diseases. Cell. Mol. Life Sci. 75, 1363–1376 (2018). https://doi.org/10.1007/s00018-017-2724-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-017-2724-5