Abstract
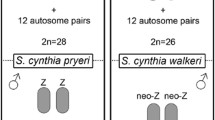
The W chromosome of the silkworm Bombyx mori is devoid of functional genes, except for the putative female-determining gene (Fem). To localize Fem, we investigated the presence of W-specific DNA markers on strains in which an autosomal fragment containing dominant marker genes was attached to the W chromosome. We produced new W-chromosomal fragments from the existing Zebra-W strain (T(W;3)Ze chromosome) by X-irradiation, and then carried out deletion mapping of these and sex-limited yellow cocoon strains (T(W;2)Y-Chu, -Abe and -Ban types) from different Japanese stock centers. Of 12 RAPD markers identified in the normal W chromosomes of most silkworm strains in Japan, the newly irradiated W(B-YL-YS)Ze chromosome contained three, the T(W;2)Y-Chu chromosome contained six, and the T(W;2)Y-Abe and -Ban chromosomes contained only one (W-Rikishi). To investigate the ability of the reduced W-chromosome translocation fragments to form heterochromatin bodies, which are found in nuclei of normal adult female sucking stomachs, we examined cells of the normal type p50 strain and the T(W;2)Y-Chu and -Abe strains. A single sex heterochromatin body was found in nuclei of p50 females, whereas we detected only small sex heterochromatin bodies in the T(W;2)Y-Chu strain and no sex heterochromatin body in the T(W;2)Y-Abe strain. Since adult females of all strains were normal and fertile, we conclude that only extremely limited region, containing the W-Rikishi RAPD sequence of the W chromosome, is required to determine femaleness. Based on a comparison of the normal W-chromosome and 7 translocation and W-deletion strains we present a map of Fem relative to the 12 W-specific RAPD markers.






Similar content being viewed by others
References
Abe H, Ohbayashi F, Harada T, Shimada T, Yokoyama T, Kobayashi M, Oshiki T (1996) An application of DNA diagnosis method for preservation of the susceptible strain to densonucleosis virus type-1 of the silkworm, Bombyx mori. J Seric Sci Jpn 65:196–299 (in Japanese with English summary)
Abe H, Kanehara M, Terada T, Ohbayashi F, Shimada T, Kawai S, Suzuki M, Sugasaki T, Oshiki T (1998a) Identification of novel random amplified polymorphic DNAs (RAPDs) on the W chromosome of the domesticated silkworm, Bombyx mori, and the wild silkworm, B. mandarina, and their retrotransposable element-related nucleotide sequences. Genes Genet Syst 73:243–254
Abe H, Ohbayashi F, Shimada T, Sugasaki T, Kawai S, Oshiki T (1998b) A complete full-length non-LTR retrotransposon, BMC1, on the W chromosome of the silkworm, Bombyx mori. Genes Genet Syst 73:353–358
Abe H, Ohbayashi F, Sugasaki T, Kanehara M, Terada T, Shimada T, Kawai S, Mita K, Kanamori Y, Yamamoto M-T, Oshiki T (2001) Two novel Pao-like retrotransposons (Kamikaze and Yamato) from the silkworm species Bombyx mori and B. mandarina: common structural features of Pao-like elements. Mol Genet Genomics 265:375–385
Abe H, Sugasaki T, Terada T, Kanehara M, Ohbayashi F, Shimada T, Kawai S, Mita K, Oshiki T (2002) Nested retrotransposons on the W chromosome of the wild silkworm Bombyx mandarina. Insect Mol Biol 11:307–314
Abe H, Mita K, Yasukochi Y, Oshiki T, Shimada T (2005a) Retrotransposable elements on the W chromosome of the silkworm, Bombyx mori. Cytogenet Genome Res 110:144–151
Abe H, Seki M, Ohbayashi F, Tanaka N, Yamashita J, Fujii T, Yokoyama T, Takahashi M, Banno Y, Sahara K, Yoshido A, Ihara J, Yasukochi Y, Mita K, Ajimura M, Suzuki MG, Oshiki T, Shimada T (2005b) Partial deletions of the W chromosome due to reciprocal translocation in the silkworm Bombyx mori. Insect Mol Biol 14:339–352
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Charlesworth D, Charlesworth B, Marais G (2005) Steps in evolution of heteromorphic sex chromosomes. Heredity 95:118–128
Doira H (1978) Genetic stocks of the silkworm. In: Tazima Y (ed) The silkworm, an important laboratory tool. Kodansha Ltd., Japan
Doira H (1992) Genetical stocks and mutations of Bombyx mori. In: Doira H (ed) Important genetic resources. Silkworm Genetic Division, Institute of Genetic resources, Faculty of Agriculture, Kyusyu University, Fukuoka, Japan, pp 1–73
