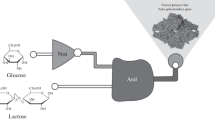
Abstract
Synthetic biology often takes cues from complex natural networks and pathways to create novel biological systems. The design modalities used in synthetic systems generally follow the different classes of gene regulation in cells such as transcriptional, post-transcriptional and post-translational regulation. The latter class is most prominent in cell signaling pathways that operate via multitude of protein–protein interactions. Engineering signaling pathways is a relatively unexplored area. This review discussed the work toward signaling pathway engineering in diverse biological contexts including bacteria, yeast, vertebrate, and plant cells. While creating synthetic signaling cascades is perhaps one of the most challenging tasks in synthetic biology, potential implications can be far-reaching and include new tools for programming cells and tissues, artificial developmental processes, and therapeutic tools.
Similar content being viewed by others
References
Acharya A et al (2002) A heterodimerizing leucine zipper coiled coil system for examining the specificity of a position interactions: amino acids I, V, L, N, A, and K. Biochemistry 41(48):14122–14131
Acuto O, Michel F (2003) CD28-mediated co-stimulation: a quantitative support for TCR signalling. Nat Rev Immunol 3(12):939–951
Akiba H et al (1999) CD28-independent costimulation of T cells by OX40 ligand and CD70 on activated B cells. J Immunol 162(12):7058–7066
Andersson J et al (2004) Differential input by Ste5 scaffold and Msg5 phosphatase route a MAPK cascade to multiple outcomes. EMBO J 23(13):2564–2576
Andrews BW, Yi TM, Iglesias PA (2006) Optimal noise filtering in the chemotactic response of Escherichia coli. PLoS Comput Biol 2(11):1407–1418
Antunes MS et al (2006) A synthetic de-greening gene circuit provides a reporting system that is remotely detectable and has a re-set capacity. Plant Biotechnol J 4(6):605–622
Antunes MS et al (2009) Engineering key components in a synthetic eukaryotic signal transduction pathway. Mol Syst Biol 5:270
Antunes MS et al (2011) Programmable ligand detection system in plants through a synthetic signal transduction pathway. PLoS ONE 6(1):e16292
Auslaender D et al (2014) A synthetic multifunctional mammalian pH sensor and CO2 transgene-control device. Mol Cell 55(3):397–408
Baim SB et al (1991) A chimeric mammalian transactivator based on the lac repressor that is regulated by temperature and isopropyl beta-d-thiogalactopyranoside. Proc Natl Acad Sci USA 88(12):5072–5076
Banghart M et al (2004) Light-activated ion channels for remote control of neuronal firing. Nat Neurosci 7(12):1381–1386
Barrett DM et al (2014) Chimeric antigen receptor therapy for cancer. Annu Rev Med 65(65):333–347
Bashor CJ et al (2008) Using engineered scaffold interactions to reshape MAP kinase pathway signaling dynamics. Science 319(5869):1539–1543
Benenson Y (2012a) Biomolecular computing systems: principles, progress and potential. Nat Rev Genet 13(7):455–468
Benenson Y (2012b) Synthetic biology with RNA: progress report. Curr Opin Chem Biol 16(3–4):278–284
Busskamp V et al (2012) Optogenetic therapy for retinitis pigmentosa. Gene Ther 19(2):169–175
Celliere G et al (2011) The plasticity of TGF-beta signaling. BMC Syst Biol 5:184
Chuong AS et al (2014) Noninvasive optical inhibition with a red-shifted microbial rhodopsin. Nat Neurosci 17(8):1123–1129
Crivici A, Ikura M (1995) Molecular and structural basis of target recognition by calmodulin. Annu Rev Biophys Biomol Struct 24:85–116
Curran KJ, Pegram HJ, Brentjens RJ (2012) Chimeric antigen receptors for T cell immunotherapy: current understanding and future directions. J Gene Med 14(6):405–415
Darrow KN et al (2015) Optogenetic stimulation of the cochlear nucleus using channelrhodopsin-2 evokes activity in the central auditory pathways. Brain Res 1599:44–56
Deuschle U, Meyer WKH, Thiesen HJ (1995) Tetracycline-reversible silencing of eukaryotic promoters. Mol Cell Biol 15(4):1907–1914
Elowitz MB, Leibler S (2000) A synthetic oscillatory network of transcriptional regulators. Nature 403(6767):335–338
Fackler OT, Grosse R (2008) Cell motility through plasma membrane blebbing. J Cell Biol 181(6):879–884
Falke JJ et al (1997) The two-component signaling pathway of bacterial chemotaxis: a molecular view of signal transduction by receptors, kinases, and adaptation enzymes. Annu Rev Cell Dev Biol 13:457–512
Ferrell JE (1996) Tripping the switch fantastic: how a protein kinase cascade can convert graded inputs into switch-like outputs. Trends Biochem Sci 21(12):460–466
