Abstract
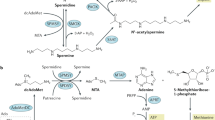
Polyamines, including spermine, spermidine, and the precursor diamine, putrescine, are naturally occurring polycationic alkylamines that are required for eukaryotic cell growth, differentiation, and survival. This absolute requirement for polyamines and the need to maintain intracellular levels within specific ranges require a highly regulated metabolic pathway primed for rapid changes in response to cellular growth signals, environmental changes, and stress. Although the polyamine metabolic pathway is strictly regulated in normal cells, dysregulation of polyamine metabolism is a frequent event in cancer. Recent studies suggest that the polyamine catabolic pathway may be involved in the etiology of some epithelial cancers. The catabolism of spermine to spermidine utilizes either the one-step enzymatic reaction of spermine oxidase (SMO) or the two-step process of spermidine/spermine N 1-acetyltransferase (SSAT) coupled with the peroxisomal enzyme N 1-acetylpolyamine oxidase. Both catabolic pathways produce hydrogen peroxide and a reactive aldehyde that are capable of damaging DNA and other critical cellular components. The catabolic pathway also depletes the intracellular concentrations of spermidine and spermine, which are free radical scavengers. Consequently, the polyamine catabolic pathway in general and specifically SMO and SSAT provide exciting new targets for chemoprevention and/or chemotherapy.


Similar content being viewed by others
References
Atiya Ali M, Poortvliet E, Strömberg R, Yngve A (2011) Polyamines in foods: development of a food database. Food Nutr Res 55:424–429
Babbar N, Casero RA Jr (2006) Tumor necrosis factor-alpha increases reactive oxygen species by inducing spermine oxidase in human lung epithelial cells: a potential mechanism for inflammation-induced carcinogenesis. Cancer Res 66(23):11125–11130
Babbar N, Hacker A, Huang Y, Casero RA Jr (2006a) Tumor necrosis factor alpha induces spermidine/spermine N1-acetyltransferase through nuclear factor kappaB in non-small cell lung cancer cells. J Biol Chem 281(34):24182–24192
Babbar N, Gerner EW, Casero RA Jr (2006c) Induction of spermidine/spermine N1-acetyltransferase (SSAT) by aspirin in Caco-2 colon cancer cells. Biochem J 394(Pt1):317–324
Bardia A, Platz EA, Yegnasubramanian S, De Marzo AM, Nelson WG (2009) Anti-inflammatory drugs, antioxidants, and prostate cancer prevention. Curr Opin Pharmacol 9(4):419–426
Bianchi M, Polticelli F, Ascenzi P, Botta M, Federico R, Mariottini P, Cona A (2006) Inhibition of polyamine and spermine oxidases by polyamine analogues. FEBS J 273(6):1115–1123
Burn J, Mathers J, Bishop DT (2013) Genetics, inheritance and strategies for prevention in populations at high risk of colorectal cancer (CRC). Recent Results Cancer Res 191:157–183
Casero RA Jr, Marton LJ (2007) Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases. Nat Rev Drug Discov 6(5):373–390
Casero RA Jr, Pegg AE (2009) Polyamine catabolism and disease. Biochem J 421(3):323–338
Casero RA Jr, Ervin SJ, Celano P, Baylin SB, Bergeron RJ (1989) Differential response to treatment with the bis(ethyl)polyamine analogues between human small cell lung carcinoma and undifferentiated large cell lung carcinoma in culture. Cancer Res 49(3):639–643
Chaturvedi R, Cheng Y, Asim M, Bussiere FI, Xu H, Gobert AP, Hacker A, Casero RA Jr, Wilson KT (2004) Induction of polyamine oxidase 1 by Helicobacter pylori causes macrophage apoptosis by hydrogen peroxide release and mitochondrial membrane depolarization. J Biol Chem 279(38):40161–40173
