Abstract
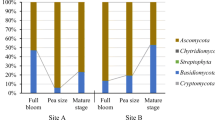
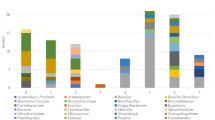
In this work, we studied the ecological interactions between grape berry microorganisms and Drosophila sp. flies involved in sour rot disease during grape ripening. After veráison the total microbial counts of grape berries affected by sour rot increased from about 2 log CFU/g of berries to more than 7 log CFU/g. Berry damage provoked a clear shift in yeast diversity from basidiomycetes to ascomycetous fermentative species. The latter were mostly Pichia terricola, Hanseniaspora uvarum, Candida zemplinina, and Zygoascus hellenicus. However, these species were not able to produce the metabolites characteristic of sour rot (gluconic and acetic acids) in inoculated berries. On the contrary, the acetic acid bacteria Gluconacetobacter saccharivorans produced high levels of these acids, mainly when berries were incubated in the presence of the insect Drosophila sp. Sour rot was not observed when grape bunches were physically separated from insects, even when berries were artificially injured. The wounds made in berry skin healed in the absence of insects, thus preventing the development of sour rot. Therefore, in the vineyard, the induction of sour rot depends on the contamination of wounded berries by a microbial consortium—yeasts and acetic acid bacteria—transported by drosophilid insects which disseminate sour rot among damaged berries. In the absence of these insects, plant defense mechanisms are effective and lead to skin healing, preventing disease spread. Thus, we showed that Drosophila sp. act as a vector for microorganisms associated with grape sour rot disease.


Similar content being viewed by others
References
Mortimer R, Polsinelli M (1999) On the origins of wine yeast. Res Microbiol 150:199–204
Buchanan GA, Amos TG (1992) Grape pests. In: Coombe BG, Dry PR (eds) Viticulture, vol 2. Winetitles, Adelaide, pp 209–231
Phaff HJ, Starmer WT (1987) Yeasts associated with plants, insects and soils. In: Rose AH, Harrison JS (eds) The yeasts, vol 1, 2 edn. Academic, New York, pp 123–180
Gilbert DG (1980) Dispersal of yeasts and bacteria by Drosophila in a temperate forest. Oecologia 46:135–137
Starmer WT, Fogleman JC (1986) Coadaptation of Drosophila and yeasts in their natural habitat. J Chem Ecol 12:1037–1055
Crotti E, Rizzi A, Chouaia B, Ricci I, Favia G, Alma A, Sacchi L, Bourtzis K, Mandrioli M, Cherif A, Bandi C, Daffonchio D (2010) Acetic acid bacteria, newly emerging symbionts of insects. Appl Environ Microbiol 76:6963–6970
Corby-Harris V, Pontaroli AC, Shimkets LJ, Bennetzen JL, Habel KE, Promislow DEL (2007) Geographical distribution and diversity of bacteria associated with natural populations of Drosophila melanogaster. Appl Environ Microbiol 73:3470–3479
Cox CR, Gilmore MS (2007) Native microbial colonization of Drosophila melanogaster and its use as a model of Enterococcus faecalis pathogenesis. Infect Immun 75:1565–1576
Ren C, Webster P, Finkel SE, Tower J (2007) Increased internal and external bacterial load during Drosophila aging without life-span trade-off. Cell Metabol 6:144–152
Roh SW, Nam Y-D, Chang H-W, Kim K-H, Kim M-S, Ryu J-H, Kim S-H, Lee W-J, Bae J-W (2008) Phylogenetic characterization of two novel commensal bacteria involved with innate immune homeostasis in Drosophila melanogaster. Appl Environ Microbiol 74:6171–6177
