Abstract
Rationale
Much evidence indicates that individuals use tobacco primarily to experience the psychopharmacological properties of nicotine. Varenicline, a partial α4β2 nicotinic acetylcholine receptor (nAChR) agonist, is effective in reducing nicotine craving and relapse in smokers, suggesting that α4β2 nAChRs may play a key role in nicotine dependence. In rats, the effect of varenicline on nicotine intake has only been studied with limited access to the drug, a model of the positive reinforcing effect of nicotine. Varenicline has not been tested on the increase in motivation to take nicotine in nicotine-dependent rats.
Objectives
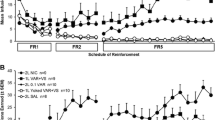
The present study evaluated the effects of varenicline on nicotine intake in rats with extended access to nicotine self-administration (23 h/day), a condition leading to the development of nicotine dependence. We hypothesized that varenicline’s effects on nicotine self-administration would be greater in rats with extended than limited access to the drug and after forced abstinence rather than during baseline self-administration.
Results
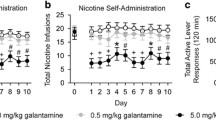
Varenicline dose-dependently decreased nicotine self-administration in rats with limited (1 h/day) and extended (23 h/day) access. Despite an increased sensitivity to the motivational effects of abstinence on nicotine intake compared with limited-access rats, varenicline was equally effective in decreasing nicotine intake in dependent rats with extended access to nicotine.
Conclusion
These results suggest that α4β2 nAChRs are critical in mediating the positive reinforcing effects of nicotine but may not be a key element underlying the negative reinforcement process responsible for the increased nicotine intake after abstinence in dependent subjects.




Similar content being viewed by others
References
Balfour DJ (1984) Nicotine and the tobacco smoking habit. In: Balfour DJK (ed) Nicotine and the tobacco smoking habit (series title: international encyclopedia of pharmacology and therapeutics, vol. 114). Pergamon, New York, pp 61–74
Benowitz NL, Jacob P 3rd (1984) Daily intake of nicotine during cigarette smoking. Clin Pharmacol Ther 35:499–504
Benwell ME, Balfour DJ, Anderson JM (1988) Evidence that tobacco smoking increases the density of (-)-[3H]nicotine binding sites in human brain. J Neurochem 50(4):1243–1247, PubMed PMID: 3346676
Breese CR, Marks MJ, Logel J, Adams CE, Sullivan B, Collins AC, Leonard S (1997) Effect of smoking history on [3H]nicotine binding in human postmortem brain. J Pharmacol Exp Ther 282(1):7–13, PubMed PMID: 9223534
Buisson B, Bertrand D (2002) Nicotine addiction: the possible role of functional upregulation. Trends Pharmacol Sci 23:130–136
Carroll FI, Yokota Y, Ma W, Lee JR, Brieaddy LE, Burgess JP, Navarro HA, Damaj MI, Martin (2008) Synthesis, nicotinic acetylcholine receptor binding, and pharmacological properties of 30-(substituted phenyl)deschloroepibatidine analogs. Bioorg Med Chem 16:746–754
Corrigall WA, Coen KM (1989) Nicotine maintains robust self-administration in rats on a limited-access schedule. Psychopharmacology 99:473–478
Corrigall WA, Franklin KB, Coen KM, Clarke PB (1992) The mesolimbic dopaminergic system is implicated in the reinforcing effects of nicotine. Psychopharmacology (Berl) 107(2–3):285–289
Corrigall WA, Coen KM, Adamson KL (1994) Self-administered nicotine activates the mesolimbic dopamine system through the ventral tegmental area. Brain Res 653:278–284
