Abstract
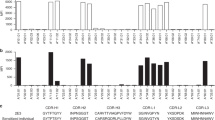
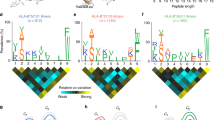
HLA-B*0801 is unique among HLA-B allotypes in having dominant amino acid anchors at positions 3 and 5 of the peptide-binding motif. HLA-B*0802 is a variant of HLA-B*0801 in which the Bw6 sequence motif is replaced by a Bw4 sequence motif. This change, involving substitutions at positions 77, 80, 81, 82, and 83 of the B*08 heavy chain, is probably the result of a single evolutionary event of interallelic conversion. Moreover, the difference between B*0802 and B*0801 is sufficient to stimulate a cytotoxic T-cell response. To assess further the functional impact of the Bw4 motif on a B8 background, we compared the peptide-binding specificity of the B*0801 and B*0802 allotypes by sequencing the mixture of peptides endogenously bound to B*0802 and 12 individual peptides purified from that mixture. The HLA-B*0802 allotype, while able to bind some peptides bound by B*0801, has a broader repertoire of endogenously bound peptides than B*0801: the peptides bound by B*0802 are more variable in length and exhibit greater diversity in the carboxyl-terminal amino acid which interacts with the F pocket.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 29 October 1997
Rights and permissions
About this article
Cite this article
Arnett, K., Huang, W., Valiante, N. et al. The Bw4/Bw6 difference between HLA-B*0802 and HLA-B*0801 changes the peptides endogenously bound and the stimulation of alloreactive T cells. Immunogenetics 48, 56–61 (1998). https://doi.org/10.1007/s002510050400
Issue Date:
DOI: https://doi.org/10.1007/s002510050400