Abstract
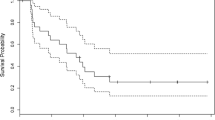
To estimate the sustained (≥8 weeks) objective response rate in pediatric patients with recurrent or progressive high-grade gliomas (HGG, Stratum A) or brainstem gliomas (BSG, Stratum B) treated with the combination of O6-benzylguanine (O6BG) and temozolomide® (TMZ). Patients received O6BG 120 mg/m2/d IV followed by TMZ 75 mg/m2/d orally daily for 5 consecutive days of each 28-day course. The target objective response rate to consider the combination active was 17%. A two-stage design was employed. Forty-three patients were enrolled; 41 were evaluable for response, including 25 patients with HGG and 16 patients with BSG. The combination of O6BG and TMZ was tolerable, and the primary toxicities were myelosuppression and gastrointestinal symptoms. One sustained (≥8 weeks) partial response was observed in the HGG cohort; no sustained objective responses were observed in the BSG cohort. Long-term (≥6 courses) stable disease (SD) was observed in 4 patients in Stratum A and 1 patient in Stratum B. Of the 5 patients with objective response or long-term SD, 3 underwent central review with 2 reclassified as low-grade gliomas. The combination of O6BG and TMZ did not achieve the target response rate for activity in pediatric patients with recurrent or progressive HGG and BSG.

Similar content being viewed by others
References
Friedman H, Dolan M, Pegg A et al (1995) Activity of temozolomide in the treatment of central nervous system xenografts. Cancer Res 55:2853–2857
Stupp R, Hegi M, Gilbert M, Chakravarti A (2007) Chemoradiotherapy in malignant glioma: Standard of care and future directions. J Clin Oncol 25:4127–4136
Yung A, Prados M, Yaya-Tur R et al (1999) Multicenter phase II trial of temozolomide in patients with anaplastic astrocytoma or anaplastic oligoastrocytoma at first relapse. J Clin Oncol 17:2762–2771
Brada M, Hoang-Xuan K, Rampling R et al (2006) Multicenter phase II trial of temozolomide in patients with glioblastoma multiforme at first relapse. Ann Oncol 12:259–266
Sankar A, Thomas D, Darling J (1999) Sensitivity of short-term cultures derived from human malignant glioma to the anti-cancer drug temozolomide. Anticancer Drugs 10:179–185
Stupp R, Dietrich P, Ostermann K et al (2002) Promising survival for patients with newly diagnosed glioblastoma multiforme treated with concomitant radiation plus temozolomide followed by adjuvant temozolomide. J Clin Oncol 20:1375–1382
Chibbaro S, Benvenuti L, Caprio A et al (2004) Temozolomide as first-line agent in treating high-grade gliomas: phase II study. J Neurooncol 67:77–81
Lashford L, Thiesse P, Jouvet A et al (2002) Temozolomide in malignant gliomas of childhood: a United Kingdom Children’s Cancer Study Group and French Society for Pediatric Oncology Intergroup Study. J Clin Oncol 20:4684–4691
Broniscer A, Iacono L, Chintagumpala M et al (2005) Role of temozolomide after radiotherapy for newly diagnosed diffuse brainstem glioma in children: results of a multiinstitutional study (SJHG-98). Cancer 103:133–139
Nicholson H, Kretschmar C, Krailo M et al (2007) Phase 2 study of temozolomide in children and adolescents with recurrent central nervous system tumors: a report from the Children’s Oncology Group. Cancer 110:1542–1550
Cohen K, Pollack I, Zhou T et al (2011) Temozolomide in the treatment of high-grade gliomas in children: a report from the Children’s Oncology Group. Neuro Oncology 13:317–323
Cohen K, Heideman R, Zhou T et al (2011) Temozolomide in the treatment of children with newly diagnosed diffuse intrinsic pontine gliomas: a report from the Children’s Oncology Group. Neuro Oncology 13:410–416
Bobola M, Silber J, Ellenbogen R et al (2005) O6-methylguanine-DNA-methyltransferase, O6-benzylguanine, and resistance to alkylators in pediatric primary brain tumor cell lines. Clin Cancer Res 11:2747–2755
Donson A, Addo-Yobo S, Handler M et al (2007) MGMT promoter methylation correlates with survival benefit and sensitivity to temozolomide in pediatric glioblastomas. Pediatr Blood Cancer 48:403–407
Denny B, Wheelhouse R, Stevens M et al (1994) NMR and molecular modeling investigation of the mechanism of activation of the antitumor drug temozolomide and its interaction with DNA. Biochemistry 33:9045–9051
Stevens M, Hickman J, Langdon S et al (1987) Antitumor activity and pharmacokinetics in mice of 8-carbamoyl-3-methyl-imidazo[5, 1-d]-1, 2, 3, 5-terazin-4(3H)-one (CCRG 81045; M & B 39831), a novel drug with potential as an alternative to dacarbazine. Cancer Res 47:5846–5852
Hegi M, Liu L, Herman J et al (2008) Correlation of O6-methylguanine methyltransferase (MGMT) promoter methylation with clinical outcomes in glioblastoma and clinical strategies to modulate MGMT activity. J Clin Oncol 26:4189–4199
Roos W, Batista L, Naumann S et al (2007) Apoptosis in malignant glioma cells triggered by temozolomide-induced DNA lesion O6-methylguanine. Oncogene 26:186–197
