Abstract
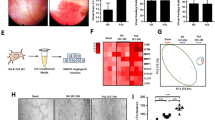
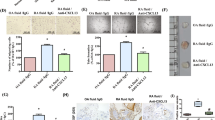
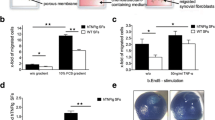
IL-11 has been detected in inflamed joints; however, its role in the pathogenesis of arthritis is not yet clear. Studies were conducted to characterize the expression and functional significance of IL-11 and IL-11Rα in rheumatoid arthritis (RA). IL-11 levels were elevated in RA synovial fluid (SF) compared to osteoarthritis (OA) SF and plasma from RA, OA and normal individuals (NLs). Morphologic studies established that IL-11 was detected in lining fibroblasts and macrophages in addition to sublining endothelial cells and macrophages at higher levels in RA compared to NL synovial tissues. Since IL-11Rα was exclusively expressed in RA fibroblasts and endothelial cells, macrophages were not involved in IL-11 effector function. Ligation of IL-11 to IL-11Rα strongly provoked fibroblast infiltration into RA joint, while cell proliferation was unaffected by this process. Secretion of IL-8 and VEGF from IL-11 activated RA fibroblasts was responsible for the indirect effect of IL-11 on endothelial cell transmigration and tube formation. Moreover, IL-11 blockade impaired RA SF capacity to elicit endothelial cell transmigration and tube formation. We conclude that IL-11 binding to endothelial IL-11Rα can directly induce RA angiogenesis. In addition, secretion of proangiogenic factors from migrating fibroblasts potentiated by IL-11 can indirectly contribute to RA neovascularization.







Similar content being viewed by others
References
Heinrich PC, Behrmann I, Haan S et al (2003) Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J 374:1–20
Putoczki TL, Thiem S, Loving A et al (2013) Interleukin-11 is the dominant IL-6 family cytokine during gastrointestinal tumorigenesis and can be targeted therapeutically. Cancer Cell 24:257–271
Paul SR, Bennett F, Calvetti JA et al (1990) Molecular cloning of a cDNA encoding interleukin 11, a stromal cell-derived lymphopoietic and hematopoietic cytokine. Proc Natl Acad Sci USA 87:7512–7516
Paul SR, Yang YC, Donahue RE et al (1991) Stromal cell-associated hematopoiesis: immortalization and characterization of a primate bone marrow-derived stromal cell line. Blood 77:1723–1733
Du X, Williams DA (1997) Interleukin-11: review of molecular, cell biology, and clinical use. Blood 89:3897–3908
Xu DH, Zhu Z, Wakefield MR et al (2016) The role of IL-11 in immunity and cancer. Cancer Lett 373:156–163
Quesniaux VF, Clark SC, Turner K et al (1992) Interleukin-11 stimulates multiple phases of erythropoiesis in vitro. Blood 80:1218–1223
Jacobsen SE, Okkenhaug C, Veiby OP et al (1994) Interleukin 13: novel role in direct regulation of proliferation and differentiation of primitive hematopoietic progenitor cells. J Exp Med 180:75–82
Orazi A, Cooper RJ, Tong J et al (1996) Effects of recombinant human interleukin-11 (Neumega rhIL-11 growth factor) on megakaryocytopoiesis in human bone marrow. Exp Hematol 24:1289–1297
Putoczki T, Ernst M (2010) More than a sidekick: the IL-6 family cytokine IL-11 links inflammation to cancer. J Leukoc Biol 88:1109–1117
Taniguchi K, Karin M (2014) IL-6 and related cytokines as the critical lynchpins between inflammation and cancer. Semin Immunol 26:54–74
Yoshizaki A, Nakayama T, Yamazumi K et al (2006) Expression of interleukin (IL)-11 and IL-11 receptor in human colorectal adenocarcinoma: IL-11 up-regulation of the invasive and proliferative activity of human colorectal carcinoma cells. Int J Oncol 29:869–876
Yamazumi K, Nakayama T, Kusaba T et al (2006) Expression of interleukin-11 and interleukin-11 receptor alpha in human colorectal adenocarcinoma; immunohistochemical analyses and correlation with clinicopathological factors. World J Gastroenterol 12:317–321
Zhang X, Tao Y, Chopra M et al (2015) IL-11 induces Th17 cell responses in patients with early relapsing-remitting multiple sclerosis. J Immunol 194:5139–5149
Walmsley M, Butler DM, Marinova-Mutafchieva L et al (1998) An anti-inflammatory role for interleukin-11 in established murine collagen-induced arthritis. Immunology 95:31–37
