Abstract
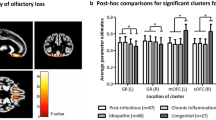
The human sense of smell is closely associated with morphological differences of the fronto-limbic system, specifically the piriform cortex and medial orbitofrontal cortex (mOFC). Still it is unclear whether cortical volume in the core olfactory areas and connected brain regions are shaped differently in individuals who suffer from lifelong olfactory deprivation relative to healthy normosmic individuals. To address this question, we examined if regional variations in gray matter volume were associated with smell ability in seventeen individuals with isolated congenital olfactory impairment (COI) matched with sixteen normosmic controls. All subjects underwent whole-brain magnetic resonance imaging, and voxel-based morphometry was used to estimate regional variations in grey matter volume. The analyses showed that relative to controls, COI subjects had significantly larger grey matter volumes in left middle frontal gyrus and right superior frontal sulcus (SFS). COI subjects with severe olfactory impairment (anosmia) had reduced grey matter volume in the left mOFC and increased volume in right piriform cortex and SFS. Within the COI group olfactory ability, measured with the “Sniffin’ Sticks” test, was positively associated with larger grey matter volume in right posterior cingulate and parahippocampal cortices whereas the opposite relationship was observed in controls. Across COI subjects and controls, better olfactory detection threshold was associated with smaller volume in right piriform cortex, while olfactory identification was negatively associated with right SFS volume. Our findings suggest that lifelong olfactory deprivation trigger changes in the cortical volume of prefrontal and limbic brain regions previously linked to olfactory memory.




Similar content being viewed by others
References
Abolmaali, N.D., Hietschold, V., Vogl, T.J., Huttenbrink, K.B., & Hummel, T. (2002). MR evaluation in patients with isolated anosmia since birth or early childhood. AJNR. American Journal of Neuroradiology, 23(1), 157–164.
Anderson, A.K., Christoff, K., Stappen, I., Panitz, D., Ghahremani, D.G., Glover, G.,.. . Sobel, N. (2003). Dissociated neural representations of intensity and valence in human olfaction. Nature Neuroscience, 6(2), 196–202. https://doi.org/10.1038/nn1001.
Andrade, K. C., Menezes, P. D., Carnaúba, A. T., Rodrigues, R. G., Leal, M. D., & Pereira, L. D. (2013). Non-flat audiograms in sensorineural hearing loss and speech perception. Clinics, 68(6), 815–819.
Arshamian, A., Iannilli, E., Gerber, J. C., Willander, J., Persson, J., Seo, H. S.,.. . Larsson, M. (2013). The functional neuroanatomy of odor evoked autobiographical memories cued by odors and words. Neuropsychologia, 51(1), 123–131. https://doi.org/10.1016/j.neuropsychologia.2012.10.023.
Ashburner, J. (2007). A fast diffeomorphic image registration algorithm. Neuroimage, 38(1), 95–113. doi:https://doi.org/10.1016/J.Neuroimage.2007.07.007.
Barbier, E. L., Marrett, S., Danek, A., Vortmeyer, A., van Gelderen, P., Duyn, J.,.. . Koretsky, A. P. (2002). Imaging cortical anatomy by high-resolution MR at 3.0T: detection of the stripe of Gennari in visual area 17. Magnetic Resonance in Medicine, 48(4), 735–738. https://doi.org/10.1002/mrm.10255.
Bitter, T., Bruderle, J., Gudziol, H., Burmeister, H. P., Gaser, C., & Guntinas-Lichius, O. (2010a). Gray and white matter reduction in hyposmic subjects–A voxel-based morphometry study. Brain Research, 1347, 42–47. https://doi.org/10.1016/j.brainres.2010.06.003.
Bitter, T., Gudziol, H., Burmeister, H. P., Mentzel, H. J., Guntinas-Lichius, O., & Gaser, C. (2010b). Anosmia leads to a loss of gray matter in cortical brain areas. Chemical Senses, 35(5), 407–415. https://doi.org/10.1093/chemse/bjq028.
Bourgeois, J. P., & Rakic, P. (1993). Changes of synaptic density in the primary visual cortex of the macaque monkey from fetal to adult stage. Journal of Neuroscience, 13(7), 2801–2820.