Chaturvedi R, de Sablet T, Peek RM, Wilson KT (2012) Spermine oxidase, a polyamine catabolic enzyme that links Helicobacter pylori CagA and gastric cancer cells. Gut Microbes 3(1):48–56
Chu FF, Esworthy RS, Chu PG, Longmate JA, Huycke MM, Wilczynski S, Doroshow JH (2004) Bacteria-induced intestinal cancer in mice with disrupted Gpx1 and Gpx2 genes. Cancer Res 64(3):962–968
De Marzo AM, Marchi VL, Epstein JI, Nelson WG (1999) Proliferative inflammatory atrophy of the prostate: implications for prostatic carcinogenesis. Am J Pathol 155(6):1985–1992
Goel A, Chang DK, Ricciardiello L, Gasche C, Boland CR (2003) A novel mechanism for aspirin-mediated growth inhibition of human colon cancer cells. Clin Cancer Res 9(1):383–390
Goodwin AC, Jadallah S, Toubaji A, Lecksell K, Hicks JL, Kowalski J, Bova GS, De Marzo AM, Netto GJ, Casero RA Jr (2008) Increased spermine oxidase expression in human prostate cancer and prostatic intraepithelial neoplasia tissues. Prostate 68(7):766–772
Goodwin AC, Destefano Shields CE, Wu S, Huso DL, Wu X, Murray-Stewart TR, Hacker-Prietz A, Rabizadeh S, Woster PM, Sears CL, Casero RA Jr (2011) Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc Natl Acad Sci USA 108(37):15354–15359
Ha HC, Sirisoma NS, Kuppusamy P, Zweier JL, Woster PM, Casero RA Jr (1998) The natural polyamine spermine functions directly as a free radical scavenger. Proc Natl Acad Sci USA 95(19):11140–11145
Hong SK, Chaturvedi R, Piazuelo MB, Coburn LA, Williams CS, Delgado AG, Casero RA Jr, Schwartz DA, Wilson KT (2010) Increased expression and cellular localization of spermine oxidase in ulcerative colitis and relationship to disease activity. Inflamm Bowel Dis 16(9):1557–1566
Hughes A, Smith NI, Wallace HM (2003) Polyamines reverse non-steroidal anti-inflammatory drug-induced toxicity in human colorectal cancer cells. Biochem J 374(Pt 2):481–488
Igarashi K, Kashiwagi K (2010) Modulation of cellular function by polyamines. Int J Biochem Cell Biol 42(1):39–51
Igarashi K, Kashiwagi K (2011) Protein-conjugated acrolein as a biochemical marker of brain infarction. Mol Nutr Food Res 55(9):1332–1341
Ignatenko NA, Besselsen DG, Roy UK, Stringer DE, Blohm-Mangone KA, Padilla-Torres JL, Guillen-R JM, Gerner EW (2006) Dietary putrescine reduces the intestinal anticarcinogenic activity of sulindac in a murine model of familial adenomatous polyposis. Nutr Cancer 56(2):172–181
Kee K, Foster BA, Merali S, Kramer DL, Hensen ML, Diegelman P, Kisiel N, Vujcic S, Mazurchuk RV, Porter CW (2004) Activated polyamine catabolism depletes acetyl-CoA pools and suppresses prostate tumor growth in TRAMP mice. J Biol Chem 279(38):40076–40083
Kobayashi M, Iseki K, Saitoh H, Miyazaki K (1992) Uptake characteristics of polyamines into rat intestinal brush-border membrane. Biochim Biophys Acta 1105(1):177–183
Kurata HT, Marton LJ, Nichols CG (2006) The polyamine binding site in inward rectifier K+ channels. J Gen Physiol 127(5):467–480
Lopatin AN, Makhina EN, Nichols CG (1994) Potassium channel block by cytoplasmic polyamines as the mechanism of intrinsic rectification. Nature 372(6504):366–369
Löser C, Eisel A, Harms D, Fölsch UR (1999) Dietary polyamines are essential luminal growth factors for small intestinal and colonic mucosal growth and development. Gut 44(1):12–16
Luk GD, Goodwin G, Marton LJ, Baylin SB (1981) Polyamines are necessary for the survival of human small-cell lung carcinoma in culture. Proc Natl Acad Sci USA 78(4):2355–2358
