Abstract
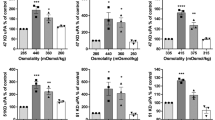
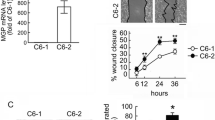
Glioma invasiveness is accomplished in part by matrix metalloproteinases (MMPs) which remodel the constraints of the three dimensional (3D) matrix of the brain parenchyma. Tissue culture studies have advanced knowledge of glioma invasiveness but the majority of studies have used a two dimensional (2D) monolayer culture system which does not reproduce the spatial constraints of invasiveness in vivo. Here, we have used a 3D matrix of type I collagen (CL) gel to address glioma invasiveness in vitro. We show that in 3D CL matrix, interleukin-1 beta (IL-1β) and tumor necrosis factor-alpha (TNF-α), cytokines which are elevated in gliomas in vivo, increased glioma cell invasiveness with correspondent elevation of MMP-2 and MMP-9. Cytokine-stimulated glioma invasiveness was blocked by three pharmacological metalloproteinase inhibitors and by small interfering RNAs to MMP-2. Thus, in 3D matrix of CL, MMP-2 expression is modulated by inflammatory cytokines with the concomitant increase in glioma invasiveness.




Similar content being viewed by others
References
Le DM, Besson A, Fogg DK et al (2003) Exploitation of astrocytes by glioma cells to facilitate invasiveness: a mechanism involving matrix metalloproteinase-2 and the urokinase-type plasminogen activator-plasmin cascade. J Neurosci 23:4034–4043
Goldbrunner RH, Bernstein JJ, Tonn JC (1998) ECM-mediated glioma cell invasion. Microsc Res Tech 43:250–257. doi :10.1002/(SICI)1097-0029(19981101)43:3<250::AID-JEMT7>3.0.CO;2-C
Forsyth PA, Wong H, Laing TD et al (1999) Gelatinase-A (MMP-2), gelatinase-B (MMP-9) and membrane type matrix metalloproteinase-1 (MT1-MMP) are involved in different aspects of the pathophysiology of malignant gliomas. Br J Cancer 79:1828–1835. doi:10.1038/sj.bjc.6690291
Yong VW, Power C, Forsyth P et al (2001) Metalloproteinases in biology and pathology of the nervous system. Nat Rev Neurosci 2:502–511. doi:10.1038/35081571
VanMeter TE, Rooprai HK, Kibble MM et al (2001) The role of matrix metalloproteinase genes in glioma invasion: co-dependent and interactive proteolysis. J Neurooncol 53:213–235. doi:10.1023/A:1012280925031
Levicar N, Nuttall RK, Lah TT (2003) Proteases in brain tumour progression. Acta Neurochir (Wien) 145:825–838. doi:10.1007/s00701-003-0097-z
Rao JS (2003) Molecular mechanisms of glioma invasiveness: the role of proteases. Nat Rev Cancer 3:489–501. doi:10.1038/nrc1121
Yong VW (2005) Metalloproteinases: mediators of pathology and regeneration in the CNS. Nat Rev Neurosci 6:931–944. doi:10.1038/nrn1807
Blazquez C, Salazar M, Carracedo A (2008) Cannabinoids inhibit glioma invasion by down-regulating matrix metalloproteinase-2 expression. Cancer Res 68(6):1945–1952. doi:10.1158/0008-5472.CAN-07-5176
Kim SY, Lee EJ, Woo MS (2008) Inhibition of matrix metalloproteinase-9 gene expression by an isoflavone metabolite, irisolidone in U87MG human astroglioma cells. Biochem Biophys Res Commun 366(2):493–499. doi:10.1016/j.bbrc.2007.11.178
Fillmore HL, VanMeter TE, Broaddus WC (2001) Membrane type matrix metalloproteinases (MT-MMPs): expression and function during glioma invasion. J Neurooncol 53:187–202. doi:10.1023/A:1012213604731
Nuttall RK, Pennington CJ, Taplin J et al (2003) Elevated membrane-type matrix metalloproteinases in gliomas revealed by profiling proteases and inhibitors in human cancer cells. Mol Cancer Res 1:333–345
Bellail AC, Hunter SB, Brat DJ (2004) Microregional extracellular matrix haterogeniety in brain modulates glioma invasion. Int J Biochem Cell Biol 36(6):1046–1069. doi:10.1016/j.biocel.2004.01.013
Sarkar S, Nuttall RK, Liu S et al (2006) Tenascin-C stimulates glioma cell invasion through matrix metalloproteinase-12. Cancer Res 66:11771–11780. doi:10.1158/0008-5472.CAN-05-0470
Wang F, Weaver VM, Petersen OW et al (1998) Reciprocal interactions between h1-integrin and epidermal growth factor receptor in three-dimensional basement membrane breast cultures: a different perspective in epithelial biology. Proc Natl Acad Sci USA 95:14821–14826. doi:10.1073/pnas.95.25.14821
