Abstract
Purpose
To compare drug deposition in the nose and olfactory region with different nasal devices and administration techniques. A Sar-Gel based colorimetry method will be developed to quantify local deposition rates.
Methods
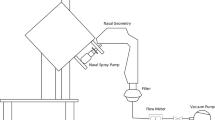
A sectional nasal airway cast was developed based on an MRI-based nasal airway model to visualize deposition patterns and measure regional dosages. Four nasal spray pumps and four nebulizers were tested with both standard and point-release administration techniques. Delivered dosages were measured using a high-precision scale. The colorimetry correlation for deposited mass was developed via image processing in Matlab and its performance was evaluated through comparison to experimental measurements.
Results
Results show that the majority of nasal spray droplets deposited in the anterior nose while only a small fraction (less than 4.6%) reached the olfactory region. For all nebulizers considered, more droplets went beyond the nasal valve, leading to distinct deposition patterns as a function of both the nebulizer type (droplet size and initial speed) and inhalation flow rate. With the point-release administration, up to 9.0% (±1.9%) of administered drugs were delivered to the olfactory region and 15.7 (±2.4%) to the upper nose using Pari Sinus.
Conclusions
Standard nasal devices are inadequate to deliver clinically significant olfactory dosages without excess drug losses in other nasal epitheliums. The Sar-Gel based colorimetry method appears to provide a simple and practical approach to visualize and quantify regional deposition.











Similar content being viewed by others
References
Born J, Lange T, Kern W, McGregor GP, Bickel U, Fehm HL. Sniffing neuropeptides: a transnasal approach to the human brain. Nat Neurosci. 2002;5(6):514–6.
Valentine R, Athanasiadis T, Thwin M, Singhal D, Weitzel EK, Wormald PJ. A prospective controlled trial of pulsed nasal nebulizer in maximally dissected cadavers. Am J Rhinol. 2008;22(4):390–4.
Moller W, Schuschnig U, Celik G, Munzing W, Bartenstein P, Haussinger K, et al. Topical drug delivery in chronic rhinosinusitis patients before and after sinus surgery using pulsating aerosols. PLoS ONE. 2013;8(9):e74991.
Djupesland PG, Skretting A, Winderen M, Holand T. Breath actuated device improves delivery to target sites beyond the nasal valve. Laryngoscope. 2006;116(3):466–72.
Washington N, McGlashan JA, Jackson SJ, Bush D, Pitt KG, Rawlins DA, et al. The effect of nasal patency on the clearance of radiolabeled saline in healthy volunteers. Pharm Res. 2000;17(6):733–6.
Suman JD, Laube BL, Lin TC, Brouet G, Dalby R. Validity of in vitro tests on aqueous spray pumps as surrogates for nasal deposition. Pharm Res. 2002;19(1):1–6.
Guo Y, Laube B, Dalby R. The effect of formulation variables and breathing patterns on the site of nasal deposition in an anatomically correct model. Pharm Res. 2005;22(11):1871–8.
Laube BL, Sharpless G, Shermer C, Nasir O, Sullivan V, Powell K. Deposition of albuterol aerosol generated by pneumatic nebulizer in the Sophia Anatomical Infant Nose-Throat (SAINT) model. Pharm Res. 2010;27(8):1722–9.
Fleming JS, Conway JH, Bolt L, Holgate ST. A comparison of planar scintigraphy and SPECT measurement of total lung deposition of inhaled aerosol. J Aerosol Med. 2003;16(1):9–19.
Bondesson E, Bengtsson T, Borgstrom L, Nilsson LE, Norrgren K, Trofast E, et al. Planar gamma scintigraphy--points to consider when quantifying pulmonary dry powder aerosol deposition. Int J Pharm. 2003;258(1–2):227–40.
Kundoor V, Dalby RN. Assessment of nasal spray deposition pattern in a silicone human nose model using a color-based method. Pharm Res. 2010;27(1):30–6.
