Abstract
Rationale
Novel stimuli paired with exposure to addictive drugs can elicit approach through Pavlovian learning. While such approach behavior, or sign tracking, has been documented for cocaine and alcohol, it has not been shown to occur with opiate drugs like heroin. Most Pavlovian conditioned approach paradigms use an operandum as the sign, so that sign tracking can be easily automated.
Objectives
We were interested in assessing whether approach behavior occurs to an audiovisual cue paired with an intravenous heroin infusion. If so, would this behavior exhibit characteristics of other Pavlovian conditioned behaviors, such as extinction and spontaneous recovery?
Methods
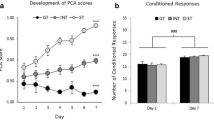
Rats were repeatedly exposed to an audiovisual cue, similar to that used in standard self-administration models, along with an intravenous heroin infusion. Sign tracking was measured in an automated fashion by analyzing motion pixels within the cue zone during each cue presentation.
Results
We were able to observe significant sign tracking after only five pairings of the conditioned stimulus (CS) with the unconditioned stimulus (US). This behavior rapidly extinguished over 2 days, but exhibited pronounced spontaneous recovery 3 weeks later.
Conclusions
We conclude that sign tracking measured by these methods exhibits all the characteristics of a classically conditioned behavior. This model can be used to examine the Pavlovian component of drug memories, alone, or in combination with self-administration methods.




Similar content being viewed by others
References
Bardo MT, Neisewander JL (1986) Single-trial conditioned place preference using intravenous morphine. Pharmacol Biochem Behav 25:1101–5
Bastle RM, Kufahl PR, Turk MN, Weber SM, Pentkowski NS, Thiel KJ, Neisewander JL (2012) Novel cues reinstate cocaine-seeking behavior and induce Fos protein expression as effectively as conditioned cues. Neuropsychopharmacology 37:2109–20
Childress AR, McLellan AT, O'Brien CP (1986) Abstinent opiate abusers exhibit conditioned craving, conditioned withdrawal and reductions in both through extinction. Br J Addict 81:655–60
Cunningham CL, Patel P (2007) Rapid induction of Pavlovian approach to an ethanol-paired visual cue in mice. Psychopharmacology (Berl) 192:231–41
Di Ciano P, Everitt BJ (2003) Differential control over drug-seeking behavior by drug-associated conditioned reinforcers and discriminative stimuli predictive of drug availability. Behav Neurosci 117:952–60
Di Ciano P, Everitt BJ (2004) Conditioned reinforcing properties of stimuli paired with self-administered cocaine, heroin or sucrose: implications for the persistence of addictive behaviour. Neuropharmacology 47(Suppl 1):202–13
Drummond DC (2000) What does cue-reactivity have to offer clinical research? Addiction 95(Suppl 2):S129–44
Fuchs RA, Lasseter HC, Ramirez DR, Xie X (2008) Relapse to drug seeking following prolonged abstinence: the role of environmental stimuli. Drug Discov Today Dis Models 5:251–258
Kearns DN, Weiss SJ (2011) A comparison of explicitly unpaired treatment and extinction: recovery of sign-tracking within a context renewal design. Behav Processes 86:364–7
Kearns DN, Weiss SJ (2004) Sign-tracking (autoshaping) in rats: a comparison of cocaine and food as unconditioned stimuli. Learn Behav 32:463–76
Krank MD (2003) Pavlovian conditioning with ethanol: sign-tracking (autoshaping), conditioned incentive, and ethanol self-administration. Alcohol Clin Exp Res 27:1592–8
Krank MD, O'Neill S, Squarey K, Jacob J (2008) Goal- and signal-directed incentive: conditioned approach, seeking, and consumption established with unsweetened alcohol in rats. Psychopharmacology (Berl) 196:397–405
Le Merrer J, Faget L, Matifas A, Kieffer BL (2012) Cues predicting drug or food reward restore morphine-induced place conditioning in mice lacking delta opioid receptors. Psychopharmacology (Berl) 223:99–106