Furukawa K, Hohmann S (2015) A fungicide-responsive kinase as a tool for synthetic cell fate regulation. Nucleic Acids Res 43(14):7162–7170
Fussenegger M et al (2000) Streptogramin-based gene regulation systems for mammalian cells. Nat Biotechnol 18(11):1203–1208
Gambetta GA, Lagarias JC (2001) Genetic engineering of phytochrome biosynthesis in bacteria. Proc Natl Acad Sci USA 98(19):10566–10571
Ganesh I et al (2013) Engineered fumarate sensing Escherichia coli based on novel chimeric two-component system. J Biotechnol 168(4):560–566
Gardner TS, Cantor CR, Collins JJ (2000) Construction of a genetic toggle switch in Escherichia coli. Nature 403(6767):339–342
Gasser C et al (2014) Engineering of a red-light-activated human cAMP/cGMP-specific phosphodiesterase. Proc Natl Acad Sci USA 111(24):8803–8808
Geiger TL, Jyothi MD (2001) Development and application of receptor-modified T lymphocytes for adoptive immunotherapy. Transfus Med Rev 15(1):21–34
Gilbert W, Mullerhi B (1967) Lac operator in DNA. Proc Natl Acad Sci USA 58(6):2415
Goldbeter A, Koshland DE (1981) An amplified sensitivity arising from covalent modification in biological systems. Proc Natl Acad Sci USA Biol Sci 78(11):6840–6844
Gossen M, Bujard H (1992) Tight control of gene-expression in mammalian-cells by tetracycline-responsive promoters. Proc Natl Acad Sci USA 89(12):5547–5551
Gramaglia I et al (1998) Ox-40 ligand: a potent costimulatory molecule for sustaining primary CD4 T cell responses. J Immunol 161(12):6510–6517
Gunaydin LA et al (2010) Ultrafast optogenetic control. Nat Neurosci 13(3):387–392
Hansen J et al (2014) Transplantation of prokaryotic two-component signaling pathways into mammalian cells. Proc Natl Acad Sci USA 111(44):15705–15710
Heinrich R, Neel BG, Rapoport TA (2002) Mathematical models of protein kinase signal transduction. Mol Cell 9(5):957–970
Hoch JA (2000) Two-component and phosphorelay signal transduction. Curr Opin Microbiol 3(2):165–170
Howell AS et al (2009) Singularity in polarization: rewiring yeast cells to make two buds. Cell 139(4):731–743
Hughes RM et al (2012) Light-mediated control of DNA transcription in yeast. Methods 58(4):385–391
Hwang I, Sheen J (2001) Two-component circuitry in Arabidopsis cytokinin signal transduction. Nature 413(6854):383–389
Jacob F, Monod J (1961) Genetic regulatory mechanisms in synthesis of proteins. J Mol Biol 3(3):318–356
Johnson LN (2009) The regulation of protein phosphorylation. Biochem Soc Trans 37:627–641
Kapp GT et al (2012) Control of protein signaling using a computationally designed GTPase/GEF orthogonal pair. Proc Natl Acad Sci USA 109(14):5277–5282
Kawahara M, Ueda H, Nagamune T (2010) Engineering cytokine receptors to control cellular functions. Biochem Eng J 48(3):283–294
Kiel C, Yus E, Serrano L (2010) Engineering signal transduction pathways. Cell 140(1):33–47
Kobayashi H et al (2004) Programmable cells: interfacing natural and engineered gene networks. Proc Natl Acad Sci USA 101(22):8414–8419
Krell T et al (2010) Bacterial sensor kinases: diversity in the recognition of environmental signals. In: Gottesman S, Harwood CS (eds) Annual Review of Microbiology, vol 64. Annual Reviews, Palo Alto, pp 539–559
Laub MT, Goulian M (2007) Specificity in two-componen signal transduction pathways. Annu Rev Genet 41:121–145
Le H et al (2013) Dynamic gene expression for metabolic engineering of mammalian cells in culture. Metab Eng 20:212–220
Lee MJ et al (2012) Sequential application of anticancer drugs enhances cell death by rewiring apoptotic signaling networks. Cell 149(4):780–794
Levskaya A et al (2005) Engineering Escherichia coli to see light: these smart bacteria ‘photograph’ a light pattern as a high-definition chemical image. Nature 438(7067):441–442
Li F et al (2010) Cell culture processes for monoclonal antibody production. Mabs 2(5):466–479
Lunt B et al (2010) Inference of direct residue contacts in two-component signaling. In: Simon MI, Crane BR, Crane A (eds) Methods in enzymology: two-component signaling systems, Part C, vol 471. Elsevier, New York, pp 17–41
Mezard M, Mora T (2009) Constraint satisfaction problems and neural networks: a statistical physics perspective. J Physiol Paris 103(1–2):107–113
Miller MB, Bassler BL (2001) Quorum sensing in bacteria. Annu Rev Microbiol 55:165–199
Mills E, Kevin T (2011) Ca2+-mediated synthetic biosystems offer protein design versatility, signal specificity, and pathway rewiring. Chem Biol 18(12):1611–1619
Mueller K et al (2014) Orthogonal optogenetic triple-gene control in mammalian cells. ACS Synth Biol 3(11):796–801