Ennis TJ (1976) Sex chromatin and chromosome numbers in Lepidoptera. Can J Cytol 18:119–130
Fujii H, Banno Y, Doira H, Kihara H, Kawaguchi Y (1998) Genetical stocks and mutations of Bombyx mori: Important genetic resources, 2nd edn. Institute of Genetic Resources, Faculty of Agriculture, Kyusyu University, Fukuoka
Fujii T, Tanaka N, Yokoyama T, Ninaki O, Oshiki T, Ohnuma A, Tazima Y, Banno Y, Ajimura M, Mkita K, Seki M, Ohbayashi F, Shimada T, Abe H (2006) The female-killing chromosome of the silkworm, Bombyx mori, was generated by translocation between the Z and W chromosomes. Genetica 127:253–265
Fujii T, Yokoyama T, Ninagi O, Kakehashi H, Obara Y, Nenoi M, Ishikawa T, Mita K, Shimada T, Abe H (2007) Isolation and characterization of sex chromosome rearrangements generating male muscle dystrophy and female abnormal oogenesis in the silkworm, Bombyx mori. Genetica 130:267–280
Goldsmith MR (1995) Genetics of the silkworm: revisiting an ancient model system. In: Goldsmith MR, Wilkins AS (eds) Molecular model systems in Lepidoptera. Cambridge University Press, New York, pp 21–76
Goldsmith MR, Shimada T, Abe H (2005) The genetics and genomics of the silkworm, Bombyx mori. Annu Rev Entomol 50:71–100
Hashimoto H (1933) The role of the W-chromosome in the sex determination of Bombyx mori. Jpn J Genet 8:245–247 (in Japanese)
Hashimoto H (1948) Sex-limited zebra, an X-ray mutation in the silkworm. J Seric Sci Jpn 16:62–64
Ito S (1977) Cytogenetical studies on the chromosomes of silk gland cells of the silkworm with special reference to the structure and behavior of the sex chromosomes. Jpn J Genet 52:327–340
Kimura K, Harada C, Aoki H (1971) Studies on the W-translocation of yellow blood gene in the silkworm (Bombyx mori). Jpn J Breed 21:199–203 (in Japanese with English summary)
Koike Y, Mita K, Suzuki MG, Maeda S, Abe H, Osoegawa K, deJong PJ, Shimada T (2003) Genomic sequence of a 320-kb segment of the Z chromosome of Bombyx mori containing a kettin ortholog. Mol Genet Genomics 269:137–149
Marec F, Traut W (1994) Sex chromosome paring and sex chromatin bodies in W-Z translocation strains of Ephestia kuehniella (Lepidoptera). Genome 37:426–435
Miao XX, Xub SJ, Li MH, Li MW, Huang JH, Dai FY, marino SW, Mills DR, Zeng P, Mita K, Jia SH, Zhang Y, Liu WB, Xiang H, Guo QH, Xu AY, Kong XY, Lin HX, Shi YZ, Lu G, Zhang X, Huang W, Yasukochi Y, Sugasaki T, Shimada T, Nagaraju J, Xiang ZH, Wang SY, Goldsmith MR, Lu C, Zhao GP, Huang YP (2005) Simple sequence repeat-based consensus linkage map of Bombyx mori. Proc Natl Acad Sci USA 102:16303–16308
Mita K, Kasahara M, Sasaki S, Nagayasu Y, Yamada T, Kanamori H, Namiki N, Kitagawa M, Yamashita H, Yasukochi Y, Kadono-Okuda K, Yamamoto K, Ajimura M, Ravikumar G, Shimomura M, Nagamura Y, Shin IT, Abe H, Shimada T, Morishita S, Sasaki T (2004) The genome sequence of silkworm, Bombyx mori. DNA Res 11:27–35
Niino T, Eguchi R, Shimazaki A, Shibukawa A (1988) Breakage by γ-rays of the +i-lem locus on the translocated 2nd chromosome in the sex-limited yellow cocoon silkworm. J Seric Sci Jpn 57:75–76
Niino T, Kanda T, Eguchi R, Shimazaki A, Shibukawa A (1987) Defects and structure of translocated chromosome in the sex-limited yellow cocoon strain of the silkworm, Bombyx mori. J Seric Sci Jpn 56:240–246 (in Japanese with English summary)
Ogura T, Okano K, Tsuchida K, Miyajima N, Tanaka H, Takeda N, Izumi S, Tomino S, Maekawa H (1994) A defective non-LTR retrotransposon is dispersed throughout the genome of the silkworm, Bombyx mori. Chromosoma 103:311–323
Pan QZ, Sugai E, Oshiki T (1986) Sex-chromatin in cell nuclei of abdominal leg of the silkworm, Bombyx mori. J Seric Sci Jpn 55:483–487 (in Japanese with English summary)
Pan QZ, Sugai E, Oshiki T (1987) Sex-chromatin observation in cell nuclei of tissues of the silkworm moth, Bombyx mori. J Seric Sci Jpn 56:436–439 (in Japanese with English summary)
Paran I, Michelmore W (1993) Development of reliable PCR-based markers linked to downy mildew resistance genes in lettuce. Theor Appl Genet 85:985–993