Wong PK, Campbell IK, Robb L et al (2005) Endogenous IL-11 is pro-inflammatory in acute methylated bovine serum albumin/interleukin-1-induced (mBSA/IL-1)arthritis. Cytokine 29:72–76
Chung SJ, Kwon YJ, Park MC et al (2011) The correlation between increased serum concentrations of interleukin-6 family cytokines and disease activity in rheumatoid arthritis patients. Yonsei Med J 52:113–120
Moreland L, Gugliotti R, King K et al (2001) Results of a phase-I/II randomized, masked, placebo-controlled trial of recombinant human interleukin-11 (rhIL-11) in the treatment of subjects with active rheumatoid arthritis. Arthritis Res 3:247–252
Arnett FC, Edworthy SM, Bloch DA et al (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheumatol 31:315–324
Pickens SR, Chamberlain ND, Volin MV et al (2011) Characterization of CCL19 and CCL21 in rheumatoid arthritis. Arthritis Rheumatol 63:914–922
Pickens SR, Chamberlain ND, Volin MV et al (2011) Characterization of interleukin-7 and interleukin-7 receptor in the pathogenesis of rheumatoid arthritis. Arthritis Rheumatol 63:2884–2893
Chamberlain ND, Vila OM, Volin MV et al (2012) TLR5, a novel and unidentified inflammatory mediator in rheumatoid arthritis that correlates with disease activity score and joint TNF-alpha levels. J Immunol 189:475–483
Kim SJ, Chen Z, Essani AB, Elshabrawy HA, Volin MV, Volkov S, Swedler W, Arami S, Sweiss N, Shahrara S (2015) Identification of a novel TLR7 endogenous ligand in RA synovial fluid that can provoke arthritic joint inflammation. Arthritis Rheumatol 68(5):1099–1110. https://doi.org/10.1002/art.39544
Chen Z, Kim SJ, Chamberlain ND et al (2013) The novel role of IL-7 ligation to IL-7 receptor in myeloid cells of rheumatoid arthritis and collagen-induced arthritis. J Immunol 190:5256–5266
Shahrara S, Pickens SR, Mandelin AM 2nd et al (2010) IL-17-mediated monocyte migration occurs partially through CC chemokine ligand 2/monocyte chemoattractant protein-1 induction. J Immunol 184:4479–4487
Morgan R, Endres J, Behbahani-Nejad N et al (2015) Expression and function of aminopeptidase N/CD13 produced by fibroblast-like synoviocytes in rheumatoid arthritis: role of CD13 in chemotaxis of cytokine-activated T cells independent of enzymatic activity. Arthritis Rheumatol 67:74–85
Curtis DJ, Hilton DJ, Roberts B et al (1997) Recombinant soluble interleukin-11 (IL-11) receptor alpha-chain can act as an IL-11 antagonist. Blood 90:4403–4412
Ota F, Maeshima A, Yamashita S et al (2003) Activin A induces cell proliferation of fibroblast-like synoviocytes in rheumatoid arthritis. Arthritis Rheumatol 48:2442–2449
Pickens SR, Volin MV, Mandelin AM 2nd et al (2010) IL-17 contributes to angiogenesis in rheumatoid arthritis. J Immunol 184:3233–3241
Pickens SR, Chamberlain ND, Volin MV et al (2012) Role of the CCL21 and CCR7 pathways in rheumatoid arthritis angiogenesis. Arthritis Rheumatol 64:2471–2481
Chen Z, Kim SJ, Essani AB et al (2015) Characterising the expression and function of CCL28 and its corresponding receptor, CCR10, in RA pathogenesis. Ann Rheum Dis 74:1898–1906
Lefevre S, Knedla A, Tennie C et al (2009) Synovial fibroblasts spread rheumatoid arthritis to unaffected joints. Nat Med 15:1414–1420
Bartok B, Firestein GS (2010) Fibroblast-like synoviocytes: key effector cells in rheumatoid arthritis. Immunol Rev 233:233–255
Elshabrawy HA, Chen Z, Volin MV et al (2015) The pathogenic role of angiogenesis in rheumatoid arthritis. Angiogenesis 18:433–448
Taki H, Sugiyama E, Mino T et al (1998) Differential inhibitory effects of indomethacin, dexamethasone, and interferon-gamma (IFN-gamma) on IL-11 production by rheumatoid synovial cells. Clin Exp Immunol 112:133–138
Taki H, Sugiyama E, Kuroda A et al (2000) Interleukin-4 inhibits interleukin-11 production by rheumatoid synovial cells. Rheumatology (Oxford) 39:728–731
Mino T, Sugiyama E, Taki H et al (1998) Interleukin-1 alpha and tumor necrosis factor alpha synergistically stimulate prostaglandin E2-dependent production of interleukin-11 in rheumatoid synovial fibroblasts. Arthritis Rheumatol 41:2004–2013
Okamoto H, Yamamura M, Morita Y et al (1997) The synovial expression and serum levels of interleukin-6, interleukin-11, leukemia inhibitory factor, and oncostatin M in rheumatoid arthritis. Arthritis Rheumatol 40:1096–1105
