Abstract
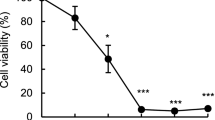
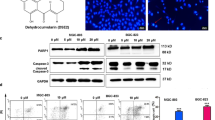
Myrtucommulone (MC) is a unique, nonprenylated acylphloroglucinol contained in the leaves of myrtle (Myrtus communis). Here, we addressed the potential of MC to induce apoptosis of cancer cells. MC potently induced cell death of different cancer cell lines (EC50 3–8 μM) with characteristics of apoptosis, visualized by the activation of caspase-3, -8 and -9, cleavage of poly(ADP-ribose)polymerase (PARP), release of nucleosomes into the cytosol, and DNA fragmentation. MC was much less cytotoxic for non-transformed human peripheral blood mononuclear cells (PBMC) or foreskin fibroblasts (EC50 cell death = 20–50 μM), and MC up to 30 μM hardly caused processing of PARP, caspase-3, -8 and -9 in human PBMC. MC-induced apoptosis was mediated by the intrinsic rather than the extrinsic death pathway. Thus, MC caused loss of the mitochondrial membrane potential in MM6 cells and evoked release of cytochrome c from mitochondria. Interestingly, Jurkat cells deficient in caspase-9 were resistant to MC-induced cell death and no processing of PARP or caspase-8 was evident. In cell lines deficient in either CD95 (Fas, APO-1) signalling, FADD or caspase-8, MC was still able to potently induce cell death and PARP cleavage. Conclusively, MC induces apoptosis in cancer cell lines, with marginal cytotoxicity for non-transformed cells, via the mitochondrial cytochrome c/Apaf-1/caspase-9 pathway.






Similar content being viewed by others
Abbreviations
- CHX:
-
Cycloheximide
- FADD:
-
Fas-associated death domain
- IBP-C:
-
Isobutyrophenone core
- 5-LO:
-
5-Lipoxygenase
- MC:
-
Myrtucommulone
- ΔΨ m :
-
Mitochondrial membrane potential
- MM6:
-
Mono Mac 6
- PARP:
-
Poly(ADP-ribose)polymerase
- PBMC:
-
Peripheral blood mononuclear cells
- S-MC:
-
Semi-myrtucommulone
- TNF:
-
Tumour necrosis factor
References
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116:205–219
Thompson CB (1995) Apoptosis in the pathogenesis and treatment of disease. Science 267:1456–1462
Fesik SW (2005) Promoting apoptosis as a strategy for cancer drug discovery. Nat Rev Cancer 5:876–885
Nicholson DW (2000) From bench to clinic with apoptosis-based therapeutic agents. Nature 407:810–816
Tan G, Gyllenhaal C, Soejarto DD (2006) Biodiversity as a source of anticancer drugs. Curr Drug Targets 7:265–277
Nagata S (1997) Apoptosis by death factor. Cell 88:355–365
Green DR, Kroemer G (2004) The pathophysiology of mitochondrial cell death. Science 305:626–629
Thornberry NA, Lazebnik Y (1998) Caspases: enemies within. Science 281:1312–1316
Galluzzi L, Larochette N, Zamzami N et al (2006) Mitochondria as therapeutic targets for cancer chemotherapy. Oncogene 25:4812–4830
Al-Saimary IE, Bakr SS, Jaffar T et al (2002) Effects of some plant extracts and antibiotics on Pseudomonas aeruginosa isolated from various burn cases. Saudi Med J 23:802–805
Elfellah MS, Akhter MH, Khan MT (1984) Anti-hyperglycaemic effect of an extract of Myrtus communis in streptozotocin-induced diabetes in mice. J Ethnopharmacol 11:275–281
Onal S, Timur S, Okutucu B et al (2005) Inhibition of alpha-glucosidase by aqueous extracts of some potent antidiabetic medicinal herbs. Prep Biochem Biotechnol 35:29–36
Levesque H, Lafont O (2000) Aspirin throughout the ages: a historical review. Rev Med Interne 21(Suppl 1):8s–17s
Rosa A, Deiana M, Casu V et al (2003) Antioxidant activity of oligomeric acylphloroglucinols from Myrtus communis L. Free Radic Res 37:1013–1019
Appendino G, Bianchi F, Minassi A et al (2002) Oligomeric acylphloroglucinols from myrtle (Myrtus communis). J Nat Prod 65:334–338
Feisst C, Franke L, Appendino G et al (2005) Identification of molecular targets of the oligomeric nonprenylated acylphloroglucinols from Myrtus communis and their implication as anti-inflammatory compounds. J Pharmacol Exp Ther 315:389–396
Samraj AK, Sohn D, Schulze-Osthoff K et al (2007) Loss of caspase-9 reveals its essential role for caspase-2 activation and mitochondrial membrane depolarization. Mol Biol Cell 18:84–93
