Abstract
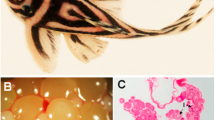
The extracellular matrix (ECM) plays a prominent role in ovarian function by participating in processes such as cell migration, proliferation, growth, and development. Although some of these signaling processes have been characterized in the mouse, the relative quantity and distribution of ECM proteins within developing follicles of the ovary have not been characterized. This study uses immunohistochemistry and real-time PCR to characterize the ECM components type I collagen, type IV collagen, fibronectin, and laminin in the mouse ovary according to follicle stage and cellular compartment. Collagen I was present throughout the ovary, with higher concentrations in the ovarian surface epithelium and follicular compartments. Collagen IV was abundant in the theca cell compartment with low-level expression in the stroma and granulosa cells. The distribution of collagen was consistent throughout follicle maturation. Fibronectin staining in the stroma and theca cell compartment increased throughout follicle development, while staining in the granulosa cell compartment decreased. Heavy staining was also observed in the follicular fluid of antral follicles. Laminin was localized primarily to the theca cell compartment, with a defined ring at the exterior of the follicular granulosa cells marking the basement membrane. Low levels of laminin were also apparent in the stroma and granulosa cell compartment. Taken together, the ECM content of the mouse ovary changes during follicular development and reveals a distinct spatial and temporal pattern. This understanding of ECM composition and distribution can be used in the basic studies of ECM function during follicle development, and could aid in the development of in vitro systems for follicle growth.





Similar content being viewed by others
References
Aharoni D, Meiri I, Atzmon R, Vlodavsky I, Amsterdam A (1997) Differential effect of components of the extracellular matrix on differentiation and apoptosis. Curr Biol 7:43–51
Akkoyunlu G, Demir R, Ustunel I (2003) Distribution patterns of TGF-alpha, laminin and fibronectin and their relationship with folliculogenesis in rat ovary. Acta Histochem 105:295–301
Auersperg N, Maclaren I AKruk PA (1991) Ovarian surface epithelium:autonomous production of connective tissue-type extracellular matrix. Biol Reprod 44:717–724
Auersperg N, Wong AS, Choi KC, Kang S KLeung PC (2001) Ovarian surface epithelium: biology, endocrinology, and pathology. Endocr Rev 22:255–288
Bortolussi M, Zanchetta R, Doliana R, Castellani I, Bressan GM, Lauria A (1989) Changes in the organization of the extracellular matrix in ovarian follicles during the preovulatory phase and atresia. An immunofluorescence study. Basic Appl Histochem 33:31–38
Bussenot I, Ferre G, Azoulay-Barjonet C, Murgo C, Vieitez G, Parinaud J (1993) Culture of human preovulatory granulosa cells: effect of extracellular matrix on steroidogenesis. Biol Cell 77:181–186
Bustin SA (2000) Absolute quantification of mRNA using real-time reverse transcription polymerase chain reaction assays. J Mol Endocrinol 25:169–193
Carnegie JA (1990) Secretion of fibronectin by rat granulosa cells occurs primarily during early follicular development. J Reprod Fertil 89:579–589
Carnegie JA, Byard R, Dardick I, Tsang BK (1988) Culture of granulosa cells in collagen gels: the influence of cell shape on steroidogenesis. Biol Reprod 38:881–890
Cortvrindt R, Smitz J (1998) Early preantral mouse follicle in vitro maturation: oocyte growth, meiotic maturation and granulosa-cell proliferation. Theriogenology 49:845–859
Figueiredo J, Hulshof SCJ, Thiry M, Van den Hurk R, Bevers MM, Nusgens B, Beckers JF (1995) Extracellular matrix proteins and basement membrane: their identification in bovine ovaries and significance for the attachment of cultured preantral follicles. Theriogenology 43:845–858
Frojdman K, Pelliniemi LJ, Virtanen I (1998) Differential distribution of type IV collagen chains in the developing rat testis and ovary. Differentiation 63:125–130
Furman A, Rotmensch S, Kohen F, Mashiach S, Amsterdam A (1986) Regulation of rat granulosa cell differentiation by extracellular matrix produced by bovine corneal endothelial cells. Endocrinology 118:1878–1885
Gentry PA, Zareie M, Liptrap RM (1996) Fibronectin concentrations correlate with ovarian follicular size and estradiol values in equine follicular fluid. Anim Reprod Sci 45:91–102
Gomes JE, Correia SC, Gouveia-Oliveira A, Cidadao AJ, Plancha CE (1999) Three-dimensional environments preserve extracellular matrix compartments of ovarian follicles and increase FSH-dependent growth. Mol Reprod Dev 54:163–172
Hovatta O, Silye R, Abir R, Krausz T, Winston RM (1997) Extracellular matrix improves survival of both stored and fresh human primordial and primary ovarian follicles in long-term culture. Hum Reprod 12:1032–1036
Huet C, Monget P, Pisselet C, Monniaux D (1997) Changes in extracellular matrix components and steroidogenic enzymes during growth and atresia of antral ovarian follicles in the sheep. Biol Reprod 56:1025–1034