Trontzas P, Kamper EF, Potamianou A et al (1998) Comparative study of serum and synovial fluid interleukin-11 levels in patients with various arthritides. Clin Biochem 31:673–679
Hermann JA, Hall MA, Maini RN et al (1998) Important immunoregulatory role of interleukin-11 in the inflammatory process in rheumatoid arthritis. Arthritis Rheumatol 41:1388–1397
Yamashita A, Yonemitsu Y, Okano S et al (2002) Fibroblast growth factor-2 determines severity of joint disease in adjuvant-induced arthritis in rats. J Immunol 168:450–457
Koch AE, Kunkel SL, Shah MR et al (1995) Growth-related gene product alpha. A chemotactic cytokine for neutrophils in rheumatoid arthritis. J Immunol 155:3660–3666
Koch AE, Kunkel SL, Harlow LA et al (1994) Epithelial neutrophil activating peptide-78: a novel chemotactic cytokine for neutrophils in arthritis. J Clin Invest 94:1012–1018
Girasole G, Passeri G, Jilka RL et al (1994) Interleukin-11: a new cytokine critical for osteoclast development. J Clin Invest 93:1516–1524
Yin TG, Schendel P, Yang YC (1992) Enhancement of in vitro and in vivo antigen-specific antibody responses by interleukin 11. J Exp Med 175:211–216
Trepicchio WL, Bozza M, Pedneault G et al (1996) Recombinant human IL-11 attenuates the inflammatory response through down-regulation of proinflammatory cytokine release and nitric oxide production. J Immunol 157:3627–3634
Trepicchio WL, Wang L, Bozza M et al (1997) IL-11 regulates macrophage effector function through the inhibition of nuclear factor-kappaB. J Immunol 159:5661–5670
Lee SY, Kwok SK, Son HJ et al (2013) IL-17-mediated Bcl-2 expression regulates survival of fibroblast-like synoviocytes in rheumatoid arthritis through STAT3 activation. Arthritis Res Ther 15:R31
Xing R, Yang L, Jin Y et al (2016) Interleukin-21 induces proliferation and proinflammatory cytokine profile of fibroblast-like synoviocytes of patients with rheumatoid arthritis. Scand J Immunol 83:64–71
Zhu J, Jia E, Zhou Y et al (2015) Interleukin-22 secreted by NKp44+ natural killer cells promotes proliferation of fibroblast-like synoviocytes in rheumatoid arthritis. Medicine (Baltimore) 94:e2137
Calmon-Hamaty F, Combe B, Hahne M et al (2011) Lymphotoxin alpha stimulates proliferation and pro-inflammatory cytokine secretion of rheumatoid arthritis synovial fibroblasts. Cytokine 53:207–214
Elias JA, Zheng T, Whiting NL et al (1994) IL-1 and transforming growth factor-beta regulation of fibroblast-derived IL-11. J Immunol 152:2421–2429
Guo X, Pan Y, Xiao C et al (2012) Fractalkine stimulates cell growth and increases its expression via NF-kappaB pathway in RA-FLS. Int J Rheum Dis 15:322–329
Garcia-Vicuna R, Gomez-Gaviro MV, Dominguez-Luis MJ et al (2004) CC and CXC chemokine receptors mediate migration, proliferation, and matrix metalloproteinase production by fibroblast-like synoviocytes from rheumatoid arthritis patients. Arthritis Rheumatol 50:3866–3877
Edhayan G, Ohara RA, Stinson WA et al (2016) Inflammatory properties of inhibitor of DNA binding 1 secreted by synovial fibroblasts in rheumatoid arthritis. Arthritis Res Ther 18:87
Acknowledgements
This work was supported in part by awards from Department of Veteran’s Affairs MERIT Award 1I01BX002286, the National Institutes of Health R43AG057312, AR056099 and AR065778, funding provided by Department of Defense PR093477. We would like to acknowledge Dr. Rhonda Kineman for critically reviewing the paper and granting us access to use her laboratory microscope.
Author information
Authors and Affiliations
Contributions
HAE, SS designed the research; HAE, MVV, ABE, ZC, SJK, KV, KP, SS performed the research; HAE, ZC, MVV, KV, MK, HMA, SJK, IBM, DAF, SS analyzed the data; SA, GZ: Provided reagents; all the authors contributed to writing the paper: Writing the paper.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Elshabrawy, H.A., Volin, M.V., Essani, A.B. et al. IL-11 facilitates a novel connection between RA joint fibroblasts and endothelial cells. Angiogenesis 21, 215–228 (2018). https://doi.org/10.1007/s10456-017-9589-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-017-9589-y