Abstract
Purpose
Early diagnosis of acute kidney injury (AKI) remains a major challenge. We developed and validated AKI prediction models in adult ICU patients and made these models available via an online prognostic calculator. We compared predictive performance against serum neutrophil gelatinase-associated lipocalin (NGAL) levels at ICU admission.
Methods
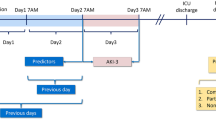
Analysis of the large multicenter EPaNIC database. Model development (n = 2123) and validation (n = 2367) were based on clinical information available (1) before and (2) upon ICU admission, (3) after 1 day in ICU and (4) including additional monitoring data from the first 24 h. The primary outcome was a comparison of the predictive performance between models and NGAL for the development of any AKI (AKI-123) and AKI stages 2 or 3 (AKI-23) during the first week of ICU stay.
Results
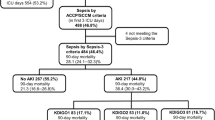
Validation cohort prevalence was 29% for AKI-123 and 15% for AKI-23. The AKI-123 model before ICU admission included age, baseline serum creatinine, diabetes and type of admission (medical/surgical, emergency/planned) and had an AUC of 0.75 (95% CI 0.75–0.75). The AKI-23 model additionally included height and weight (AUC 0.77 (95% CI 0.77–0.77)). Performance consistently improved with progressive data availability to AUCs of 0.82 (95% CI 0.82–0.82) for AKI-123 and 0.84 (95% CI 0.83–0.84) for AKI-23 after 24 h. NGAL was less discriminant with AUCs of 0.74 (95% CI 0.74–0.74) for AKI-123 and 0.79 (95% CI 0.79–0.79) for AKI-23.
Conclusions
AKI can be predicted early with models that only use routinely collected clinical information and outperform NGAL measured at ICU admission. The AKI-123 models are available at http://akipredictor.com/.
Trial registration Clinical Trials.gov NCT00512122



Similar content being viewed by others
References
Bellomo R, Kellum JA, Ronco C (2012) Acute kidney injury. Lancet 380:756–766. doi:10.1016/S0140-6736(11)61454-2
Gunst J, Vanhorebeek I, Casaer MP et al (2013) Impact of early parenteral nutrition on metabolism and kidney injury. J Am Soc Nephrol 24:995–1005. doi:10.1681/ASN.2012070732
Joannidis M, Metnitz B, Bauer P et al (2009) Acute kidney injury in critically ill patients classified by AKIN versus RIFLE using the SAPS 3 database. Intensive Care Med 35:1692–1702. doi:10.1007/s00134-009-1530-4
Nisula S, Kaukonen K-M, Vaara ST et al (2013) Incidence, risk factors and 90-day mortality of patients with acute kidney injury in Finnish intensive care units: the FINNAKI study. Intensive Care Med 39:420–428. doi:10.1007/s00134-012-2796-5
Sutherland SM, Chawla LS, Kane-Gill SL et al (2016) Utilizing electronic health records to predict acute kidney injury risk and outcomes: workgroup statements from the 15th ADQI Consensus Conference. Can J Kidney Heal Dis 3:11. doi:10.1186/s40697-016-0099-4
Chawla LS, Eggers PW, Star RA, Kimmel PL (2014) Acute kidney injury and chronic kidney disease as interconnected syndromes. N Engl J Med 371:58–66. doi:10.1056/NEJMra1214243
Ostermann M, Joannidis M (2015) Biomarkers for AKI improve clinical practice: no. Intensive Care Med 41:618–622. doi:10.1007/s00134-014-3540-0
Park M, Coca SG, Nigwekar SU et al (2010) Prevention and treatment of acute kidney injury in patients undergoing cardiac surgery: a systematic review. Am J Nephrol 31:408–418. doi:10.1159/000296277
Burns KEA, Chu MWA, Novick RJ et al (2005) Perioperative N-acetylcysteine to prevent renal dysfunction in high-risk patients undergoing CABG surgery. JAMA 294:342–350. doi:10.1001/jama.294.3.342
Billings FT, Hendricks PA, Schildcrout JS et al (2016) High-dose perioperative atorvastatin and acute kidney injury following cardiac surgery. JAMA 315:877–888. doi:10.1001/jama.2016.0548
Garg AX, Kurz A, Sessler DI et al (2014) Perioperative aspirin and clonidine and risk of acute kidney injury: a randomized clinical trial. JAMA 312:2254–2264. doi:10.1001/jama.2014.15284