Lecca D, Valentini V, Cacciapaglia F, Acquas E, Di Chiara G (2007) Reciprocal effects of response contingent and noncontingent intravenous heroin on in vivo nucleus accumbens shell versus core dopamine in the rat: a repeated sampling microdialysis study. Psychopharmacology (Berl) 194:103–16
Lee RS, Criado JR, Koob GF, Henriksen SJ (1999) Cellular responses of nucleus accumbens neurons to opiate-seeking behavior: I. Sustained responding during heroin self-administration Synapse 33:49–58
Marissen MA, Franken IH, Blanken P, van den Brink W, Hendriks VM (2007) Cue exposure therapy for the treatment of opiate addiction: results of a randomized controlled clinical trial. Psychother Psychosom 76:97–105
Marissen MA, Franken IH, Waters AJ, Blanken P, van den Brink W, Hendriks VM (2006) Attentional bias predicts heroin relapse following treatment. Addiction 101:1306–12
McClernon FJ, Hiott FB, Liu J, Salley AN, Behm FM, Rose JE (2007) Selectively reduced responses to smoking cues in amygdala following extinction-based smoking cessation: results of a preliminary functional magnetic resonance imaging study. Addict Biol 12:503–12
Price KL, Saladin ME, Baker NL, Tolliver BK, DeSantis SM, McRae-Clark AL, Brady KT (2010) Extinction of drug cue reactivity in methamphetamine-dependent individuals. Behav Res Ther 48:860–5
Quirk GJ (2002) Memory for extinction of conditioned fear is long-lasting and persists following spontaneous recovery. Learn Mem 9:402–7
Rescorla RA (2004) Spontaneous recovery. Learn Mem 11:501–9
Sanchez H, Quinn JJ, Torregrossa MM, Taylor JR (2010) Reconsolidation of a cocaine-associated stimulus requires amygdalar protein kinase A. J Neurosci 30:4401–7
Shippenberg TS, Elmer GI (1998) The neurobiology of opiate reinforcement. Crit Rev Neurobiol 12:267–303
Sinha R, Li CS (2007) Imaging stress- and cue-induced drug and alcohol craving: association with relapse and clinical implications. Drug Alcohol Rev 26:25–31
Su ZI, Wenzel J, Baird R, Ettenberg A (2011) Comparison of self-administration behavior and responsiveness to drug-paired cues in rats running an alley for intravenous heroin and cocaine. Psychopharmacology (Berl) 214:769–78
Torregrossa MM, Sanchez H, Taylor JR (2010) D-cycloserine reduces the context specificity of pavlovian extinction of cocaine cues through actions in the nucleus accumbens. J Neurosci 30:10526–33
Uslaner JM, Acerbo MJ, Jones SA, Robinson TE (2006) The attribution of incentive salience to a stimulus that signals an intravenous injection of cocaine. Behav Brain Res 169:320–4
Wise RA (1989) Opiate reward: sites and substrates. Neurosci Biobehav Rev 13:129–33
Yager LM, Robinson TE (2013) A classically conditioned cocaine cue acquires greater control over motivated behavior in rats prone to attribute incentive salience to a food cue. Psychopharmacology (Berl) 226:217–28
Acknowledgments
The authors thank Jasper A. Heinsbroek for editing the Supplemental Video and Dr. Matthew W. Feltenstein for helpful advice on the use of the backpack catheters.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
This video illustrates the acquisition of sign-tracking behavior from day 1 of Conditioning to day 5. The video has been edited to show only the "cue on" periods ±10 s before and after the cue. Sign tracking is absent on day 1, but evident in all four rats from this quad unit on day 5. Bouts of sign tracking are indicated by white arrows. (MP4 31427 kb)
Rights and permissions
About this article
Cite this article
Peters, J., De Vries, T.J. Pavlovian conditioned approach, extinction, and spontaneous recovery to an audiovisual cue paired with an intravenous heroin infusion. Psychopharmacology 231, 447–453 (2014). https://doi.org/10.1007/s00213-013-3258-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-013-3258-7