Ryu J-H, Kim S-H, Lee H-Y, Bai JY, Nam Y-D, Bae J-W, Lee DG, Shin SC, Ha E-M, Lee W-J (2008) Innate immune homeostasis by the homeobox gene caudal and commensal-gut mutualism in Drosophila. Science 319:777–782
Bisiach M, Minervini G, Zerbetto F (1986) Possible integrated control of grapevine sour rot. Vitis 25:118–128
Cantoni A (1984) Osservazioni sulla distribuizione di Drosophila fasciata Mg. e sulla sua correlazione con il marciume acido dell’uva nei vigneti lombardi. Tesi di laurea, Universitàdi Milano
Wolf TK, Zoecklein BW, Cook MK, Cottingham CK (1990) Shoot topping and ethephon effects on white Riesling grapes and grapevines. Am J Enol Vitic 41:330–341
Zoecklein BW, Wolf TK, Duncan NW, Judge JM, Cook MK (1992) Effects of fruit zone leaf removal on yield, fruit composition, and fruit rot incidence of Chardonnay and white Riesling (Vitis vinifera L.) grapes. Am J Enol Vitic 43:139–148
Barata A, Campo E, Malfeito-Ferreira M, Vl L, Cacho J, Ferreira V (2011) Analytical and sensorial characterization of the aroma of wines produced with sour rotten grapes using GC-O and GC-MS: identification of key aroma compounds. J Agric Food Chem 59:2543–2553
Barata A, Pais A, Malfeito-Ferreira M, Loureiro V (2011) Influence of sour rotten grapes on the chemical composition and quality of grape must and wine. Eur Food Res Technol 233:183–194
Barata A, González S, Malfeito-Ferreira M, Querol A, Loureiro V (2008) Sour rot-damaged grapes are sources of wine spoilage yeasts. FEMS Yeast Res 8:1008–1017
Barata A, Seborro F, Belloch C, Malfeito-Ferreira M, Loureiro V (2008) Ascomycetous yeast species recovered from grapes damaged by honeydew and sour rot. J Appl Microbiol 104:1182–1191
Gravot E, Blancard D, Fermaud M, Lonvaud A, Joyeux A (2001) La Pourriture acide. I. Étiologie: recherché de causes de cette pourriture dans le vignoble bordelaise. Phytoma 543:36–39
Blancard D, Gravot E, Jailloux F, Fermaud M (2000) Etiology of sour rot in vineyards located in south-west of France. IOBC/wprs Bulletin “Integrated Control in Viticulture” 23:51-54
Fermaud M, Gravot E, Blancard D (2002) La Pourriture acide dans le vignoble bordelais. II. Vection par les drosophiles des micro-organismes pathogènes. Phytoma 547:41–44
Rodrigues F, Goncalves G, Pereira-da-Silva S, Malfeito-Ferreira M, Loureiro V (2001) Development and use of a new medium to detect yeasts of the genera Dekkera/Brettanomyces. J Appl Microbiol 90:588–599
Schuller D, Côrte-Real M, Leão C (2000) A differential medium for the enumeration of the spoilage yeast Zygosaccharomyces bailii in wine. J Food Protect 63:1570–1575
FOSS (2002) GrapeScan calibration. Must-sanitary state, Application Note 212, Issue 1GB. Foss Electric, Hilleroed
Malfeito-Ferreira M, Tareco M, Loureiro V (1997) Fatty acid profiling: a feasible typing system to trace yeast contamination in wine bottling plants. Int J Food Microbiol 38:143–155
Esteve-Zarzoso B, Belloch C, Uruburu F, Querol A (1999) Identification of yeasts by RFLP analysis of the 5.8S rRNA gene and the two ribosomal internal transcribed spacers. Int J Syst Bacteriol 49:329–337
Cryer DR, Ecclesmall R, Marmur J (1975) Isolation of yeast DNA. In: Prescott DM (ed) Methods in cell biology, vol 12. Academic, New York, p 39
Hall T (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/NT. Nucleic Acids Symposium Series 41:95–98
Smith MT (1998) Hanseniaspora Zikes. In: Kurtzman CP, Fell JW (eds) The yeasts, a taxonomic study. Elsevier, Amsterdam, pp 214–220
