Abstract
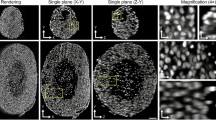
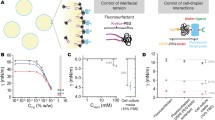
A spheroid is an in vitro multicellular aggregate that provides a microenvironment resembling that of normal tissue in vivo. Although cell adhesion molecules such as integrins and cadherins have been implicated in participating in the process of spheroid formation, little is known about the timing of their action. In this study, we have employed an image-based quantitative method to investigate the compactness of cell aggregates during hepatoma spheroid formation in a dynamic fashion. By modulating β1-integrin and E-cadherin activity with specific blocking antibodies, ion chelators, and RGD-sequence-containing peptides, we show that these cell adhesion molecules mediate the formation of spheroids through the establishment of complex cell-cell and cell-extracellular matrix (ECM) interactions. The dynamics of spheroid formation can be separated into three stages. In the first stage, ECM fibers act as a long-chain linker for the attachment of dispersed single-cells to form loose aggregations through the binding of integrins. This is followed by a delay period in which cell aggregates pause in compaction, presumably because of the accumulation of sufficient amounts of E-cadherins. In the third stage, strong homophilic interaction of E-cadherins is a major factor for the morphological transition from loose cell aggregates to compact spheroids. These findings thus provide comprehensive information on the molecular mechanisms and dynamics of hepatoma spheroid formation.








Similar content being viewed by others
References
Abu-Absi SF, Friend JR, Hansen LK, Hu WS (2002) Structural polarity and functional bile canaliculi in rat hepatocyte spheroids. Exp Cell Res 274:56–67
Alpaugh ML, Tomlinson JS, Ye Y, Barsky SH (2002) Relationship of sialyl-Lewis(x/a) underexpression and E-cadherin overexpression in the lymphovascular embolus of inflammatory breast carcinoma. Am J Pathol 161:619–628
Angres B, Barth A, Nelson WJ (1996) Mechanism for transition from initial to stable cell-cell adhesion: kinetic analysis of E-cadherin-mediated adhesion using a quantitative adhesion assay. J Cell Biol 134:549–557
Angst BD, Marcozzi C, Magee AI (2001) The cadherin superfamily: diversity in form and function. J Cell Sci 114:629–641
Arusha O, Timothy M, Shoukat D (2004) Regulation of E-cadherin expression and β-catenin/Tcf transcriptional activity by the integrin-linked kinase. Biochem Biophys Acta 1691:1–15
Bates RC, Edwards NS, Yates JD (2000) Spheroids and cell survival. Crit Rev Oncol Hematol 36:61–74
Byers SW, Sommers CL, Hoxter B, Mercurio AM, Tozeren A (1995) Role of E-cadherin in the response of tumor cell aggregates to lymphatic, venous and arterial flow: measurement of cell-cell adhesion strength. J Cell Sci 108:2053–2064
Casey RC, Burleson KM, Skubitz KM, Pambuccian SE, Oegema TR Jr, Ruff LE, Skubitz AP (2001) Beta 1-integrins regulate the formation and adhesion of ovarian carcinoma multicellular spheroids. Am J Pathol 159:2071–2080
Cavallaro U, Christofori G (2004) Cell adhesion and signalling by cadherins and Ig-CAMs in cancer. Nat Rev Cancer 4:118–132
Conacci-Sorrell M, Simcha I, Ben-Yedidia T, Blechman J, Savagner P, Ben-Ze’ev A (2003) Autoregulation of E-cadherin expression by cadherin-cadherin interactions: the roles of beta-catenin signaling, Slug, and MAPK. J Cell Biol 163:847–857
Desoize B (2000) Contribution of three-dimensional culture to cancer research. Crit Rev Oncol Hematol 36:59–60
Enmon RM Jr, O’Connor KC, Lacks DJ, Schwartz DK, Dotson RS (2001) Dynamics of spheroid self-assembly in liquid-overlay culture of DU 145 human prostate cancer cells. Biotechnol Bioeng 72:579–591
Enmon RM Jr, O’Connor KC, Song H, Lacks DJ, Schwartz DK (2002) Aggregation kinetics of well and poorly differentiated human prostate cancer cells. Biotechnol Bioeng 80:580–588
Frisch SM, Ruoslahti E (1997) Integrins and anoikis. Curr Opin Cell Biol 9:701–706
Frisch SM, Screaton RA (2001) Anoikis mechanisms. Curr Opin Cell Biol 13:555–562
Frixen UH, Behrens J, Sachs M, Eberle G, Voss B, Warda A, Lochner D, Birchmeier W (1991) E-cadherin-mediated cell-cell adhesion prevents invasiveness of human carcinoma cells. J Cell Biol 113:173–185
Fukuda J, Mizumoto H, Nakazawa K, Kajiwara T, Funatsu K (2004) Hepatocyte organoid culture in elliptic hollow fibers to develop a hybrid artificial liver. Int J Artif Organs 27:1091–1099
Glinsky VV, Glinsky GV, Glinskii OV, Huxley VH, Turk JR, Mossine VV, Deutscher SL, Pienta KJ, Quinn TP (2003) Intravascular metastatic cancer cell homotypic aggregation at the sites of primary attachment to the endothelium. Cancer Res 63:3805–3811
Guo W, Giancotti FG (2004) Integrin signalling during tumour progression. Nat Rev Mol Cell Biol 5:816–826
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100:57–70
Holtfreter J (1944) A study of the mechanics of gastrulation. J Exp Zool 95:171–212
