Abstract
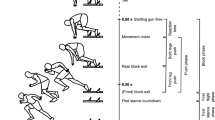
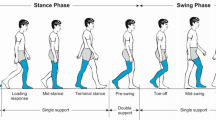
Objective. The evaluation of variability of biological rhythmic activities through measures such as Approximate Entropy (ApEn) has provided important information regarding pathology in disciplines such as cardiology and neurology. This research lead to the “loss of complexity hypothesis” where decreased variability is associated with loss of healthy flexibility rendering the system more rigid and unable to adapt to stresses. ApEn as a measure of variability and complexity, correlates well with pathology while, in some cases, it is predictive of subsequent clinical changes. The study of human gait could benefit from the application of ApEn since it is also a rhythmical oscillation. Our aim was to assess the variability of the ACL deficient knee, since ACL rupture is a common musculoskeletal injury and is accompanied by altered gait patterns and future pathology in the joint. We hypothesized that the ACL deficient knee will exhibit more regular and less variable walking patterns than the contralateral intact knee. Methods. Ten subjects with unilateral deficiency walked on a treadmill at their self-selected speed, 20% faster, and 20% slower, while kinematics were collected (50 Hz) from 80 consecutive strides for each condition. The ApEn of the resulted knee joint flexion-extension time series was calculated. Results. Significantly smaller ApEn values were found in the ACL deficient knee when compared with the contralateral intact (F = 5.57, p = 0.022), for all speeds. ApEn values significantly increased (F = 5.79, p = 0.005) with increases in walking speed. Conclusions. The altered properties of the ACL deficient knee, which exhibits more regular and less variable patterns than the contralateral intact knee, may decrease the adaptability of the system rendering it less able to adjust to perturbations. This could explain the increased future pathology found in the deficient knee. ApEn can be an important tool in assessing pathology and therapeutic interventions in orthopaedics.
Similar content being viewed by others
References
Goldberger AL, Rigney DR, Mietus J, Antman EM, Greewald S. Nonlinear dynamics in sudden cardiac death syndrome: Heartrate oscillations and bifurcations. Experientia 1988; 44(11–12): 983–987.
Babloyantz A, Destexhe A. Low dimensional chaos in an instance of epilepsy. Proc Natl Acad Sci USA 1986; 83: 3513–3517.
Kleiger RE, Miller JP, Bigger JT Jr, Moss AJ. Decreased heart rate variability and its association with increased mortality after acute myocardial infraction. Am J Cardiol 1987; 59(4): 256–262.
Pool R. Is it healthy to be chaotic? Science 1989; 243(4891): 604–607.
Goldberger AL, Amaral LAN, Hausdorff JM, Ivanov PC, Peng CK, Stanley HE. Fractal dynamics in physiology: Alterations with disease and aging. Proc Natl Acad Sci 2002; 99(Suppl. 1): 2466–2472.
Hausdorff JM, Peng CK, Ladin Z, Wei JY, Goldberger AL. Is walking a random walk? Evidence for long-range correlations in the stride interval of human gait. J Appl Physiol 1995; 78: 349–358.
Dingwell JB, Cusumano JP. Nonlinear time series analysis of normal and pathological human walking. Chaos 2000; 10: 848–863.
Hausdorff JM, Lertratanakul A, Cudkowitz ME, Peterson AL, Kaliton D, Goldberger AL. Dynamic markers of altered gait rhythm in amyotrophic lateral sclerosis. J Appl Physiol 2000; 88(6): 2045–2053.
Buzzi UH, Stergiou N, Kurz MJ, Hageman PA, Heidel J. Nonlinear dynamics indicates aging affects variability during gait. Clin Biomech 2003; 18: 435–443.
Stergiou N, Moraiti C, Giakas G, Ristanis S, Georgoulis AD. The effect of the walking speed on the stability of the anterior cruciate ligament deficient knee. Clin Biomech 2004; 19: 957–963.
Griffin LY, Agel J, Albohm MJ, Arendt EA, Dick RW, Garrett WE, Garrick JG, Hewett TE, Huston L, Ireland ML, Johnson RJ, Kibler WB, Lephart S, Lewis JL, Lindenfeld TN, Mandelbaum BR, Marchak P, Teitz CC, Wojtys EM. Non contact anterior cruciate ligament injuries: Risk factors and prevention strategies. J Am Acad Orthop Surg 2000; 8: 141–150.
Murrell GAC, Maddali S, Horovitz N, Oakley SP, Warren RF. The effects of time course after anterior cruciate ligament injury in correlation with meniscal and cartilage loss. Am J Sports Med 2001; 29(1): 9–14.
Lysholm M, Messner K. Sagittal plane translation of the tibia in anterior cruciate ligament deficient knees during commonly used rehabilitation exercises. Scand J med Sci Sports 1995; 5(1): 49–56.
Georgoulis AD, Papadonikolakis A, Papageorgiou CD, Mitsou A, Stergiou N. Three-Dimensional tibiofemoral kinematics of the anterior cruciate ligament-deficient and reconstructed knee during walking. Am J Sports Med 2003; 31: 75–79.
Ristanis S, Giakas G, Papageorgiou CD, Moraiti T, Stergiou N, Georgoulis AD. The effects of anterior cruciate ligament reconstruction on tibial rotation during pivoting after descending stairs. Knee Surg Sports Traumatol Arthrosc 2003 Nov; 11(6): 360–365.
Wexler G, Hurwitz DE, Bush-Joseph CA, Andriacchi TP, Bach BR. Functional gait adaptations in patients with anterior cruciate ligament deficiency over time. Clin Orthop Rel Res 1998; 348: 166–175.