Luk GD, Goodwin G, Gazdar AF, Baylin SB (1982) Growth-inhibitory effects of DL-alpha-difluoromethylornithine in the spectrum of human lung carcinoma cells in culture. Cancer Res 42(8):3070–3073
Macarthur M, Hold GL, El-Omar EM (2004) Inflammation and Cancer II. Role of chronic inflammation and cytokine gene polymorphisms in the pathogenesis of gastrointestinal malignancy. Am J Physiol Gastrointest Liver Physiol 286(4):G515–G520
Mandal S, Mandal A, Johansson HE, Orjalo AV, Park MH (2013) Depletion of cellular polyamines, spermidine and spermine, causes a total arrest in translation and growth in mammalian cells. Proc Natl Acad Sci USA 110(6):2169–2174
Merentie M, Uimari A, Pietilä M, Sinervirta R, Keinänen TA, Vepsäläinen J, Khomutov A, Grigorenko N, Herzig KH, Jänne J, Alhonen L (2007) Oxidative stress and inflammation in the pathogenesis of activated polyamine catabolism-induced acute pancreatitis. Amino Acids 33(2):323–330
Murray-Stewart T, Wang Y, Devereux W, Casero RA Jr (2002) Cloning and characterization of multiple human polyamine oxidase splice variants that code for isoenzymes with different biochemical characteristics. Biochem J 368(Pt3):673–677
Murray-Stewart T, Wang Y, Goodwin A, Hacker A, Meeker A, Casero RA Jr (2008) Nuclear localization of human spermine oxidase isoforms-possible implications in drug response and disease etiology. FEBS J 275(11):2795–2806
Obayashi M, Matsui-Yuasa I, Matsumoto T, Kitano A, Kobayashi K, Otani S (1992) Polyamine metabolism in colonic mucosa from patients with ulcerative colitis. Am J Gastroenterol 87(6):736–740
O’Hagan HM, Wang W, Sen S, Destefano Shields C, Lee SS, Zhang YW, Clements EG, Cai Y, Van Neste L, Easwaran H, Casero RA Jr, Sears CL, Baylin SB (2011) Oxidative damage targets complexes containing DNA methyltransferases, SIRT1, and polycomb members to promoter CpG Islands. Cancer Cell 20(5):606–619
Park MH, Nishimura K, Zanelli CF, Valentini SR (2010) Functional significance of eIF5A and its hypusine modification in eukaryotes. Amino Acids 38(2):491–500
Pegg AE (2006) Regulation of ornithine decarboxylase. J Biol Chem 281(21):14529–14532
Pegg AE (2008) Spermidine/spermine-N(1)-acetyltransferase: a key metabolic regulator. Am J Physiol Endocrinol Metab 294(6):E995–E1010
Pegg AE (2009) S-Adenosylmethionine decarboxylase. Essays Biochem 46:25–45
Pegg AE, Casero RA Jr (2011) Current status of the polyamine research field. Methods Mol Biol 720:3–35
Pledgie-Tracy A, Billam M, Hacker A, Sobolewski MD, Woster PM, Zhang Z, Casero RA Jr, Davidson NE (2010) The role of the polyamine catabolic enzymes SSAT and SMO in the synergistic effects of standard chemotherapeutic agents with a polyamine analogue in human breast cancer cell lines. Cancer Chemother Pharmacol 65(6):1067–1081
Poulin R, Casero RA Jr, Soulet D (2012) Recent advances in the molecular biology of metazoan polyamine transport. Amino Acids 42(2–3):711–723
Raj KP, Zell JA, Rock CL, McLaren CE, Zoumas-Morse C, Gerner EW, Meyskens FL (2013) Role of dietary polyamines in a phase III clinical trial of difluoromethylornithine (DFMO) and sulindac for prevention of sporadic colorectal adenomas. Br J Cancer 108(3):512–518
Ray RM, Zimmerman BJ, McCormack SA, Patel TB, Johnson LR (1999) Polyamine depletion arrests cell cycle and induces inhibitors p21(Waf1/Cip1), p27(Kip1), and p53 in IEC-6 cells. Am J Physiol 276(3 Pt 1):C684–C691
Reuter S, Gupta SC, Chaturvedi MM, Aggarwal BB (2010) Oxidative stress, inflammation, and cancer: how are they linked? Free Radic Biol Med 49(11):1603–1616
