Abstract
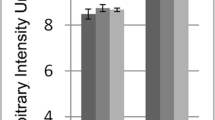
Bromate (BrO3 −), a by-product of ozonation of drinking water, induces nephrotoxicity in male rats at much lower doses than in female rats. This difference appears to be related to the development of α-2u-globulin nephropathy in males. To determine sex-dependent changes in mRNA and protein expression in the renal cortex attributable to α-2u-globulin nephropathy, we performed microarray and immunohistochemical analyses in proximal renal tubules of male and female F344 rats treated with KBrO3 for 28 days. Particular attention was paid to molecular biomarkers of renal tubular injury. Microarray analysis of male and female rats treated with BrO3 − at low doses (125 mg/L KBrO3) displayed marked sex-dependent changes in renal gene expression. The greatest differences were seen in genes encoding for cellular differentiation, apoptosis, ion transport, and cell proliferation. Differences by sex were especially prominent for the cell cycle checkpoint gene p21, the renal injury protein Kim-1, and the kidney injury and cancer biomarker protein osteopontin. Dose-related nephrotoxicity, assessed by hematoxylin and eosin staining, was greater in males compared to female rats, as was cellular proliferation, assessed by bromodeoxyuridine staining. The fraction of proximal renal cells with elevated 8-oxodeoxyguanosine (8-OH-dG) was only increased at the high dose and did not differ by sex. Dose-dependent increases in the expression of osteopontin were detected immunohistochemically only in male rats and were localized in proximal tubule cells. Similarly, BrO3 − treatment increased clusterin and Kim-1 staining in the proximal tubules; however, staining for these proteins did not differ appreciably between males and females. These data demonstrate both qualitative and quantitative differences in the response of male versus female kidneys to BrO3 −-treatment. These sex-dependent effects likely contribute to renal carcinogenesis of BrO3 − in the male rat.










Similar content being viewed by others
References
Abbas T, Dutta A (2009) p21 in cancer: intricate networks and multiple activities. Nat Rev Cancer 9(6):400–414
Ahlborn GJ, Delker DA, Roop BC et al (2009) Early alterations in protein and gene expression in rat kidney following bromate exposure. Food Chem Toxicol 47(6):1154–1160
Arai T, Kelly VP, Minowa O, Noda T, Nishimura S (2002) High accumulation of oxidative DNA damage, 8-hydroxyguanine, in Mmh/Ogg1 deficient mice by chronic oxidative stress. Carcinogenesis 23(12):2005–2010
Baldi P, Long AD (2001) A Bayesian framework for the analysis of microarray expression data: regularized t-test and statistical inferences of gene changes. Bioinformatics 17(6):509–519. doi:10.1093/bioinformatics/17.6.509
Ballmaier D, Epe B (2006) DNA damage by bromate: mechanism and consequences. Toxicology 221(2–3):166–171
Camacho L, Kelly KP, Beland FA, Gamboa da Costa G (2011) Gene expression of biomarkers of nephrotoxicity in F344 rats co-exposed to melamine and cyanuric acid for seven days. Toxicol Lett 206(2):166–171. doi:10.1016/j.toxlet.2011.07.009
Chipman JK, Parsons JL, Beddowes EJ (2006) The multiple influences of glutathione on bromate genotoxicity: implications for the dose–response relationship. Toxicology 221(2–3):187–189
de Hoon MJ, Imoto S, Nolan J, Miyano S (2004) Open source clustering software. Bioinformatics 20(9):1453–1454. doi:10.1093/bioinformatics/bth078
DeAngelo AB, George MH, Kilburn SR, Moore TM, Wolf DC (1998) Carcinogenicity of potassium bromate administered in the drinking water to male B6C3F1 mice and F344/N rats. Toxicol Pathol 26(5):587–594
Delker D, Hatch G, Allen J et al (2006) Molecular biomarkers of oxidative stress associated with bromate carcinogenicity. Toxicology 221(2–3):158–165
EPA (2002) Toxicological Review of Bromate (CAS No. 15541-45-4). In: Agency USEP (ed)
EPA US (2012) IRIS for Bromate. In. http://www.epa.gov/iris/subst/1002.htm
Geter DR, Ward WO, Knapp GW et al (2006) Kidney toxicogenomics of chronic potassium bromate exposure in F344 male rats. Transl Oncogenomics 1:33–52
Gold LS, Slone TH, Manley MB, Garfinkel GB, Ames BN (2012) Carincogen potency database (CPD). In: http://potency.berkeley.edu/chempages/BROMATE,%20POTASSIUM.html
Han C, Demetris AJ, Michalopoulos G, Shelhamer JH, Wu T (2002) 85-kDa cPLA(2) plays a critical role in PPAR-mediated gene transcription in human hepatoma cells. Am J Physiol Gastrointest Liver Physiol 282(4):G586–G597
(IARC) Iafroc (1986) Summaries and evaluations: potassium bromate (Group 2B)
Irizarry RA, Hobbs B, Collin F et al (2003) Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4(2):249–264. doi:10.1093/biostatistics/4.2.249
Kawanishi S, Murata M (2006) Mechanism of DNA damage induced by bromate differs from general types of oxidative stress. Toxicology 221(2–3):172–178