Promboon A, Shimada T, Fujiwara H, Kobayashi M (1995) Linkage map of random amplified polymorphic DNAs (RAPDs) in the silkworm, Bombyx mori. Genet Res Camb 66:1–7
Sahara K, Yoshido A, Kawamura N, Ohnuma A, Abe H, Mita K, Oshiki T, Shimada T, Asano SI, Bando H, Yasukochi Y (2003) W-derived BAC probes as a new tool for identification of the W chromosome and its aberrations in Bombyx mori. Chromosoma 112:48–55
Sakaguchi B (1978) Gametogenesis, fertilization and embryogenesis of the silkworm: In: Tazima Y (ed) The silkworm, an important laboratory tool. Kodansha Ltd., Japan
Tan YD, Wan C, Zhu Y, Lu C, Xiang Z, Deng HW (2001) An amplified fragment length polymorphism map of the silkworm. Genetics 157:1277–1284
Tanaka N, Yokoyama Y, Irobe Y, Abe H, Ninagi O, Oshiki T (2000) Breakage and subsequent return for W chromosome-p B fragment-fifth linkage group fusion chromosome in the sex-limited p B silkworm strain (TWPB), Bombyx mori. J Seric Sci Jpn 69:327–330
Tanaka Y (1916) Genetic studies in the silkworm. J Coll Agric Sapporo 6:1–33
Tazima Y (1941) A simple method of sex discrimination by means of larval markings in Bombyx mori. J Seric Sci Jpn 12:184–188 (in Japanese)
Tazima Y (1944) Studies on chromosome aberrations in the silkworm. II. Translocation involving second and W-chromosomes. Bull Seric Exp Stn 12:109–181 (in Japanese with English summary)
Tazima Y, Harada C, Ohta N (1951) On the sex discriminating method by colouring genes of silkworm eggs. I. Induction of translocation between the W and tenth chromosomes. Jpn J Breed 1:47–50 (in Japanese with English summary)
Traut W, Weith A, Traut G (1986) Structural mutants of the W chromosome in Ephestia (Insecta, Lepidoptera). Genetica 70:69–79
Traut W, Marec F (1996) Sex chromatin in Lepidoptera. Quart Rev Biol 71:239–256
Wu C, Asakawa S, Shimizu N, Kawasaki S, Yasukochi Y (1999) Construction and characterization of bacterial artificial chromosome libraries from the silkworm, Bombyx mori. Mol Gen Genet 261:698–706
Xia Q, Zhou Z, Lu C, Cheng D, Dai F, Li B, Zhao P, Zha X, Cheng T, Chai C, Pan G, Xu J, Liu C, Lin Y, Qian J, Hou Y, Wu Z, Li G, Pan M, Li C, Shen Y, Lan X, Yuan L, Li T, Xu H, Yang G, Wan Y, Zhu Y, Yu M, Shen W, Wu D, Xiang Z, Yu J, Wang J, Li R, Shi J, Li H, Li G, Su J, Wang X, Li G, Zhang Z, Wu Q, Li J, Zhang Q, Wei N, Xu J, Sun H, Dong L, Liu D, Zhao S, Zhao X, Meng Q, Lan F, Huang X, Li Y, Fang L, Li C, Li D, Sun Y, Zhang Z, Yang Z, Huang Y, Xi Y, Qi Q, He D, Huang H, Zhang X, Wang Z, Li W, Cao Y, Yu Y, Yu H, Li J, Ye J, Chen H, Zhou Y, Liu B, Wang J, Ye J, Ji H, Li S, Ni P, Zhang J, Zhang Y, Zheng H, Mao B, Wang W, Ye C, Li S, Wang J, Wong GK, Yang H, Biology Analysis Group (2004) A draft sequence for the genome of the domesticated silkworm (Bombyx mori). Science 306:1937–1940
Xiong Y, Burke WD, Eickbush TH (1993) Pao, a highly divergent retrotransposable element from Bombyx mori containing long terminal repeats with tandem copies of the putative R region. Nucleic Acids Res 21:2117–2123
Yamamoto K, Narukawa J, Kadono-Okuda K, Nohata J, Sasanuma M, Suetsugu Y, Banno Y, Fujii H, Goldsmith MR, Mita K (2006) Construction of a single nucleotide polymorphism linkage map for the silkworm, Bombyx mori, based on bacterial artificial chromosome end sequences. Genetics 173:151–161
Yasukochi Y (1998) A dense genetic map of the silkworm, Bombyx mori, covering all chromosomes based on 1,018 molecular markers. Genetics 150:1513–1525
Yasukochi Y (2002) PCR-based screening for bacterial artificial chromosome libraries. Methods Mol Biol 192:401–410
Acknowledgements
This work was supported by grants from BRAIN (to K. M.), Grants-in-Aid for Scientific Research, MEXT (Nos. 17052006, 17580044, 16011263 and 17018007) and JSPS (No. 16208006), the Insect Technology Project, MAFF/NIAS (No. 1216), and the National Bioresource Project, MEXT.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abe, H., Fujii, T., Tanaka, N. et al. Identification of the female-determining region of the W chromosome in Bombyx mori . Genetica 133, 269–282 (2008). https://doi.org/10.1007/s10709-007-9210-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-007-9210-1