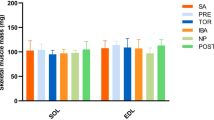
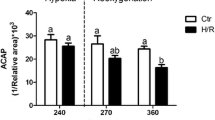
Abstract
Birds seem to employ powerful physiological strategies to curb the harmful effects of reactive oxygen species (ROS) because they generally live longer than predicted by the free radical theory of aging. However, little is known about the physiological mechanisms that confer protection to birds against excessive ROS generation. Hence, we investigated the ability of birds to control mitochondrial ROS generation during physiologically stressful periods. In our study, we analyzed the relationship between the thyroid status and the function of intermyofibrillar and subsarcolemmal mitochondria located in glycolytic and oxidative muscles of ducklings. We found that the intermyofibrillar mitochondria of both glycolytic and oxidative muscles down regulate ROS production when plasma T3 levels rise. The intermyofibrillar mitochondria of the gastrocnemius muscle (an oxidative muscle) produced less ROS and were more sensitive than the pectoralis muscle (a glycolytic muscle) to changes in plasma T3. Such differences in the ROS production by glycolytic and oxidative muscles were associated with differences in the membrane proton permeability and in the rate of free radical leakage within the respiratory chain. This is the first evidence which shows that in birds, the amount of ROS that the mitochondria release is dependent on: (1) their location within the muscle; (2) the type of muscle (glycolytic or oxidative) and (3) on the thyroid status. Reducing muscle mitochondrial ROS generation might be an important mechanism in birds to limit oxidative damage during periods of physiological stress.


Similar content being viewed by others
References
Adhihetty PJ, Ljubicic V, Menzies KJ, Hood DA (2005) Differential susceptibility of subsarcolemmal and intermyofibrillar mitochondria to apoptotic stimuli. Am J Physiol Cell Physiol 289:C994–C1001
Anderson EJ, Neufer PD (2006) Type II skeletal myofibers possess unique properties that potentiate mitochondrial H(2)O(2) generation. Am J Physiol Cell Physiol 290:C844–C851
Brand MD (2000) Uncoupling to survive? The role of mitochondrial inefficiency in ageing. Exp Gerontol 35:811–820
Chanseaume E, Malpuech-Brugere C, Patrac V, Bielicki G, Rousset P, Couturier K, Salles J, Renou JP, Boirie Y, Morio B (2006) Diets high in sugar, fat, and energy induce muscle type-specific adaptations in mitochondrial functions in rats. J Nutr 136:2194–2200
Cogswell AM, Stevens RJ, Hood DA (1993) Properties of skeletal muscle mitochondria isolated from subsarcolemmal and intermyofibrillar regions. Am J Physiol Cell Physiol 264:C383–C389
Cohen AA, McGraw KJ, Robinson WD (2009) Serum antioxidant levels in wild birds vary in relation to diet, season, life history strategy, and species. Oecologia 161:673–683
Costantini D, Dell’ariccia G, Lipp HP (2008a) Long flights and age affect oxidative status of homing pigeons (Columba livia). J Exp Biol 211:377–381
Costantini D, Fanfani A, Dell’Omo G (2008b) Effects of corticosteroids on oxidative damage and circulating carotenoids in captive adult kestrel (Falco tinnunculus). J Comp Physiol B 178:829–835
Duchamp C, Barre H, Rouanet JL, Lanni A, Cohen-Adad F, Berne G, Brebion P (1991) Nonshivering thermogenesis in king penguin chicks. I. Role of skeletal muscle. Am J Physiol Regul Integr Comp Physiol 261:R1438–R1445
Duchamp C, Chatonnet J, Dittmar A, Barre H (1993) Increased role of skeletal muscle in the calorigenic response to glucagon of cold-acclimated ducklings. Am J Physiol Regul Integr Comp Physiol 265:R1084–R1091
Ferreira R, Vitorino R, Alves RM, Appell HJ, Powers SK, Duarte JA, Amado F (2010) Subsarcolemmal and intermyofibrillar mitochondria proteome differences disclose functional specializations in skeletal muscle. Proteomics 10:3142–3154
Goglia F, Lanni A, Duchamp C, Rouanet JL, Barre H (1993) Effect of cold acclimation on oxidative capacity and respiratory properties of liver and muscle mitochondria in ducklings, Cairina moschata. Comp Biochem Physiol 106:95–101
Holmes DJ, Fluckiger R, Austad SN (2001) Comparative biology of aging in birds: an update. Exp Gerontol 36:869–883
Horak P, Zilmer M, Saks L, Ots I, Karu U, Zilmer K (2006) Antioxidant protection, carotenoids and the costs of immune challenge in greenfinches. J Exp Biol 209:4329–4338
Hulbert AJ, Faulks S, Buttemer WA, Else PL (2002) Acyl composition of muscle membranes varies with body size in birds. J Exp Biol 205:3561–3569
Iossa S, Lionetti L, Mollica MP, Crescenzo R, Botta M, Samec S, Dulloo AG, Liverini G (2001) Differences in proton leak kinetics, but not in UCP3 protein content, in subsarcolemmal and intermyofibrillar skeletal muscle mitochondria from fed and fasted rats. FEBS Lett 505:53–56