Cukierman E, Pankov R, Stevens DR et al (2001) Taking cell-matrix adhesions to the third dimension. Science 294:1708–1712. doi:10.1126/science.1064829
Hotary KB, Allen ED, Brooks PC et al (2003) Membrane type I matrix metalloproteinase usurps tumor growth control imposed by the three dimensional extracellular matrix. Cell 114:33–45. doi:10.1016/S0092-8674(03)00513-0
Maestro RD, Shivers R, McDonald W et al (2001) Dynamics of C6 astrocytoma invasion into three dimensional collagen gels. J Neurooncol 53:87–98. doi:10.1023/A:1012236830230
Bellon G, Caulet T, Cam Y et al (1985) Immunohistochemical localisation of macromolecules of the basement membrane and extracellular matrix of human gliomas and meningiomas. Acta Neuropathol 66:245–252. doi:10.1007/BF00688590
Paulus W, Roggendorf W, Schuppan D (1988) Immunohistochemical investigation of collagen subtypes in human glioblastomas. Virchows Arch A Pathol Anat Histol 413:325–332
Akabani G, Reardon DA, Coleman RE et al (2005) Dosimetry and radiographic analysis of 131I-labeled anti-tenascin 81C6 murine monoclonal antibody in newly diagnosed patients with malignant gliomas: a phase II study. J Nucl Med 46:1042–1051
Zamecnik J, Vargova L, Homola A (2004) Extracellular matrix glycoproteins and diffusion barriers in human astrocytic tumours. Neuropathol Appl Neurobiol 30:338–350. doi:10.1046/j.0305-1846.2003.00541.x
Annabi B, Thibeault S, Moumdjian R (2004) Hyaluronan cell surface binding is induced by type I collagen and regulated by caveolae in glioma cells. J Biol Chem 279:21888–21896. doi:10.1074/jbc.M313694200
Graeber MB, Scheithauer BW, Kreutzberg GW (2002) Microglia in brain tumors. Glia 40:252–259
Uhm JH, Dooley NP, Yong VW et al (1996) Glioma invasion in vitro: regulation by matrix metalloprotease-2 and protein kinase C. Clin Exp Metastasis 14:421–433. doi:10.1007/BF00128958
Tamaki M, Mc Donald W, Amberger VR (1997) Implantation of C6 astrocytoma spheroid into collagen type1 gels: invasive, proliferative and enzymatic characterizations. J Neurosurg 87:602–609
Tara HL, Davis SJ, Madri JA (1998) Three-dimensional type I collagen lattices induce coordinate expression of matrix metalloproteinases MT1- MMP and MMP-2 in microvascular endothelial cells. J Biol Chem 273:3604–3610. doi:10.1074/jbc.273.6.3604
Morioka T, Baba T, Black KL, Streit WJ (1992) Inflammatory cell infiltrates vary in experimental primary and metastatic brain tumors. Neurosurgery 30:891–896
Stevens A, Kloter I, Roggendorf W (1988) Inflammatory infiltrates and natural killer cell presence in human brain tumors. Cancer 61:738–743. doi :10.1002/1097-0142(19880215)61:4<738::AID-CNCR2820610417>3.0.CO;2-E
Laeurum OD, Bjerkvig R, Steinsvag SK et al (1984) Invasiveness of primary tumors. Cancer Metastasis Rev 3:223–236. doi:10.1007/BF00048386
Lung HL, Shan SW, Tsang D et al (2005) Tumor necrosis factor-alpha mediates the proliferation of rat C6 glioma cells via beta adrenergic receptors. J Neuroimmunol 166:102–112. doi:10.1016/j.jneuroim.2005.05.011
Ueda Y, Nakagawa T, Kubota T et al (2005) Glioma cells under hypoxic conditions block the brain microvascular endothelial cell death induced by serum starvation. J Neurochem 95:99–110. doi:10.1111/j.1471-4159.2005.03343.x
Zhou Y, Larsen PH, Yong VW (2002) CXCR4 is a major chemokine receptor on glioma cells and mediates their survival. J Biol Chem 277:49481–49487. doi:10.1074/jbc.M206222200
Zhang J, Sarkar S, Yong VW (2005) The chemokine stromal cell derived factor-1 (CXCL12) promotes glioma invasiveness through MT2-matrix metalloproteinase. Carcinogenesis 26(12):2069–2077. doi:10.1093/carcin/bgi183
Acknowledgments
This study was supported by an operating grant from the Canadian Institutes of Health Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarkar, S., Yong, V.W. Inflammatory cytokine modulation of matrix metalloproteinase expression and invasiveness of glioma cells in a 3-dimensional collagen matrix. J Neurooncol 91, 157–164 (2009). https://doi.org/10.1007/s11060-008-9695-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-008-9695-1