Riddell RH, Goldman H, Ransohoff DF, Appelman HD, Fenoglio CM, Haggitt RC, Ahren C, Correa P, Hamilton SR, Morson BC et al (1983) Dysplasia in inflammatory bowel disease: standardized classification with provisional clinical applications. Hum Pathol 14(11):931–968
Ruschoff J, Wallinger S, Dietmaier W et al (1998) Aspirin suppresses the mutator phenotype associated with hereditary nonpolyposis colorectal cancer by genetic selection. Proc Natl Acad Sci USA 95:11301–11306
Schipper RG, Penning LC, Verhofstad AA (2000) Involvement of polyamines in apoptosis. Facts and controversies: effectors or protectors? Semin Cancer Biol 10(1):55–68
Schreck R, Rieber P, Baeuerle PA (1991) Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J 10(8):2247–2258
Smirnova OA, Isaguliants MG, Hyvonen MT, Keinanen TA, Tunitskaya VL, Vepsalainen J, Alhonen L, Kochetkov SN, Ivanov AV (2012) Chemically induced oxidative stress increases polyamine levels by activating the transcription of ornithine decarboxylase and spermidine/spermine-N1-acetyltransferase in human hepatoma HUH7 cells. Biochimie 94(9):1876–1883
ter Steege JC, Forget PP, Buurman WA (1999) Oral spermine administration inhibits nitric oxide-mediated intestinal damage and levels of systemic inflammatory mediators in a mouse endotoxin model. Shock 11(2):115–119
Tucker JM, Murphy JT, Kisiel N, Diegelman P, Barbour KW, Davis C, Medda M, Alhonen L, Jänne J, Kramer DL, Porter CW, Berger FG (2005) Potent modulation of intestinal tumorigenesis in Apcmin/+mice by the polyamine catabolic enzyme spermidine/spermine N1-acetyltransferase. Cancer Res 65(12):5390–5398
Turchanowa L, Dauletbaev N, Milovic V, Stein J (2001) Nonsteroidal anti-inflammatory drugs stimulate spermidine/spermine acetyltransferase and deplete polyamine content in colon cancer cells. Eur J Clin Invest 31(10):887–893
Wang Y, Devereux W, Woster P, Stewart T, Hacker A, Casero RA Jr (2001) Cloning and characterization of a human polyamine oxidase that is inducible by polyamine analogue exposure. Cancer Res 61(14):5370–5373
Wang Y, Murray-Stewart T, Devereux W, Hacker A, Frydman B, Woster PM, Casero RA Jr (2003) Properties of purified recombinant human polyamine oxidase, PAOh1/SMO. Biochem Biophys Res Commun 304(4):605–611
Wang Y, Hacker A, Murray-Stewart T, Fleischer JG, Woster PM, Casero RA Jr (2005) Induction of human spermine oxidase SMO(PAOh1) is regulated at the levels of new mRNA synthesis, mRNA stabilization and newly synthesized protein. Biochem J 386(Pt 3):543–547
Wang X, Feith DJ, Welsh P, Coleman CS, Lopez C, Woster PM, O’Brien TG, Pegg AE (2007) Studies of the mechanism by which increased spermidine/spermine N1-acetyltransferase activity increases susceptibility to skin carcinogenesis. Carcinogenesis 28(11):2404–2411
Weinberg F, Chandel NS (2009) Reactive oxygen species-dependent signaling regulates cancer. Cell Mol Life Sci 66(23):3663–3673
Weiss TS, Herfarth H, Obermeier F, Ouart J, Vogl D, Schölmerich J, Jauch KW, Rogler G (2004) Intracellular polyamine levels of intestinal epithelial cells in inflammatory bowel disease. Inflamm Bowel Dis 10(5):529–535
Williams K (1997) Modulation and block of ion channels: a new biology of polyamines. Cell Signal 9(1):1–13
Wu T, Ling KQ, Sayre LM, McIntire WS (2005) Inhibition of murine N1-acetylated polyamine oxidase by an acetylenic amine and the allenic amine, MDL 72527. Biochem Biophys Res Commun 326(2):483–490