Judge S, Jang YM, Smith A, Selman C, Phillips T, Speakman JR, Hagen T, Leeuwenburgh C (2005) Exercise by lifelong voluntary wheel running reduces subsarcolemmal and interfibrillar mitochondrial hydrogen peroxide production in the heart. Am J Physiol Regul Integr Comp Physiol 289:R1564–R1572
Korshunov SS, Skulachev VP, Starkov AA (1997) High protonic potential actuates a mechanism of production of reactive oxygen species in mitochondria. FEBS Lett 416:15–18
Lambert AJ, Boysen HM, Buckingham JA, Yang T, Podlutsky A, Austad SN, Kunz TH, Buffenstein R, Brand MD (2007) Low rates of hydrogen peroxide production by isolated heart mitochondria associate with long maximum lifespan in vertebrate homeotherms. Aging Cell 6:607–618
Ljubicic V, Hood DA (2008) Kinase-specific responsiveness to incremental contractile activity in skeletal muscle with low and high mitochondrial content. Am J Physiol Endocrinol Metab 295:E195–E204
Lombardi A, Damon M, Vincent A, Goglia F, Herpin P (2000) Characterisation of oxidative phosphorylation in skeletal muscle mitochondria subpopulations in pig: a study using top-down elasticity analysis. FEBS Lett 475:84–88
Lopez-Torres M, Romero M, Barja G (2000) Effect of thyroid hormones on mitochondrial oxygen free radical production and DNA oxidative damage in the rat heart. Mol Cell Endocrinol 168:127–134
Mollica MP, Lionetti L, Crescenzo R, D’Andrea E, Ferraro M, Liverini G, Iossa S (2006) Heterogeneous bioenergetic behaviour of subsarcolemmal and intermyofibrillar mitochondria in fed and fasted rats. Cell Mol Life Sci 63:358–366
Mujahid A, Yoshiki Y, Akiba Y, Toyomizu M (2005) Superoxide radical production in chicken skeletal muscle induced by acute heat stress. Poult Sci 84:307–314
Mujahid A, Sato K, Akiba Y, Toyomizu M (2006) Acute heat stress stimulates mitochondrial superoxide production in broiler skeletal muscle, possibly via downregulation of uncoupling protein content. Poult Sci 85:1259–1265
Picard M, Hepple RT, Burelle Y (2012) Mitochondrial functional specialization in glycolytic and oxidative muscle fibers: tailoring the organelle for optimal function. Am J Physiol Cell Physiol 302:C629–C641
Rey B, Roussel D, Romestaing C, Belouze M, Rouanet JL, Desplanches D, Sibille B, Servais S, Duchamp C (2010) Up-regulation of avian uncoupling protein in cold-acclimated and hyperthyroid ducklings prevents reactive oxygen species production by skeletal muscle mitochondria. BMC Physiol 10:5
Rey B, Roussel D, Teulier L, Eyenga P, Degletagne C, Belouze M, Duchamp C (2011) Functional argument for the existence of an avian nitric oxide synthase in muscle mitochondria: effect of cold acclimation. FEBS Lett 585:173–177
Roussel D, Rouanet JL, Duchamp C, Barre H (1998) Effects of cold acclimation and palmitate on energy coupling in duckling skeletal muscle mitochondria. FEBS Lett 439:258–262
Roussel D, Chainier F, Rouanet J, Barre H (2000) Increase in the adenine nucleotide translocase content of duckling subsarcolemmal mitochondria during cold acclimation. FEBS Lett 477:141–144
Servais S, Couturier K, Koubi H, Rouanet JL, Desplanches D, Sornay-Mayet MH, Sempore B, Lavoie JM, Favier R (2003) Effect of voluntary exercise on H2O2 release by subsarcolemmal and intermyofibrillar mitochondria. Free Radic Biol Med 35:24–32
Silva JE (1995) Thyroid hormone control of thermogenesis and energy balance. Thyroid 5:481–492
Simonides WS, van Hardeveld C (2008) Thyroid hormone as a determinant of metabolic and contractile phenotype of skeletal muscle. Thyroid 18:205–216
Stowe DF, Camara AK (2009) Mitochondrial reactive oxygen species production in excitable cells: modulators of mitochondrial and cell function. Antioxid Redox Signal 11:1373–1414
Suarez RK (1992) Hummingbird flight: sustaining the highest mass-specific metabolic rates among vertebrates. Experientia 48:565–570
Toyomizu M, Ueda M, Sato S, Seki Y, Sato K, Akiba Y (2002) Cold-induced mitochondrial uncoupling and expression of chicken UCP and ANT mRNA in chicken skeletal muscle. FEBS Lett 529:313–318
Venditti P, Puca A, Di Meo S (2003a) Effect of thyroid state on rate and sites of H2O2 production in rat skeletal muscle mitochondria. Arch Biochem Biophys 411:121–128
Venditti P, Puca A, Di Meo S (2003b) Effects of thyroid state on H2O2 production by rat heart mitochondria: sites of production with complex I- and complex II-linked substrates. Horm Metab Res 35:55–61
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by I.D. Hume.
Rights and permissions
About this article
Cite this article
Rey, B., Roussel, D., Rouanet, JL. et al. Differential effects of thyroid status on regional H2O2 production in slow- and fast-twitch muscle of ducklings. J Comp Physiol B 183, 135–143 (2013). https://doi.org/10.1007/s00360-012-0692-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-012-0692-5