Fowler CD, Arends MA, Kenny PJ (2008) Subtypes of nicotinic acetylcholine receptors in nicotine reward, dependence, and withdrawal: evidence from genetically modified mice. Behav Pharmacol 19:461–484
Fu Y, Matta SG, Kane VB, Sharp BM (2003) Norepinephrine release in amygdala of rats during chronic nicotine self-administration: an in vivo microdialysis study. Neuropharmacology 45:514–523
George O, Ghozland S, Azar MR, Cottone P, Zorrilla EP, Parsons LH, O’Dell LE, Richardson HN, Koob GF (2007) CRF-CRF1 system activation mediates withdrawal-induced increases in nicotine self-administration in nicotine-dependent rats. Proc Natl Acad Sci USA 104(43):17198–17203, Epub 2007 Oct 5
Gonzales D, Rennard SI, Nides M, Oncken C, Azoulay S, Billing CB, Watsky EJ, Gong J, Williams KE, Reeves KR (2006) Varenicline, an α4β2 nicotinic acetylcholine receptor partial agonist, vs sustained-release bupropion and placebo for smoking cessation: a randomized controlled trial. J Am Med Assoc 296:47–55
Govind AP, Vezina P, Green WN (2009) Nicotine-induced upregulation of nicotinic receptors: underlying mechanisms and relevance to nicotine addiction. Biochem Pharmacol 78:756–765
Grottick AJ, Trube G, Corrigall WA, Huwyler J, Malherbe P, Wyler R, Higgins GA (2000) Evidence that nicotinic α7 receptors are not involved in the hyperlocomotor and rewarding effects of nicotine. J Pharmacol Exp Ther 294:1112–1119
Hughes JR, Stead LF, Lancaster T (2007) Antidepressants for smoking cessation. Cochrane Database Syst Rev 1:CD000031
Isaac PF, Rand MJ (1972) Cigarette smoking and plasma levels of nicotine. Nature 236:308–310
Jorenby DE, Hays JT, Rigotti NA, Azoulay S, Watsky EJ, Williams KE, Billing CB, Gong J, Reeves KR (2006) Efficacy of varenicline, an α4β2 nicotinic acetylcholine receptor partial agonist, vs placebo or sustained-release bupropion for smoking cessation: a randomized controlled trial. J Am Med Assoc 296:56–63
LeSage MG, Keyler DE, Collins G, Pentel PR (2003) Effects of continuous nicotine infusion on nicotine self-administration in rats: relationship between continuously infused and self-administered nicotine doses and serum concentrations. Psychopharmacology (Berl) 170(3):278–286, Epub 2003 Jul 25
Marks MJ, Burch JB, Collins AC (1983) Effects of chronic nicotine infusion on tolerance development and nicotinic receptors. J Pharmacol Exp Ther 226(3):817–825, PubMed PMID: 6887012
Marks MJ, Stitzel JA, Collins AC (1985) Time course study of the effects of chronic nicotine infusion on drug response and brain receptors. J Pharmacol Exp Ther 235(3):619–628, PubMed PMID: 4078726
Marks MJ, Pauly JR, Gross SD, Deneris ES, Hermans-Borgmeyer I, Heinemann SF, Collins AC (1992) Nicotine binding and nicotinic receptor subunit RNA after chronic nicotine treatment. J Neurosci 12(7):2765–2784, PubMed PMID: 1613557
Marubio LM, Gardier AM, Durier S, David D, Klink R, Arroyo-Jimenez MM, McIntosh JM, Rossi F, Champtiaux N, Zoli M, Changeux JP (2003) Effects of nicotine in the dopaminergic system of mice lacking the α4 subunit of neuronal nicotinic acetylcholine receptors. Eur J Neurosci 17:1329–1337
Maskos U, Molles BE, Pons S, Besson M, Guiard BP, Guilloux JP, Evrard A, Cazala P, Cormier A, Mameli-Engvall M, Dufour N, Cloez-Tayarani I, Bemelmans AP, Mallet J, Gardier AM, David V, Faure P, Granon S, Changeux JP (2005) Nicotine reinforcement and cognition restored by targeted expression of nicotinic receptors. Nature 436:103–107
Mihalak KB, Carroll FI, Luetje CW (2006) Varenicline is a partial agonist at α4β2 and a full agonist at α7 neuronal nicotinic receptors. Mol Pharmacol 70:801–805
Moreno AY, Azar MR, Warren NA, Dickerson TJ, Koob GF, Janda KD (2010) A critical evaluation of a nicotine vaccine within a self-administration behavioral model. Mol Pharm 7(2):431–441