Nagel G et al (2003) Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci USA 100(24):13940–13945
Novak B, Tyson JJ (2008) Design principles of biochemical oscillators. Nat Rev Mol Cell Biol 9(12):981–991
O’Shaughnessy EC et al (2011) Tunable signal processing in synthetic MAP kinase cascades. Cell 144(1):119–131
Park SH, Zarrinpar A, Lim WA (2003) Rewiring MAP kinase pathways using alternative scaffold assembly mechanisms. Science 299(5609):1061–1064
Pawson T, Warner N (2007) Oncogenic re-wiring of cellular signaling pathways. Oncogene 26(9):1268–1275
Procaccini A et al (2011) Dissecting the specificity of protein–protein interaction in bacterial two-component signaling: orphans and crosstalks. PLoS ONE 6(5):e19729
Ptashne M (1967) Specific binding of lambda phage repressor to lambda DNA. Nature 214(5085):232
Pule MA et al (2005) A chimeric T cell antigen receptor that augments cytokine release and supports clonal expansion of primary human T cells. Mol Ther 12(5):933–941
Puttick J, Baker EN, Delbaere LTJ (2008) Histidine phosphorylation in biological systems. Biochim Biophys Acta Proteins Proteom 1784(1):100–105
Qin L et al (2000) A monomeric histidine kinase derived from EnvZ, an Escherichia coli osmosensor. Mol Microbiol 36(1):24–32
Regot S et al (2011) Distributed biological computation with multicellular engineered networks. Nature 469(7329):207–211
Rinaudo K et al (2007) A universal RNAi-based logic evaluator that operates in mammalian cells. Nat Biotechnol 25(7):795–801
Rowley JD (1990) The philadelphia-chromosome translocation: a paradigm for understanding leukemia. Cancer 65(10):2178–2184
Sadelain M, Riviere I, Brentjens R (2003) Targeting tumours with genetically enhanced T lymphocytes. Nat Rev Cancer 3(1):35–45
Shimizu TS, Tu YH, Berg HC (2010) A modular gradient-sensing network for chemotaxis in Escherichia coli revealed by responses to time-varying stimuli. Mol Syst Biol 6(1):382
Skerker JM et al (2008) Rewiring the specificity of two-component signal transduction systems. Cell 133(6):1043–1054
Tabor JJ et al (2009) A synthetic genetic edge detection program. Cell 137(7):1272–1281
Tay S et al (2010) Single-cell NF-kappa B dynamics reveal digital activation and analogue information processing. Nature 466(7303):267–271
Thattai M, van Oudenaarden A (2002) Attenuation of noise in ultrasensitive signaling cascades. Biophys J 82(6):2943–2950
Tyson JJ, Chen KC, Novak B (2003) Sniffers, buzzers, toggles and blinkers: dynamics of regulatory and signaling pathways in the cell. Curr Opin Cell Biol 15(2):221–231
Tyszkiewicz AB, Muir TW (2008) Activation of protein splicing with light in yeast. Nat Methods 5(4):303–305
Wang R, Brattain MG (2007) The maximal size of protein to diffuse through the nuclear pore is larger than 60 kDa. FEBS Lett 581(17):3164–3170
Wang X et al (2014) Optogenetic stimulation of locus ceruleus neurons augments inhibitory transmission to parasympathetic cardiac vagal neurons via activation of brainstem alpha 1 and beta 1 receptors. J Neurosci 34(18):6182–6189
Weber W et al (2002) Macrolide-based transgene control in mammalian cells and mice. Nat Biotechnol 20(9):901–907
Weigt M et al (2009) Identification of direct residue contacts in protein–protein interaction by message passing. Proc Natl Acad Sci USA 106(1):67–72
Wilkinson MG, Millar JBA (2000) Control of the eukaryotic cell cycle by MAP kinase signaling pathways. Faseb J 14(14):2147–2157
Wu CL et al (2003) Phosphorylation of the MAPKKK regulator Ste50p in Saccharomyces cerevisiae: a casein kinase I phosphorylation site is required for proper mating function. Eukaryot Cell 2(5):949–961
Yamamoto K et al (2005) Functional characterization in vitro of all two-component signal transduction systems from Escherichia coli. J Biol Chem 280(2):1448–1456
Yang X et al (2013) A light-inducible organelle-targeting system for dynamically activating and inactivating signaling in budding yeast. Mol Biol Cell 24(15):2419–2430
Ye HF et al (2011) A synthetic optogenetic transcription device enhances blood-glucose homeostasis in mice. Science 332(6037):1565–1568
Yeh BJ et al (2007) Rewiring cellular morphology pathways with synthetic guanine nucleotide exchange factors. Nature 447(7144):596–600
Zhang F et al (2006) Channelrhodopsin-2 and optical control of excitable cells. Nat Methods 3(10):785–792
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hansen, J., Benenson, Y. Synthetic biology of cell signaling. Nat Comput 15, 5–13 (2016). https://doi.org/10.1007/s11047-015-9526-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11047-015-9526-1