Abstract.
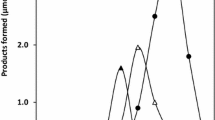
Geranyldiphosphate:4-hydroxybenzoate 3-geranyltransferase is a regulatory enzyme in the biosynthesis of shikonin, a phytoalexin and pharmaceutical produced by cell cultures of Lithospermum erythrorhizon Sieb. et Zucc.. In Linsmaier-Skoog medium, the activity of this enzyme could be enhanced more than 200-fold by addition of methyl jasmonate, and this culture material was used for the solubilization and purification of the enzyme. Of various detergents examined, digitonin was the most suitable for the solubilization of the enzyme. The solubilized enzyme was purified 800-fold by chromatography over diethylaminoethyl (DEAE)-Sephacel, Heparin-Sepharose, Reactive Green 19-Agarose, and Cholic Acid-Agarose. The purified enzyme required magnesium ions as cofactor and was highly specific for geranyldiphosphate (GPP) and 4-hydroxybenzoate (4HB) as substrates. The K m values for 4HB and GPP were calculated by the method of Lineweaver and Burk as 18.4 μM and 13.8 μM, respectively.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 2 July 1997 / Accepted: 14 October 1997
Rights and permissions
About this article
Cite this article
Mühlenweg, A., Melzer, M., Li, SM. et al. 4-Hydroxybenzoate 3-geranyltransferase from Lithospermum erythrorhizon: purification of a plant membrane-bound prenyltransferase. Planta 205, 407–413 (1998). https://doi.org/10.1007/s004250050337
Issue Date:
DOI: https://doi.org/10.1007/s004250050337