Xie SQ, Zhang YH, Li Q, Wang JH, Li JH, Zhao J, Wang CJ (2011) COX-2-independent induction of apoptosis by celecoxib and polyamine naphthalimide conjugate mediated by polyamine depression in colorectal cancer cell lines. Int J Colorectal Dis 27(7):861–868
Xu H, Chaturvedi R, Cheng Y, Bussiere FI, Asim M, Yao MD, Potosky D, Meltzer SJ, Rhee JG, Kim SS, Moss SF, Hacker A, Wang Y, Casero RA Jr, Wilson KT (2004) Spermine oxidation induced by Helicobacter pylori results in apoptosis and DNA damage: implications for gastric carcinogenesis. Cancer Res 64(23):8521–8525
Yao Y, Fan Y, Wu J, Wan H, Wang J, Lam S, Lam WL, Girard L, Gazdar AF, Wu Z, Zhou Q (2012) Potential application of non-small cell lung cancer-associated autoantibodies to early cancer diagnosis. Biochem Biophys Res Commun 423(3):613–619
Yoshida M, Kashiwagi K, Shigemasa A, Taniguchi S, Yamamoto K, Makinoshima H, Ishihama A, Igarashi K (2004) A unifying model for the role of polyamines in bacterial cell growth, the polyamine modulon. J Biol Chem 279(44):46008–46013
Yuan Q, Ray RM, Johnson LR (2002) Polyamine depletion prevents camptothecin-induced apoptosis by inhibiting the release of cytochrome c. Am J Physiol Cell Physiol 282(6):C1290–C1297
Zahedi K, Wang Z, Barone S, Prada AE, Kelly CN, Casero RA Jr, Yokota N, Porter CW, Rabb H, Soleimani M (2003) Expression of SSAT, a novel biomarker of tubular cell damage, increases in kidney ischemia-reperfusion injury. Am J Physiol Renal Physiol 284(5):F1046–F1055
Zahedi K, Lentsch AB, Okaya T, Barone S, Sakai N, Witte DP, Arend LJ, Alhonen L, Jell J, Jänne J, Porter CW, Soleimani M (2009) Spermidine/spermine-N1-acetyltransferase ablation protects against liver and kidney ischemia-reperfusion injury in mice. Am J Physiol Gastrointest Liver Physiol 296(4):G899–G909
Zahedi K, Barone S, Kramer DL, Amlal H, Alhonen L, Jänne J, Porter CW, Soleimani M (2010a) The role of spermidine/spermine N1-acetyltransferase in endotoxin-induced acute kidney injury. Am J Physiol Cell Physiol 299(1):C164–C174
Zahedi K, Huttinger F, Morrison R, Murray-Stewart T, Casero RA Jr, Strauss KI (2010b) Polyamine catabolism is enhanced after traumatic brain injury. J Neurotrauma 27(3):515–525
Zahedi K, Barone SL, Xu J, Steinbergs N, Schuster R, Lentsch AB, Amlal H, Wang J, Casero RA Jr, Soleimani M (2012) Hepatocyte-specific ablation of spermine/spermidine-N1-acetyltransferase gene reduces the severity of CCl4-induced acute liver injury. Am J Physiol Gastrointest Liver Physiol 303(5):G546–G560
Zell JA, McLaren CE, Chen WP, Thompson PA, Gerner EW, Meyskens FL (2010) Ornithine decarboxylase-1 polymorphism, chemoprevention with eflornithine and sulindac, and outcomes among colorectal adenoma patients. J Natl Cancer Inst 102(19):1513–1516
Zhang M, Caragine T, Wang H, Cohen PS, Botchkina G, Soda K, Bianchi M, Ulrich P, Cerami A, Sherry B, Tracey KJ (1997) Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: a counterregulatory mechanism that restrains the immune response. J Exp Med 185(10):1759–1768
Acknowledgments
Portions of the work described in this manuscript were supported by the NIEHS T32 training grant ES07141 and NCI grants CA51085 and CA98454.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
V. Battaglia and C. DeStefano Shields contributed equally to this work.
Rights and permissions
About this article
Cite this article
Battaglia, V., DeStefano Shields, C., Murray-Stewart, T. et al. Polyamine catabolism in carcinogenesis: potential targets for chemotherapy and chemoprevention. Amino Acids 46, 511–519 (2014). https://doi.org/10.1007/s00726-013-1529-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-013-1529-6