Kundoor V, Dalby RN. Effect of formulation- and administration-related variables on deposition pattern of nasal spray pumps evaluated using a nasal cast. Pharm Res. 2011;28(8):1895–904.
Cheng YS, Holmes TD, Gao J, Guilmette RA, Li S, Surakitbanharn Y, et al. Characterization of nasal spray pumps and deposition pattern in a replica of the human nasal airway. J Aerosol Med Pulm D. 2001;14(2):267–80.
Wang J, Bentz J, Anderson R. Nasal device for delivery to the olfactory region. Patent: US 20070119451 A1; 2007.
Gizurarson S. A method for administration of active substances to the olfactory region. Patent: CA 2298596 A1; 1999.
Hoekman JD, Ho RJY. Effects of localized hydrophilic mannitol and hydrophobic nelfinavir administration targeted to olfactory epithelium on brain distribution. AAPS PharmSciTech. 2011;12(2):534–43.
Hoekman JD, Ho RJY. Enhanced analgesic responses after preferential delivery of morphine and fentanyl to the olfactory epithelium in rats. Anesth Analg. 2011;113(3):641–51.
Corley RA, Kabilan S, Kuprat AP, Carson JP, Minard KR, Jacob RE, et al. Comparative computational modeling of airflows and vapor dosimetry in the respiratory tracts of rat, monkey, and human. Toxicol Sci. 2012;128(2):500–16.
Xi J, Kim J, Si XA, Corley RA, Zhou Y. Modeling of inertial depositions in scaled models of rat and human nasal airways: towards in vitro regional dosimetry in small animals. J Aerosol Sci. 2016; Accepted.
Si X, Xi J, Kim J, Zhou Y, Zhong H. Modeling of release position and ventilation effects on olfactory aerosol drug delivery. Respir Physiol Neurobiol. 2013;186(1):22–32.
Xi J, Longest PW. Characterization of submicrometer aerosol deposition in extrathoracic airways during nasal exhalation. Aerosol Sci Technol. 2009;43(8):808–27.
Guilmette RA, Wicks JD, Wolff RK. Morphometry of human nasal airways in vivo using Magnetic Resonance Imaging. J Aerosol Med. 1989;2(4):365–77.
Cheng KH, Cheng YS, Yeh HC, Swift DL. Deposition of ultrafine aerosols in the head airways during natural breathing and during simulated breath-holding using replicate human upper airway casts. Aerosol Sci Technol. 1995;23(3):465–74.
Kelly JT, Asgharian B, Kimbell JS, Wong B. Particle depositon in human nasal airway replicas manufactured by different methds. Part I: Inertial regime particles. Aerosol Sci Technol. 2004;38:1063–71.
Schroeter JD, Kimbell JS, Asgharian B. Analysis of particle deposition in the turbinate and olfacotry regions using a human nasal computational fluid dynamics model. J Aerosol Med. 2006;19(3):301–13.
Xi J, Berlinski A, Zhou Y, Greenberg B, Ou X. Breathing resistance and ultrafine particle deposition in nasal-laryngeal airways of a newborn, an infant, a child, and an adult. Ann Biomed Eng. 2012;40(12):2579–95.
Kimbell JS, Subramaniam RP. Use of computational fluid dynamics models for dosimetry of inhaled gases in the nasal passages. Inhal Toxicol. 2001;13(5):325–34.
Subramaniam RP, Richardson RB, Morgan KT, Kimbell JS, Guilmette RA. Computational fluid dynamics simulations of inspiratory airflow in the human nose and nasopharynx. Inhal Toxicol. 1998;10(2):91–120.
Xi J, Si X, Kim JW, Berlinski A. Simulation of airflow and aerosol deposition in the nasal cavity of a 5-year-old child. J Aerosol Sci. 2011;42(3):156–73.
Xi J, Si X, Gaide R. Electrophoretic particle guidance significantly enhances olfactory drug delivery: a feasibility study. PLoS One. 2014;9(1):e86593.