Friedman H, McLendon R, Kerby T et al (1998) DNA mismatch repair and O6-alkylguanine-DNA alkyltransferase analysis and response to Temodal in newly diagnosed malignant glioma. J Clin Oncol 16:3851–3857
Kokkinakis D, Bocangel D, Schold S et al (2001) Thresholds of O6-alkylguanine-DNA alkyltransferase which confer significant resistance of human glial tumor xenografts to treatment with 1, 3-bis(2-chloroethyl)-1-nitrosourea or temozolomide. Clin Cancer Res 7:421–428
Gerson S (2002) Clinical relevance of MGMT in the treatment of cancer. J Clin Oncol 20:2388–2399
Ishii D, Natsume A, Wakabayashi T et al (2007) Efficacy of temozolomide is correlated with 1p loss and methylation of the deoxyribonucleic acid repair gene MGMT in malignant gliomas. Neurol Med Chir (Tokyo) 47:341–349
Friedman H, Keir S, Pegg A et al (2002) O6-benzylguanine-mediated enhancement of chemotherapy. Mol Cancer Ther 1:943–948
Wedge S, Newlands E (1996) O6-benzylguanine enhances the sensitivity of a glioma xenograft with low O6-alkylguanine-DNA alkyltransferase activity to temozolomide and BCNU. Br J Cancer 73:1049–1052
Warren K, Aikin A, Libucha M et al (2005) Phase I study of O6-benzylguanine and temozolomide administered daily for 5 days to pediatric patients with solid tumors. J Clin Oncol 23:7646–7653
Broniscer A, Gururangan S, MacDonald T et al (2007) Phase I trial of single-dose temozolomide and continuous administration of O6-benzylguanine in children with brain tumors: a pediatric brain tumor consortium report. Clin Cancer Res 13:6712–6718
Maxwell J, Johnson S, Quinn J et al (2006) Quantitative analysis of O6-alkylguanine-DNA-alkyltransferase in malignant glioma. Mol Cancer Ther 5:2531–2539
Rich J, Sathornsumetee S, Keir S et al (2005) ZD6474, a novel tyrosine kinase inhibitor of vascular endothelial growth factor receptor and epidermal growth factor receptor, inhibits tumor growth of multiple nervous system tumors. Clin Cancer Res 11:8145–8157
Brandes A, Ermani M, Basso U et al (2002) Temozolomide in patients with glioblastoma at second relapse after first line nitrosourea-procarbazine failure: a phase II study. Oncology 63:38–41
Quinn J, Jiang S, Reardon D et al (2009) Phase II trial of temozolomide plus O6-benzylguanine in adults with recurrent, temozolomide-resistant malignant glioma. J Clin Oncol 27:1262–1267
Quinn J, Pluda J, Dolan M et al (2002) Phase II trial of carmustine plus O6-benzylguanine for patients with nitrosourea-resistant recurrent or progressive malignant glioma. J Clin Oncol 20:2277–2283
Brandes A, Tosoni A, Cavallo G et al (2006) Correlations between O6-methylguanine DNA methyltransferase promoter methylation status, 1p and 19q deletions, and response to temozolomide in anaplastic and recurrent oligodendroglioma: a prospective GICNO study. J Clin Oncol 24:4746–4753
Donson A, Addo-Yobo S, Handler M et al (2007) MGMT promoter methylation correlates with survival benefit and sensitivity to temozolomide in pediatric glioblastoma. Pediatr Blood Cancer 48:403–407
Newlands E (1992) Phase I trial of temozolomide (CCRG 81045: M&B 39831: NSC 362856). Br J Cancer 65:287–291
Patel M, McCully C, Godwin K et al (2003) Plasma and cerebrospinal fluid pharmacokinetics of intravenous temozolomide in non-human primates. J Neurooncol 61:203–207
Panetta J, Kirstein M, Gajjar A et al (2003) Population pharmacokinetics of temozolomide and metabolites in infants and children with primary central nervous system tumors. Cancer Chemother Pharmacol 52:435–441
Berg S, Gerson S, Godwin K et al (1995) Plasma and cerebrospinal fluid pharmacokinetics of O6-benzylguanine and time course of peripheral blood mononuclear cell O6-methylguanine-DNA methyltransferase inhibition in the nonhuman primate. Cancer Res 55:4604–4610
Friedman H, Kokkinakis D, Pluda J et al (1998) Phase I trial of O6-benzylguanine for patients undergoing surgery for malignant glioma. J Clin Oncol 16:3570–3575
Karran P, Bignami M (1994) DNA damage tolerance, mismatch repair and genome instability. Bioessays 16:833–839
Acknowledgments
This work was supported in part by NIH grant U01 CA81457 for the Pediatric Brain Tumor Consortium, NIH grant 5M01RR000188, and the American Lebanese Syrian Associated Charities, and the intramural program of the NIH. The authors and the PBTC acknowledge the protocol management of Mr. Christopher Smith, the central pathology review participation of Dr. Adesina Adekunle and Dr. Veena Rajaram and the statistical support of Mr. Shawn Lesh.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Warren, K.E., Gururangan, S., Geyer, J.R. et al. A phase II study of O6-benzylguanine and temozolomide in pediatric patients with recurrent or progressive high-grade gliomas and brainstem gliomas: a Pediatric Brain Tumor Consortium study. J Neurooncol 106, 643–649 (2012). https://doi.org/10.1007/s11060-011-0709-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-011-0709-z