Islam S, Carey TE, Wolf GT, Wheelock MJ, Johnson KR (1996) Expression of N-cadherin by human squamous carcinoma cells induces a scattered fibroblastic phenotype with disrupted cell-cell adhesion. J Cell Biol 135:1643–1654
Jakab K, Neagu A, Mironov V, Markwald RR, Forgacs G (2004) Engineering biological structures of prescribed shape using self-assembling multicellular systems. Proc Natl Acad Sci USA 101:2864–2869
Kale S, Biermann S, Edwards C, Tarnowski C, Morris M, Long MW (2000) Three-dimensional cellular development is essential for ex vivo formation of human bone. Nat Biotechnol 18:954–958
Kelm JM, Fussenegger M (2004) Microscale tissue engineering using gravity-enforced cell assembly. Trends Biotechnol 22:195–202
Kelm JM, Timmins NE, Brown CJ, Fussenegger M, Nielsen LK (2003) Method for generation of homogeneous multicellular tumor spheroids applicable to a wide variety of cell types. Biotechnol Bioeng 83:173–180
Korff T, Augustin HG (1998) Integration of endothelial cells in multicellular spheroids prevents apoptosis and induces differentiation. J Cell Biol 143:1341–1352
Kunz-Schughart LA, Kreutz M, Knuechel R (1998) Multicellular spheroids: a three-dimensional in vitro culture system to study tumour biology. Int J Exp Pathol 79:1–23
Landry J, Bernier D, Ouellet C, Goyette R, Marceau N (1985) Spheroidal aggregate culture of rat liver cells: histotypic reorganization, biomatrix deposition, and maintenance of functional activities. J Cell Biol 101:914–923
Larue L, Antos C, Butz S, Huber O, Delmas V, Dominis M, Kemler R (1996) A role for cadherins in tissue formation. Development 122:3185–3194
Lazar A, Peshwa MV, Wu FJ, Chi CM, Cerra FB, Hu WS (1995) Formation of porcine hepatocyte spheroids for use in a bioartificial liver. Cell Transplant 4:259–268
Marrs JA, Andersson-Fisone C, Jeong MC, Cohen-Gould L, Zurzolo C, Nabi IR, Rodriguez-Boulan E, Nelson WJ (1995) Plasticity in epithelial cell phenotype: modulation by expression of different cadherin cell adhesion molecules. J Cell Biol 129:507–519
Moscona A, Moscona H (1952) The dissociation and aggregation of cells from organ rudiments of the early chick embryo. J Anat 86:287–301
Mueller-Klieser W (1997) Three-dimensional cell cultures: from molecular mechanisms to clinical applications. Am J Physiol 273:C1109–C1123
Nagar B, Overduin M, Ikura M, Rini JM (1996) Structural basis of calcium-induced E-cadherin rigidification and dimerization. Nature 380:360–364
Nakamura M, Shinji T, Ujike K, Hirasaki S, Koide N, Tsuji T (2002) Cytoskeletal inhibitors, anti-adhesion molecule antibodies, and lectins inhibit hepatocyte spheroid formation. Acta Med Okayama 56:43–50
Narayanan RA, Rink A, Beattie CW, Hu WS (2002) Differential gene expression analysis during porcine hepatocyte spheroid formation. Mamm Genome 13:515–523
Neelamegham S, Munn LL, Zygourakis K (1997) A model for the kinetics of homotypic cellular aggregation under static conditions. Biophys J 72:51–64
O’Brien LE, Zegers MM, Mostov KE (2002) Building epithelial architecture: insights from three-dimensional culture models. Nat Rev Mol Cell Biol 3:531–537
Robinson EE, Zazzali KM, Corbett SA, Foty RA (2003) Alpha5beta1 integrin mediates strong tissue cohesion. J Cell Sci 116:377–386
Robinson EE, Foty RA, Corbett SA (2004) Fibronectin matrix assembly regulates alpha5beta1-mediated cell cohesion. Mol Biol Cell 15:973–981
Shimaoka M, Takagi J, Springer TA (2002) Conformational regulation of integrin structure and function. Annu Rev Biophys Biomol Struct 31:485–516
Shimazui T, Schalken JA, Kawai K, Kawamoto R, Bockhoven A van, Oosterwijk E, Akaza H (2004) Role of complex cadherins in cell-cell adhesion evaluated by spheroid formation in renal cell carcinoma cell lines. Oncol Rep 11:357–360
Tomlinson JS, Alpaugh ML, Barsky SH (2001) An intact overexpressed E-cadherin/alpha,beta-catenin axis characterizes the lymphovascular emboli of inflammatory breast carcinoma. Cancer Res 61:5231–5241
Wong CW, Song C, Grimes MM, Fu W, Dewhirst MW, Muschel RJ, Al-Mehdi AB (2002) Intravascular location of breast cancer cells after spontaneous metastasis to the lung. Am J Pathol 161:749–753
Wong BW, Luk JM, Ng IO, Hu MY, Liu KD, Fan ST (2003) Identification of liver-intestine cadherin in hepatocellular carcinoma—a potential disease marker. Biochem Biophys Res Commun 311:618–624
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the National Program of Genome Medicine, ROC (NSC 93-3112-B007-001) and Veteran General Hospitals University System of Taiwan Joint Research Program (VGHUST94-G6-06-3).
Rights and permissions
About this article
Cite this article
Lin, RZ., Chou, LF., Chien, CC.M. et al. Dynamic analysis of hepatoma spheroid formation: roles of E-cadherin and β1-integrin. Cell Tissue Res 324, 411–422 (2006). https://doi.org/10.1007/s00441-005-0148-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-005-0148-2