Kurokawa Y, Hayashi Y, Maekawa A, Takahashi M, Kokubo T (1982) Induction of renal cell tumors in F-344 rats by oral administration of potassium bromate, a food additive. Gann 73(2):335–338
Kurokawa Y, Hayashi Y, Maekawa A, Takahashi M, Kokubo T, Odashima S (1983) Carcinogenicity of potassium bromate administered orally to F344 rats. J Natl Cancer Inst 71(5):965–972
Kurokawa Y, Aoki S, Imazawa T, Hayashi Y, Matsushima Y, Takamura N (1985) Dose-related enhancing effect of potassium bromate on renal tumorigenesis in rats initiated with N-ethyl-N-hydroxyethyl-nitrosamine. Jpn J Cancer Res (Gann) 76(7):583–589
Kurokawa Y, Aoki S, Matsushima Y, Takamura N, Imazawa T, Hayashi Y (1986) Dose–response studies on the carcinogenicity of potassium bromate in F344 rats after long-term oral administration. J Natl Cancer Inst 77(4):977–982
Kurokawa Y, Maekawa A, Takahashi M, Hayashi Y (1990) Toxicity and carcinogenicity of potassium bromate—a new renal carcinogen. Environ Health Perspect 87:309–335
Matusan K, Dordevic G, Stipic D, Mozetic V, Lucin K (2006) Osteopontin expression correlates with prognostic variables and survival in clear cell renal cell carcinoma. J Surg Oncol 94(4):325–331
Moore MM, Chen T (2006) Mutangenicity of bromate: implications for cancer risk assessment. Toxicology 221(2–3):190–196
Rossig L, Jadidi AS, Urbich C, Badorff C, Zeiher AM, Dimmeler S (2001) Akt-dependent phosphorylation of p21(Cip1) regulates PCNA binding and proliferation of endothelial cells. Mol Cell Biol 21(16):5644–5657
Umemura T, Kurokawa Y (2006) Etiology of bromate-induced cancer and possible modes of action-studies in Japan. Toxicology 221(2–3):154–157
Umemura T, Sai K, Takagi A, Hasegawa R, Kurokawa Y (1995) A possible role for oxidative stress in potassium bromate (KBrO3) carcinogenesis. Carcinogenesis 16(3):593–597. doi:10.1093/carcin/16.3.593
Umemura T, Takagi A, Sai K, Hasegawa R, Kurokawa Y (1998) Oxidative DNA damage and cell proliferation in kidneys of male and female rats during 13-weeks exposure to potassium bromate (KBrO3). Arch Toxicol 72(5):264–269
Umemura T, Kitamura Y, Kanki K et al (2004) Dose-related changes of oxidative stress and cell proliferation in kidneys of male and female F344 rats exposed to potassium bromate. Cancer Sci 95(5):393–398
Vaidya VS, Ramirez V, Ichimura T, Bobadilla NA, Bonventre JV (2006) Urinary kidney injury molecule-1: a sensitive quantitative biomarker for early detection of kidney tubular injury. Am J Physiol Renal Physiol 290(2):F517–F529
von Gunten U, Hoigne J (1994) Bromate formation during ozonization of bromide-containing waters: interaction of ozone and hydroxyl radical reactions. Environ Sci Technol 28(7):1234–1242. doi:10.1021/es00056a009
Wolf DC, Crosby LM, George MH et al (1998) Time- and dose-dependent development of potassium bromate-induced tumors in male Fischer 344 rats. Toxicol Pathol 26(6):724–729
Xie Y, Sakatsume M, Nishi S, Narita I, Arakawa M, Gejyo F (2001) Expression, roles, receptors, and regulation of osteopontin in the kidney. Kidney Int 60(5):1645–1657
Zhang X, De Silva D, Sun B et al (2010) Cellular and molecular mechanisms of bromate-induced cytotoxicity in human and rat kidney cells. Toxicology 269(1):13–23
Acknowledgments
We thank Dr. James Bruckner for use of his lab facilities to conduct many of the in vivo studies. We also thank Dr. Robert Arnold for use of his microscope for the immunohistochemistry studies. This work was supported by the Georgia Cancer Coalition and Joseph Cotruvo & Associates LLC with grants from the Water Research Foundation Project #4042–International Ozone Assoc., Environment Agency of Abu Dhabi, Veolia Water, Metropolitan Water District of Southern Calif., Los Angeles Department of Water and Power, Singapore Public Utilities Bureau, National Water Research Institute, Walkerton Clean Water Centre, Calleguas Municipal Water District, Long Beach Water Department, and in-kind contributions of the participants. We gratefully acknowledge and thank the Water Research Foundation and all of the sponsors for their financial, technical, and administrative assistance in funding the project through which this information was discovered.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclaimer: This document has been reviewed in accordance with United States Food and Drug Administration (FDA) policy and approved for publication. Approval does not signify that the contents necessarily reflect the position or opinions of the FDA nor does mention of trade names or commercial products constitute endorsement or recommendation for use. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the FDA.
Rights and permissions
About this article
Cite this article
Kolisetty, N., Delker, D.A., Muralidhara, S. et al. Changes in mRNA and protein expression in the renal cortex of male and female F344 rats treated with bromate. Arch Toxicol 87, 1911–1925 (2013). https://doi.org/10.1007/s00204-013-1052-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-013-1052-2