Gizurarson S. Anatomical and histological factors affecting intranasal drug and vaccine delivery. Curr Drug Deliv. 2012;9(6):566–82.
Landis MS, Boyden T, Pegg S. Nasal-to-CNS drug delivery: where are we now and where are we heading? An industrial perspective. Ther Deliv. 2012;3(2):195–208.
Schroeter JD, Tewksbury EW, Wong BA, Kimbell JS. Experimental measurements and computational predictions of regional particle deposition in a sectional nasal model. J Aerosol Med Pulm Drug Deliv. 2015;28(1):20–9.
Djupesland PG, Messina JC, Mahmoud RA. The nasal approach to delivering treatment for brain diseases: an anatomic, physiologic, and delivery technology overview. Ther Deliv. 2014;5(6):709–33.
Leopold DA, Hummel T, Schwob JE, Hong SC, Knecht M, Kobal G. Anterior distribution of human olfactory epithelium. Laryngoscope. 2000;110(3):417–21.
Shi H, Kleinstreuer C, Zhang Z. Laminar airflow and nanoparticle or vapor deposition in a human nasal cavity model. J Biomech Eng. 2006;128:697–706.
Sengoku R, Matsushima S, Bono K, Sakuta K, Yamazaki M, Miyagawa S, et al. Olfactory function combined with morphology distinguishes Parkinson’s disease. Parkinsonism Relat Disord. 2015;21(7):771–7.
Shiga H, Taki J, Okuda K, Watanabe N, Tonami H, Furukawa M, et al. Prognostic value of olfactory nerve assessment with olfacto-scintigraphy in patients with olfactory disorders. Chem Senses. 2015;40(7):643–4.
Kikuchi A, Baba T, Hasegawa T, Sugeno N, Konno M, Aoki M, et al. Differentiating Parkinson’s disease from multiple system atrophy by I-123 meta-iodobenzylguanidine myocardial scintigraphy and olfactory test. Mov Disord. 2013;28:S156.
Prado GL, Itabashi Y, Noda H, Miura H, Mariya Y, Abe Y. Olfactory neuroblastoma visualized by Technetium-99m-ECD SPECT. Radiat Med. 2001;19(5):267–70.
Xi J, Longest PW. Numerical predictions of submicrometer aerosol deposition in the nasal cavity using a novel drift flux approach. Int J Heat Mass Transf. 2008;51(23–24):5562–77.
Si X, Xi J, Kim J. Effect of laryngopharyngeal anatomy on expiratory airflow and submicrometer particle deposition in human extrathoracic airways. Open J Fluid Dyn. 2013;3(4):286–301.
Xi J, Longest PW, Martonen TB. Effects of the laryngeal jet on nano- and microparticle transport and deposition in an approximate model of the upper tracheobronchial airways. J Appl Physiol. 2008;104(6):1761–77.
Berg EJ, Weisman JL, Oldham MJ, Robinson RJ. Flow field analysis in a compliant acinus replica model using particle image velocimetry (PIV). J Biomech. 2010;43(6):1039–47.
Inthavong K, Fung MC, Tong X, Yang W, Tu J. High resolution visualization and analysis of nasal spray drug delivery. Pharm Res. 2014;31(8):1930–7.
Inthavong K, Yang W, Fung MC, Tu JY. External and near-nozzle spray characteristics of a continuous spray atomized from a nasal spray device. Aerosol Sci Technol. 2012;46(2):165–77.
Kimbell JS, Segal RA, Asgharian B, Wong BA, Schroeter JD, Southall JP, et al. Characterization of deposition from nasal spray devices using a computational fluid dynamics model of the human nasal passages. J Aerosol Med Pulm D. 2007;20(1):59–74.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors report no conflicts of interest in this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xi, J., Yuan, J.E., Zhang, Y. et al. Visualization and Quantification of Nasal and Olfactory Deposition in a Sectional Adult Nasal Airway Cast. Pharm Res 33, 1527–1541 (2016). https://doi.org/10.1007/s11095-016-1896-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-016-1896-2