Stergiou N, Buzzi UH, Kurz MJ, Heidel J. Nonlinear tools in human movement. In: Stergiou N, ed., Innovative Analyses of Human Movement. Champaign: Human Kinetics, 2004: 63–90.
Pincus SM, Goldberger AL. Physiological time series analysis: what does regularity quantify? Am J Physiol 1994; 266: H1643–H1656.
Vaillancourt DE, Newell KM. The dynamics of resting and postural tremor in Parkinson's disease. Clin Neurophysiol 2000; 111: 2046–2056.
Bhattacharya J. Complexity analysis of spontaneous EEG. Acta Neurobiol Exp (Wars). 2000; 60(4): 495–501.
Pincus SM, Viscarello RR. Approximate entropy: A regularity measure for fetal heart rate analysis. Obstet Gynecol 1992; 79(2): 249–255.
Van den Berg G, Pincus SM, Frolich M, Veldhuis JD, Roelfsema F. Reduced disorderliness of growth hormone release in biochemically inactive acromegaly after pituitary surgery. Eur J Endocrinol 1998; 138: 164–169.
Kaplan DT, Furman MI, Pincus SM, Ryan SM, Lipsitz LA, Goldberger AL. Aging and the complexity of cardiovascular dynamics. Biophys J 1991; 59: 945–949.
Ryan SM, Goldberger AL, Pincus SM, Mietus J, Lipsitz LA. Gender and age-related differences in heart rate dynamics: Are women more complex than men? J Am Coll Cardiol 1994; 24: 1700–1707.
Bruhn J, Ropcke H, Hoeft A. Approximate Entropy as an electroencephalographic measure of anesthetic drug effect during desflurane anesthesia. Anesthesiology 2000; 92(3): 715–726.
Vikman S, Makikallio TH, Yli-Mayry S, Pikkujamsa S, Koivisto AM, Reinikainen P, Airaksinen KE, Huikuri HV. Altered complexity and correlation properties of R-R interval dynamics before the spontaneous onset of paroxysmal atrial fibrillation. Circulation 1999; 100: 2079–2084.
Newell KM. Degrees of freedom and the development of center of pressure profiles. In: Newell KM and Molenaar PMC, eds, Applications of nonlinear dynamics to developmental process modeling. Hillslade: Erlbaum, 1997: 63–84.
Harbourne RT, Stergiou N. Nonlinear analysis of the development of sitting postural control. Dev Psychobiol 2003; 42(4): 368–377.
Davis R, Ounpuu S, Tyburski D, Gage J. A gait analysis data collection and reduction technique. Hum Mov Sci 1991; 10: 575–587.
Cappozzo A, Catani F, Leardini A, Benedetti MG, Croce CD. Position and orientation in space of bones during movement: Experimental artefacts. Clin Biomech 1996; 11: 90–100.
Rapp PE. A guide to dynamical analysis. Integr Physiol Behav Sci 1994; 29: 311–327.
Areblad M, Nigg BM, Ekstrand J, Olsson KO, Ekstrom H. Three-dimensional measurement of rearfoot motion during running. J Biomech 1990; 23(9): 933–940.
Matsas A, Taylor N, Mcburney H. Knee joint kinematics from familiarised treadmill walking can be generalised to overground walking in young unimpaired subjects. Gait Posture 2000; 11: 56–53.
Laurent M, Pailhous J. A note on modulation of gait in man: effect of constraining stride length and frequency. Hum Mov Sci 1986; 5: 333–343.
Murray MP, Kory RC, Clarkson BH, Sepic SB. Comparison of free and fast walking patterns of normal man. Am J Phys Med 1966; 45: 8–24.
Diedrich FJ, Warren WH Jr. Why change gaits? Dynamins of the walk-run transition. J Exp Psychol Hum Percept Perform 1995; 21(1): 183–202.
Voloshin A. The influence of walking speed on dynamic loading on the human musculoskeletal system. Med Sci Sports Exerc 2000; 32: 1156–1159.
Mees AI, Judd K, 1993. Dangers of geometric filtering. Physica D 68, 427–436.
Pincus S, Kalman RE. Not all (possibly) “random” sequences are created equal. Proc Natl Acad Sci USA 1997; 94(8): 3513–3518.
Hausdorff JM, Zemany L, Peng CK, Goldberger AL. Maturation of gait dynamics: Stride-to-stride variability and its temporal organization in children. J Appl Physiol 1999; 86: 1040–1047.
Schultz RA, Miller DC, Kerr CS, Micheli L. Mechanoreceptors in human cruciate ligaments. J Bone Jt Surg 1984; 66A: 1072–1076.
Valeriani M, Restuccia D, DiLazzaro VD, Franceschi F, Fabbriciani C, Tonali P. Central nervous system modifications in patients with lesion of the anterior cruciate ligament of the knee. Brain 1996; 119: 1751–1762.
Dingwell JB, Cusumano JP, Cavanagh PR, Sternad D. Local dynamic stability versus kinematic variability of continuous overground and treadmill walking. J Biomech Eng 2001; 123: 27–32.
Minetti AE, Ardigo LP, Capodaglio EM, Saibene F. Energetics and mechanics of human walking at oscillating speeds. Amer Zool 2001; 41: 205–210.
Weinstein RB. Terrestrial intermittent exercise: Common issues for human athletics and comperative animal locomotion. Amer Zool 2001; 41: 219–228.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Georgoulis, A.D., Moraiti, C., Ristanis, S. et al. A Novel Approach to Measure Variability in the Anterior Cruciate Ligament Deficient Knee During Walking: The Use of the Approximate Entropy in Orthopaedics. J Clin Monit Comput 20, 11–18 (2006). https://doi.org/10.1007/s10877-006-1032-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-006-1032-7