Young P, Bailey M, Beasley R et al (2015) Effect of a buffered crystalloid solution vs saline on acute kidney injury among patients in the intensive care unit. JAMA 314:1701–1710. doi:10.1001/jama.2015.12334
Wilson FP, Shashaty M, Testani J et al (2015) Automated, electronic alerts for acute kidney injury: a single-blind, parallel-group, randomised controlled trial. Lancet 385:1966–1974. doi:10.1016/S0140-6736(15)60266-5
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group (2012) KDIGO clinical practice guideline for acute kidney injury. Kidney inter 2(Suppl1):1–138
Joannidis M, Druml W, Forni LG et al (2010) Prevention of acute kidney injury and protection of renal function in the intensive care unit. Intensive Care Med 36:392–411. doi:10.1007/s00134-009-1678-y
Shemin D, Dworkin LD (2011) Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for early acute kidney injury. Crit Care Clin 27:379–389. doi:10.1016/j.ccc.2010.12.003
Kashani K, Al-Khafaji A, Ardiles T et al (2013) Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit Care 17:R25. doi:10.1186/cc12503
de Geus HRH, Bakker J, Lesaffre EMEH, le Noble JLML (2011) Neutrophil gelatinase-associated lipocalin at ICU admission predicts for acute kidney injury in adult patients. Am J Respir Crit Care Med 183:907–914. doi:10.1164/rccm.200908-1214OC
Mishra J, Dent C, Tarabishi R et al (2005) Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365:1231–1238. doi:10.1016/S0140-6736(05)74811-X
Zhang Z (2015) Biomarkers, diagnosis and management of sepsis-induced acute kidney injury: a narrative review. Hear Lung Vessel 7:64–73
Bagshaw SM, Langenberg C, Haase M et al (2007) Urinary biomarkers in septic acute kidney injury. Intensive Care Med 33:1285–1296. doi:10.1007/s00134-007-0656-5
Mårtensson J, Bell M, Oldner A et al (2010) Neutrophil gelatinase-associated lipocalin in adult septic patients with and without acute kidney injury. Intensive Care Med 36:1333–1340. doi:10.1007/s00134-010-1887-4
Bell M, Larsson A, Venge P et al (2015) Assessment of cell-cycle arrest biomarkers to predict early and delayed acute kidney injury. Dis Markers 2015:1–9. doi:10.1155/2015/158658
Prowle JR (2015) Measurement of AKI biomarkers in the ICU: still striving for appropriate clinical indications. Intensive Care Med. doi:10.1007/s00134-015-3662-z
Bellazi R, Zupan B, Bellazzi R, Zupan B (2008) Predictive data mining in clinical medicine: current issues and guidelines. Int J Med Inform 77:81–97. doi:10.1016/j.ijmedinf.2006.11.006
Eagle KA, Lim MJ, Dabbous OH et al (2004) A validated prediction model for all forms of acute coronary syndrome. JAMA 291:2727–2733
Casaer MP, Mesotten D, Hermans G et al (2011) Early versus late parenteral nutrition in critically ill adults. N Engl J Med 365:506–517. doi:10.1056/NEJMoa1102662
Breiman L (2001) Random forests. Mach Learn 45:5–32. doi:10.1023/A:1010933404324
Efron B, Tibshirani R (1997) Improvements on cross-validation: the 632 + bootstrap method. J Am Stat Assoc 92:548–560. doi:10.1080/01621459.1997.10474007
Steyerberg EW, Bleeker SE, Moll HA et al (2003) Internal and external validation of predictive models: a simulation study of bias and precision in small samples. J Clin Epidemiol 56:441–447. doi:10.1016/S0895-4356(03)00047-7
Van Hoorde K, Vergouwe Y, Timmerman D et al (2014) Assessing calibration of multinomial risk prediction models. Stat Med 33:2585–2596. doi:10.1002/sim.6114
Swets J (1988) Measuring the accuracy of diagnostic systems. Science 240:1285–1293. doi:10.1126/science.3287615
Leening MJG, Steyerberg EW, Van Calster B et al (2014) Net reclassification improvement and integrated discrimination improvement require calibrated models: relevance from a marker and model perspective. Stat Med 33:3415–3418. doi:10.1002/sim.6133