O’Dell LE, Koob GF (2007) “Nicotine deprivation effect” in rats with intermittent 23-hour access to intravenous nicotine self-administration. Pharmacol Biochem Behav 86:346–353
Paterson NE, Markou A (2004) Prolonged nicotine dependence associated with extended access to nicotine self-administration in rats. Psychopharmacology 173:64–72
Picciotto MR, Zoli M, Rimondini R, Lena C, Marubio LM, Merlo-Pich E, Fuxe K, Changeux JP (1998) Acetylcholine receptors containing the β2 subunit are involved in the reinforcing properties of nicotine. Nature 391:173–177
Picciotto MR, Addy NA, Mineur YS, Brunzell DH (2008) It is not “either/or”: activation and desensitization of nicotinic acetylcholine receptors both contribute to behaviors related to nicotine addiction and mood. Prog Neurobiol 84:329–342
Pontieri FE, Tanda G, Orzi R, Di Chiara G (1996) Effects of nicotine on the nucleus accumbens and similarity to those of addictive drugs. Nature 382:255–257
Rollema H, Chambers LK, Coe JW, Glowa J, Hurst RS, Lebel LA, Lu Y, Mansbach RS, Mather RJ, Rovetti CC, Sands SB, Schaeffer E, Schulz DW, Tingley FD 3rd, Williams KE (2007a) Pharmacological profile of the α4β2 nicotinic acetylcholine receptor partial agonist varenicline, an effective smoking cessation aid. Neuropharmacology 52:985–994
Rollema H, Coe JW, Chambers LK, Hurst RS, Stahl SM, Williams KE (2007b) Rationale, pharmacology and clinical efficacy of partial agonists of α4β2 nACh receptors for smoking cessation. Trends Pharmacol Sci 28:316–325
Schwartz RD, Kellar KJ (1983) Nicotinic cholinergic receptor binding sites in the brain: regulation in vivo. Science 220(4593):214–216, PubMed PMID: 6828889
Silagy C, Lancaster T, Stead L, Mant D, Fowler G (2004) Nicotine replacement therapy for smoking cessation. Cochrane Database Syst Rev 3:CD000146
Stolerman IP (1991) Behavioural pharmacology of nicotine: multiple mechanisms. Br J Addict 86:533–536
Tonstad S, Tønnesen P, Hajek P, Williams KE, Billing CB, Reeves KR (2006) Effect of maintenance therapy with varenicline for smoking cessation: a randomized controlled trial. J Am Med Assoc 296:64–71
Valentine JD, Hokanson JS, Matta SG, Sharp BM (1997) Self-administration in rats allowed unlimited access to nicotine. Psychopharmacology 133:300–304
Walters CL, Brown S, Changeux JP, Martin B, Damaj MI (2006) The β2 but not α7 subunit of the nicotinic acetylcholine receptor is required for nicotine-conditioned place preference in mice. Psychopharmacology 184:339–344
Zaniewska M, McCreary AC, Stefański R, Przegaliński E, Filip M (2008) Effect of varenicline on the acute and repeated locomotor responses to nicotine in rats. Synapse 62:935–939
Acknowledgments
This is publication number 20551 from The Scripps Research Institute. This work was supported by the Tobacco-Related Disease Research Program (TRDRP) from the State of California (grant 17RT-0095), the Pearson Center for Alcoholism and Addiction Research, and the National Institute on Drug Abuse (DA12001 and DA023597)). The authors thank Robert Lintz, Shayla Vu, Yanabel Grant, and Molly Brennan for their technical assistance and Taryn Grieder for helpful discussions. We also thank Michael Arends for his editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
George, O., Lloyd, A., Carroll, F.I. et al. Varenicline blocks nicotine intake in rats with extended access to nicotine self-administration. Psychopharmacology 213, 715–722 (2011). https://doi.org/10.1007/s00213-010-2024-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-010-2024-3