Cinatl J, Herneiz P, Rabenau H et al (1994) Induction of myogenic differentiation in a human rhabdomyosarcoma cell line by phenylacetate. Cancer Lett 78:41–48
Kotchetkov R, Driever PH, Cinatl J et al (2005) Increased malignant behavior in neuroblastoma cells with acquired multi-drug resistance does not depend on P-gp expression. Int J Oncol 27:1029–1037
Michaelis M, Kohler N, Reinisch A et al (2004) Increased human cytomegalovirus replication in fibroblasts after treatment with therapeutical plasma concentrations of valproic acid. Biochem Pharmacol 68:531–538
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Michaelis M, Suhan T, Cinatl J et al (2004) Valproic acid and interferon-alpha synergistically inhibit neuroblastoma cell growth in vitro and in vivo. Int J Oncol 25:1795–1799
Nicoletti I, Migliorati G, Pagliacci MC et al (1991) A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J Immunol Methods 139: 271–279
Zwelling LA, Altschuler E, Cherif A et al (1991) N-(5,5-diacetoxypentyl)doxorubicin: a novel anthracycline producing DNA interstrand cross-linking and rapid endonucleolytic cleavage in human leukemia cells. Cancer Res 51:6704–6707
Lazebnik YA, Kaufmann SH, Desnoyers S et al (1994) Cleavage of poly(ADP-ribose) polymerase by a proteinase with properties like ICE. Nature 371:346–347
Werz O, Steinhilber D (2006) Therapeutic options for 5-lipoxygenase inhibitors. Pharmacol Ther 112:701–718
Budihardjo I, Oliver H, Lutter M et al (1999) Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol 15:269–290
Peterson EJ, Latinis KM, Koretzky GA (1998) Molecular characterization of a CD95 signaling mutant. Arthritis Rheum 41:1047–1053
Ghaemmaghami M, Jett JR (1998) New agents in the treatment of small cell lung cancer. Chest 113:86S–91S
Cragg GM, Newman DJ (2005) Plants as a source of anti-cancer agents. J Ethnopharmacol 100:72–79
Brown JM, Attardi LD (2005) The role of apoptosis in cancer development and treatment response. Nat Rev Cancer 5:231–237
Loo DT, Rillema JR (1998) Measurement of cell death. Methods Cell Biol 57:251–264
Luo X, Budihardjo I, Zou H et al (1998) Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94:481–490
Maccarrone M, Taccone-Gallucci M and Finazzi-Agro A (2003) 5-Lipoxygenase-mediated mitochondrial damage and apoptosis of mononuclear cells in ESRD patients. Kidney Int Suppl:S33–S36
Ding XZ, Iversen P, Cluck MW et al (1999) Lipoxygenase inhibitors abolish proliferation of human pancreatic cancer cells. Biochem Biophys Res Commun 261:218–223
Ghosh J, Myers CE (1997) Arachidonic acid stimulates prostate cancer cell growth: critical role of 5-lipoxygenase. Biochem Biophys Res Commun 235:418–423
Romano M, Catalano A, Nutini M et al (2001) 5-lipoxygenase regulates malignant mesothelial cell survival: involvement of vascular endothelial growth factor. FASEB J 15:2326–2336
Benchimol S (2001) p53-dependent pathways of apoptosis. Cell Death Differ 8:1049–1051
Lowe SW, Ruley HE, Jacks T et al (1993) p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74:957–967
Brown JM, Wouters BG (1999) Apoptosis, p53, and tumor cell sensitivity to anticancer agents. Cancer Res 59:1391–1399
Wolf D, Rotter V (1985) Major deletions in the gene encoding the p53 tumor antigen cause lack of p53 expression in HL-60 cells. Proc Natl Acad Sci USA 82:790–794
Rokhlin OW, Bishop GA, Hostager BS et al (1997) Fas-mediated apoptosis in human prostatic carcinoma cell lines. Cancer Res 57:1758–1768
Strasser A (2005) The role of BH3-only proteins in the immune system. Nat Rev Immunol 5:189–200
Author information
Authors and Affiliations
Corresponding author
Additional information
I. Tretiakova and D. Blaesius contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tretiakova, I., Blaesius, D., Maxia, L. et al. Myrtucommulone from Myrtus communis induces apoptosis in cancer cells via the mitochondrial pathway involving caspase-9. Apoptosis 13, 119–131 (2008). https://doi.org/10.1007/s10495-007-0150-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-007-0150-0