Huet C, Pisselet C, Mandon-Pepin B, Monget P, Monniaux D (2001) Extracellular matrix regulates ovine granulosa cell survival, proliferation and steroidogenesis: relationships between cell shape and function. J Endocrinol 169:347–360
Irving-Rodgers HF, Mussard ML, Kinder JE, Rodgers RJ (2002) Composition and morphology of the follicular basal lamina during atresia of bovine antral follicles. Reproduction 123:97–106
Iwahashi M, Muragaki Y, Ooshima A, Nakano R (2000) Type VI collagen expression during growth of human ovarian follicles. Fertil Steril 74:343–347
Kreeger PK, Fernandes NN, Woodruff TK, Shea LD (2005) Regulation of mouse follicle development by follicle-stimulating hormone in a three-dimensional in vitro culture system is dependent on follicle stage and dose. Biol Reprod 73:942–950
Kreeger PK, Deck JW, Woodruff TK, Shea LD (2006) The in vitro regulation of ovarian follicle development using alginate-extracellular matrix gels. Biomaterials 27:714–723
Le Bellego F, Pisselet C, Huet C, Monget P, Monniaux D (2002) Laminin-alpha6beta1 integrin interaction enhances survival and proliferation and modulates steroidogenesis of ovine granulosa cells. J Endocrinol 172:45–59
Lee VH, Britt JH, Dunbar BS (1996) Localization of laminin proteins during early follicular development in pig and rabbit ovaries. J Reprod Fertil 108:115–122
Matzuk MM (2000) Revelations of ovarian follicle biology from gene knockout mice. Mol Cell Endocrinol 163:61–66
McArthur ME, Irving-Rodgers HF, Byers S, Rodgers RJ (2000) Identification and immunolocalization of decorin, versican, perlecan, nidogen, and chondroitin sulfate proteoglycans in bovine small-antral ovarian follicles. Biol Reprod 63:913–924
O’Brien MJ, Pendola JK, Eppig JJ (2003) A revised protocol for in vitro development of mouse oocytes from primordial follicles dramatically improves their developmental competence. Biol Reprod 68:1682–1686
Oktay K, Karlikaya G, Akman O, Ojakian GK, Oktay M (2000) Interaction of extracellular matrix and activin-A in the initiation of follicle growth in the mouse ovary. Biol Reprod 63:457–461
Rajah R, Sundaram GS (1994) Protein distribution and gene expression of collagen type IV in the neonatal rat ovary during follicle formation. Cell Mol Biol (Noisy-le-grand) 40:769–780
Rodgers HF, Irvine CM, van Wezel IL, Lavranos TC, Luck MR, Sado Y, Ninomiya Y, Rodgers RJ (1998) Distribution of the alpha1 to alpha6 chains of type IV collagen in bovine follicles. Biol Reprod 59:1334–1341
Rodgers RJ, Lavranos TC, van Wezel IL, Irving-Rodgers HF (1999a) Development of the ovarian follicular epithelium. Mol Cell Endocrinol 151:171–179
Rodgers RJ, van Wezel IL, Irving-Rodgers HF, Lavranos TC, Irvine CM, Krupa M (1999b) Roles of extracellular matrix in follicular development. J Reprod Fertil Suppl 54:343–352
Rodgers RJ, Irving-Rodgers HF, van Wezel IL, Krupa M, Lavranos TC (2001) Dynamics of the membrana granulosa during expansion of the ovarian follicular antrum. Mol Cell Endocrinol 171:41–48
Smitz JE, Cortvrindt RG (2002) The earliest stages of folliculogenesis in vitro. Reproduction 123:185–202
Spears N, Murray AA, Allison V, Boland NI, Gosden RG (1998) Role of gonadotrophins and ovarian steroids in the development of mouse follicles in vitro. J Reprod Fertil 113:19–26
Tsuiki A, Preyer J, Hung TT (1988) Fibronectin and glycosaminoglycans in human preovulatory follicular fluid and their correlation to follicular maturation. Hum Reprod 3:425–429
Wang T, Brown MJ (1999) mRNA quantification by real time TaqMan polymerase chain reaction:validation and comparison with RNase protection. Anal Biochem 269:198–201
van Wezel IL, Rodgers HF, Rodgers RJ (1998) Differential localization of laminin chains in bovine follicles. J Reprod Fertil 112:267–278
Yamada S, Fujiwara H, Honda T, Higuchi T, Nakayama T, Inoue T, Maeda M, Fujii S (1999) Human granulosa cells express integrin alpha2 and collagen type IV: possible involvement of collagen type IV in granulosa cell luteinization. Mol Hum Reprod 5:607–617
Zhao Y, Luck MR (1995) Gene expression and protein distribution of collagen, fibronectin and laminin in bovine follicles and corpora lutea. J Reprod Fertil 104:115–123
Acknowledgements
This research was supported by the NICHD/NIH through cooperative agreement U54 HD041857 as part of the Specialized Cooperative Centers Program in Reproductive Research and the NIH Biotechnology Training Grant for C.B.B. We would like to thank the Pathology Core Laboratory (Northwestern University, Chicago, IL, USA), especially Andrew Lisowski, for the paraffin embedding and sectioning of all ovarian tissue. A special thanks to Sarah Bristol and Joanna Burdette for their technical assistance and support with both the immunohistochemistry and real-time PCR. We would also like to thank Erin West for her review of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berkholtz, C.B., Lai, B.E., Woodruff, T.K. et al. Distribution of extracellular matrix proteins type I collagen, type IV collagen, fibronectin, and laminin in mouse folliculogenesis. Histochem Cell Biol 126, 583–592 (2006). https://doi.org/10.1007/s00418-006-0194-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-006-0194-1