Fitzgerald M, Saville BR, Lewis RJ (2015) Decision curve analysis. JAMA 313:409–410. doi:10.1001/jama.2015.37
Steyerberg EW, Vickers AJ, Cook NR et al (2010) Assessing the performance of prediction models: a framework for some traditional and novel measures. Epidemiology 21:128–138. doi:10.1097/EDE.0b013e3181c30fb2.Assessing
Pedregosa F, Varoquax G, Gramfort A et al (2011) Scikit-learn: machine learning in python. J Mach Learn Res 12:2825–2830
Concato J (1993) The risk of determining risk with multivariable models. Ann Intern Med 118:201–210. doi:10.7326/0003-4819-118-3-199302010-00009
Hoste EAJ, Bagshaw SM, Bellomo R et al (2015) Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med 41:1411–1423. doi:10.1007/s00134-015-3934-7
Cruz DN, de Cal M, Garzotto F et al (2010) Plasma neutrophil gelatinase-associated lipocalin is an early biomarker for acute kidney injury in an adult ICU population. Intensive Care Med 36:444–451. doi:10.1007/s00134-009-1711-1
Haase M, Bellomo R, Devarajan P et al (2009) Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis 54:1012–1024. doi:10.1053/j.ajkd.2009.07.020
Englberger L, Suri RM, Li Z et al (2010) Validation of clinical scores predicting severe acute kidney injury after cardiac surgery. Am J Kidney Dis 56:623–631. doi:10.1053/j.ajkd.2010.04.017
Bagshaw SM, Bennett M, Haase M et al (2010) Plasma and urine neutrophil gelatinase-associated lipocalin in septic versus non-septic acute kidney injury in critical illness. Intensive Care Med 36:452–461. doi:10.1007/s00134-009-1724-9
Zhang A, Cai Y, Wang P et al (2016) Diagnosis and prognosis of neutrophil gelatinase-associated lipocalin for acute kidney injury with sepsis: a systematic review and meta-analysis. Crit Care 20:41. doi:10.1186/s13054-016-1212-x
Vanmassenhove J, Vanholder R, Nagler E, Van Biesen W (2013) Urinary and serum biomarkers for the diagnosis of acute kidney injury: an in-depth review of the literature. Nephrol Dial Transplant 28:254–273. doi:10.1093/ndt/gfs380
Schley G, Köberle C, Manuilova E et al (2015) Comparison of plasma and urine biomarker performance in acute kidney injury. PLoS One 10:e0145042. doi:10.1371/journal.pone.0145042
Forni LG, Dawes T, Sinclair H, Cheek E, Bewick V, Dennis M et al (2013) Identifying the patient at risk of acute kidney injury: a predictive scoring system for the development of acute kidney injury in acute medical patients. Nephron-Clin Pract 123:143–150
Mehta RL, Cerdá J, Burdmann EA et al (2015) International Society of Nephrology’s 0 by 25 initiative for acute kidney injury (zero preventable deaths by 2025): a human rights case for nephrology. Lancet 385:2616–2643. doi:10.1016/S0140-6736(15)60126-X
Acknowledgements
MF receives funding from the Fonds Wetenschappelijk Onderzoek (FWO) as a PhD fellow (11Y1116 N). GM receives funding from FWO as senior clinical investigator (1846113 N). MC receives funding from the UZLeuven KOF. GVdB, through the KULeuven, receives long-term research financing via the Flemish government Methusalem–program. The authors would like to thank Romain Pirson for his input during the development of the akipredictor.com website.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Take-home message: AKI can be predicted early with models that only use routinely collected clinical information and outperform NGAL measured at ICU admission. The AKI-123 models are available at http://akipredictor.com/.
M. Flechet and F. Güiza contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Flechet, M., Güiza, F., Schetz, M. et al. AKIpredictor, an online prognostic calculator for acute kidney injury in adult critically ill patients: development, validation and comparison to serum neutrophil gelatinase-associated lipocalin. Intensive Care Med 43, 764–773 (2017). https://doi.org/10.1007/s00134-017-4678-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-017-4678-3