Cadez N, Raspor P, de Cock AWAM, Boekhout T, Smith MT (2002) Molecular identification and genetic diversity within species of the genera Hanseniaspora and Kloeckera. FEMS Yeast Res 1:279–289
Nisiotou AA, Nychas GJE (2007) Yeast populations residing on healthy or Botrytis-infected grapes from a vineyard in Attica, Greece. Appl Environ Microb 73:2765–2768
Kurtzman CP (2003) Phylogenetic circumscription of Saccharomyces, Kluyveromyces and other members of the Saccharomycetaceae, and the proposal of the new genera Lachancea, Nakaseomyces, Naumovia, Vanderwaltozyma and Zygotorulaspora. FEMS Yeast Res 4:233–245
Kurtzman CP, Fell JW (eds) (1998) The yeasts, a taxonomic study. Elsevier, Amsterdam
Sipiczki M (2004) Species identification and comparative molecular and physiological analysis of Candida zemplinina and Candida stellata. J Basic Microbiol 44:471–479
González Á, Guillamón JM, Mas A, Poblet M (2006) Application of molecular methods for routine identification of acetic acid bacteria. Int J Food Microbiol 108:141–146
Ruiz A, Poblet M, Mas A, Guillamon J (2000) Identification of acetic acid bacteria by RFLP of PCR-amplified 16S rDNA and 16S–23S rDNA intergenic spacer. Int J Syst Evol Microbiol 50:1981–1987
Fonseca Á, Inácio J (2006) Phylloplane Yeasts. In: Péter G, Rosa C (eds) Biodiversity and ecophysiology of yeasts. The yeast handbook. Springer, Berlin, pp 263–301
Fermaud M, Gravot E, Blancard D, Jailloux F, Stockel J (2000) Association of Drosophilae with microorganisms in Bordeaux vineyards affected by sour rot. IOBC/wprs Bulletin “Integrated Control in Viticulture” 23:55-58
Barata A, Malfeito-Ferreira M, Loureiro V (2012) Changes in sour rotten grape berry microbiota during ripening and wine fermentation. Int J Food Microbiol. doi:10.1016/j.ijfoodmicro.2011.12.029
West AS (1961) Chemical attractants for adult Drosophila species. J Econ Entomol 54:677–681
Hoffmann AA (1985) Interspecific variation in the response of Drosophila to chemicals and fruit odours in a wind tunnel. Aust J Zool 33:451–460
Zhu J, Park K-C, Baker TC (2003) Identification of odors from overripe mango that attract vinegar flies Drosophila melanogaster. J Chem Ecol 29:899–909
Stensmyr MC, Giordano E, Balloi A, Angioy A-M, Hansson BS (2003) Novel natural ligands for Drosophila olfactory receptor neurones. J Exp Biol 206:715–724
Wightman F, Lighty DL (1982) Identification of phenylacetic acid as a natural auxin in the shoots of higher plants. Physiol Plantarum 55:17–24
Cossé AA, Bartelt RJ, Weaver DK, Zilkowski BW (2002) Pheromone components of the wheat stem sawfly: identification, electrophysiology, and field bioassay. J Chem Ecol 28:407–423
Acknowledgments
This work was partially funded by Portuguese Science and Technology Foundation (FCT) and by POCI 2010, participated by the European fund FEDER under the projects POCI/AGR/56771/2004 and PTDC/AGR-ALI/101393/2008. The authors gratefully thank the Analytical Services of Adega Cooperativa de Borba winery for the grape musts FTIR analyses. A. Barata was the recipient of a PhD grant (Ref. SFRH/BD/28451/2006) from the FCT.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 367 kb)
Rights and permissions
About this article
Cite this article
Barata, A., Santos, S.C., Malfeito-Ferreira, M. et al. New Insights into the Ecological Interaction Between Grape Berry Microorganisms and Drosophila Flies During the Development of Sour Rot. Microb Ecol 64, 416–430 (2012). https://doi.org/10.1007/s00248-012-0041-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-012-0041-y