Abstract
Purpose
The aim of the review was to provide an overview of studies investigating the pharmacokinetics of exogenous melatonin in humans and if possible, to provide recommendations for clinical use.
Methods
The review was conducted in accordance to PRISMA guidelines. A systematic literature search was performed in PubMed and Embase databases. The pharmacokinetic variables included maximal plasma/serum concentration (Cmax), time to maximal plasma/serum concentration (Tmax), elimination half-life (T1/2), area-under-the-curve plasma/serum concentrations (AUC), clearance (Cl), volume of distribution (VD), and bioavailability.
Results
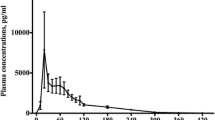
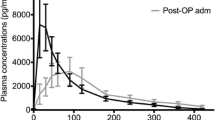
The literature search identified 392 records. Twenty-two studies were included in the review. Melatonin dosages varied between 0.3 and 100 mg and were administered either orally or intravenously. Cmax ranged from 72.1 (10 ml/h; 0.02 mg, IV) to 101,163 pg/ml (100 mg, oral). Tmax ranged between 15 (2 mg) and 210 min (10 mg). T1/2 ranged from 28 (0.005 mg, IV) to 126 min (4 mg, oral), whereas AUC ranged between 5400 (0.005 mg, IV) and 6.56 × 1010 pg/ml × min (1 mg, oral). Cl ranged from 0.97 (0.005 mg, IV) to 132.50 L/min (6 mg, oral), whereas VD ranged between 35 (0.005 mg, IV) and 1602 L (4 mg, oral). Bioavailability of oral melatonin ranged from 9 to 33 %. Pharmacokinetics was affected by age, caffeine, smoking, oral contraceptives, feeding status, and fluvoxamine. Critically ill patients displayed accelerated absorption and compromised elimination.
Conclusions
Despite methodological differences between the included studies, Tmax was approximately 50 min following oral immediate-release formulations of melatonin. T1/2 was 45 min in both administration routes. Cmax, AUC, Cl, and VD varied extensively between studies. Bioavailability of oral melatonin was approximately 15 %.

Similar content being viewed by others
References
Brzezinski A (1997) Melatonin in humans. N Engl J Med 336:186–195
Andersen LPH, Werner MU, Rosenberg J, Gögenur I (2014) A systematic review of peri-operative melatonin. Anaesthesia 69:1163–1171
Bourne RS, Mills GH, Minelli C (2008) Melatonin therapy to improve nocturnal sleep in critically ill patients: encouraging results from a small randomized controlled trail. Crit Care 12:R52
Borazan H, Tuncer S, Yalcin N, Erol A, Otelcioglu S (2010) Effects of preoperative oral melatonin medication on postoperative analgesia, sleep quality, and sedation in patients undergoing elective prostatectomy: a randomized clinical trial. J Anesth 24:155–160
Caumo W, Torres F, Moreia NL, Auzani JAS, Monteiro CA, Londero G, Ribeiro DFM, Hidalgo MPL (2007) The clinical impact of preoperative melatonin on postoperative outcomes in patients undergoing abdominal hysterectomy. Anesth Analg 105:1263–1271
Gitto E, Romeo C, Reiter RJ, Impellizzeri P, Pesce S, Basile M, Antonuccio P, Trimarchi G, Gentile C, Barberi I, Zuccarello B (2004) Melatonin reduces oxidative stress in surgical neonates. J Pediatr Surg 39:184–189
Kücükakin B, Lykkesfeldt J, Nielsen HJ, Reiter RJ, Rosenberg J, Gögenur I (2008) Utility of melatonin to treat surgical stress after major vascular surgery—a safety study. J Pineal Res 44:426–431
DeMuro RL, Nafziger AN, Blask DE, Menhinick AM, Bertino JS (2000) The absolute bioavailability of oral melatonin. J Clin Pharmacol 40:781–784
Di WL, Kadva A, Johnston A, Silman R (1997) Variable bioavailability of oral melatonin. N Engl J Med 336:1028–1029
Moher D, Liberati A, Tetzlaff J, Altman DG (2010) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8:336–341
Zhdanova IV, Wurtman RJ, Balcioglu A, Kartashov AI, Lynch HJ (1998) Endogenous melatonin levels and the fate of exogenous melatonin: age effects. J Gerontol 53:B293–B298
Cavallo A, Ritschel WA (1996) Pharmacokinetics of melatonin in human sexual maturation. J Clin Endocrinol Metab 81:1882–1886
Vakkuri O, Leppäluoto J, Kauppila A (1985) Oral administration and distribution of melatonin in human serum, saliva and urine. Life Sci 37:489–495
Shirakawa SI, Tsuchiya S, Tsutsumi Y, Kotorii T, Uchimura N, Sakamoto T, Yamada S (1998) Time course of saliva and serum melatonin levels after ingestion of melatonin. Psychiatry Clin Neurosci 52:266–267
Waldhauser F, Waldhauser M, Lieberman HR, Deng MH, Lynch HJ, Wurtman RJ (1984) Bioavailability of oral melatonin in humans. Neuroendocrinology 39:307–311
Mistraletti G, Sabbatini G, Taverna M, Figini MA, Umbrello M, Magni P, Ruscica M, Dozio E, Esposti R, DeMartini G, Fraschini F, Rezzani R, Reiter RJ, Iapichino G (2010) Pharmacokinetics of orally administered melatonin in critically ill patients. J Pineal Res 48:142–147
Härtter S, Nordmark A, Rose DM, Bertilsson L, Tybring G, Laine K (2003) Effects of caffeine intake on the pharmacokinetics of melatonin, a probe drug for CYP1A2 activity. Br J Clin Pharmacol 56:679–682
Mallo C, Zaîdan R, Galy G, Vermeulen E, Brun J, Chazot G, Claustrat B (1990) Pharmacokinetics of melatonin in man after intravenous infusion and bolus injection. Eur J Clin Pharmacol 38:297–301
Ursing C, Bhar CV, Brismar K, Röjdmark S (2005) Influence of cigarette smoking on melatonin levels in man. Eur J Clin Pharmacol 61:197–201
Fourtillan JB, Brisson AM, Gobin P, Ingrand I, Decourt JPH, Girault J (2000) Bioavailability of melatonin in humans after day-time administration of D7 Melatonin. Biopharm Drug Dispos 21:15–22
Gooneratne NS, Edwards AYZ, Zhou C, Cuellar N, Grandner MA, Barrett JS (2012) Melatonin pharmacokinetics following two different oral surge-sustained release doses in older adults. J Pineal Res 52:437–445
Markantonis SL, Tsakalozou E, Paraskeva A, Staikou C, Fassoulaki A (2008) Melatonin pharmacokinetics in premenopausal and postmenopausal healthy female volunteers. J Clin Pharmacol 48:240–245
Hilli J, Korhonen T, Turpeinen M, Hokkanen J, Mattila S, Laine K (2008) The effect of oral contraceptives on the pharmacokinetics of melatonin in healthy subjects with CYP1A2 g.-163C > A polymorphism. J Clin Pharmacol 48:986–994
Le Bars D, Thivolle P, Vitte PA, Bojkowski C, Chazot G, Arendt J, Frackowiak RSJ, Claustrat B (1991) PET and plasma pharmacokinetic studies after bolus intravenous administration of [11C]melatonin in humans. Nucl Med Biol 18:357–362
Aldhous M, Franey C, Wright J, Arendt J (1985) Plasma concentrations of melatonin in man following oral absorption of different preparations. J Clin Pharmacol 19:517–521
López-Gamboa M, Canales-Gómez JS, Castro Sandoval TJ, Tovar EN, Mejía MA, Baltazar MAM, Palma-Aguirre JA (2010) Bioavailability of long acting capsules of melatonin in Mexican healthy volunteers. J Bioequiv Availab 2:116–119
Proietti S, Carlomagno G, Dinicola S, Bizzarri M (2014) Soft gel capsules improve melatonin’s bioavailability in humans. Expert Opin Drug Metab Toxicol 10:1193–1198
Hoffmann H, Dittgen M, Hoffmann A, Bartsch C, Breitbarth H, Timpe C, Farker K, Schmidt U, Mellinger U, Zimmermann H, Gräser T, Oettel M (1998) Evaluation of an oral pulsatile delivery system for melatonin in humans. Pharmazie 53:462–466
Härrter S, Grözinger M, Wiegmann H, Röschke J, Hiemke C (2000) Increased bioavailability of oral melatonin after fluvoxamine coadministration. Clin Pharmacol Ther 67:1–6
Tan DX, Manchester LC, Terron MP, Flores LJ, Reiter RJ (2007) One molecule, many derivatives: a never-ending interaction of melatonin with reactive oxygen and nitrogen species? J Pineal Res 42:28–42
Higgins JPT, Green S (eds) (2011) Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration. http://www.cochrane-handbook.org. Accessed 16 Dec 2014
Jadad AR, Moore RA, Carrol D, Jenkinson C, Reynolds JM, Gavaghan DJ, McQuay HJ (1996) Assessing the quality of reports of randomized clinical trails: is blinding necessary? Contemp Clin Trails 17:1–12
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harpsøe, N.G., Andersen, L.P.H., Gögenur, I. et al. Clinical pharmacokinetics of melatonin: a systematic review. Eur J Clin Pharmacol 71, 901–909 (2015). https://doi.org/10.1007/s00228-015-1873-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-015-1873-4