Bridge, H., Cowey, A., Ragge, N., & Watkins, K. (2009). Imaging studies in congenital anophthalmia reveal preservation of brain architecture in ‘visual’ cortex. Brain, 132(Pt 12), 3467–3480. https://doi.org/10.1093/brain/awp279.
Burmeister, H. P., Baltzer, P. A., Moslein, C., Bitter, T., Gudziol, H., Dietzel, M.,.. . Kaiser, W. A. (2011). Reproducibility and repeatability of volumetric measurements for olfactory bulb volumetry: which method is appropriate? An update using 3 T MRI. Academic Radiology, 18(7), 842–849. https://doi.org/10.1016/j.acra.2011.02.018.
Burmeister, H. P., Bitter, T., Heiler, P. M., Irintchev, A., Frober, R., Dietzel, M.,.. . Kaiser, W. A. (2012). Imaging of lamination patterns of the adult human olfactory bulb and tract: in vitro comparison of standard- and high-resolution 3T MRI, and MR microscopy at 9.4 T. Neuroimage, 60(3), 1662–1670. https://doi.org/10.1016/j.neuroimage.2012.01.101.
Buschhuter, D., Smitka, M., Puschmann, S., Gerber, J. C., Witt, M., Abolmaali, N. D., & Hummel, T. (2008). Correlation between olfactory bulb volume and olfactory function. Neuroimage, 42(2), 498–502. https://doi.org/10.1016/j.neuroimage.2008.05.004.
Carmichael, S. T., Clugnet, M. C., & Price, J. L. (1994). Central olfactory connections in the macaque monkey. J Comp Neurol, 346(3), 403–434. https://doi.org/10.1002/cne.903460306.
Catani, M., Dell’acqua, F., & Thiebaut de Schotten, M. (2013). A revised limbic system model for memory, emotion and behaviour. Neuroscience and Biobehavioral Reviews, 37(8), 1724–1737. https://doi.org/10.1016/j.neubiorev.2013.07.001.
Delon-Martin, C., Plailly, J., Fonlupt, P., Veyrac, A., & Royet, J. P. (2013). Perfumers’ expertise induces structural reorganization in olfactory brain regions. Neuroimage, 68, 55–62. https://doi.org/10.1016/j.neuroimage.2012.11.044.
Duvernoy, H. M. (1999). The Human Brain (Second edition). Wien New York: Springer.
Eickhoff, S., Walters, N. B., Schleicher, A., Kril, J., Egan, G. F., Zilles, K.,.. . Amunts, K. (2005). High-resolution MRI reflects myeloarchitecture and cytoarchitecture of human cerebral cortex. Human Brain Mapping, 24(3), 206–215. https://doi.org/10.1002/hbm.20082.
Eklund, A., Nichols, T. E., & Knutsson, H. (2016). Cluster failure: Why fMRI inferences for spatial extent have inflated false-positive rates. Proceedings of the National Academy of Sciences of the United States of America, 113(28), 7900–7905. https://doi.org/10.1073/pnas.1602413113.
Forkel, S. J., Thiebaut de Schotten, M., Kawadler, J. M., Dell’Acqua, F., Danek, A., & Catani, M. (2014). The anatomy of fronto-occipital connections from early blunt dissections to contemporary tractography. Cortex, 56, 73–84. https://doi.org/10.1016/j.cortex.2012.09.005.
Frasnelli, J., Fark, T., Lehmann, J., Gerber, J., & Hummel, T. (2013). Brain structure is changed in congenital anosmia. Neuroimage, 83, 1074–1080. https://doi.org/10.1016/j.neuroimage.2013.07.070.
Frasnelli, J., Lundstrom, J. N., Boyle, J. A., Djordjevic, J., Zatorre, R. J., & Jones-Gotman, M. (2010). Neuroanatomical correlates of olfactory performance. Experimental Brain Research, 201(1), 1–11. https://doi.org/10.1007/s00221-009-1999-7.
Gagnon, L., Kupers, R., & Ptito, M. (2013). Reduced taste sensitivity in congenital blindness. Chemical Senses, 38(6), 509–517. https://doi.org/10.1093/chemse/bjt021.
Gagnon, L., Vestergaard, M., Madsen, K., Karstensen, H. G., Siebner, H., Tommerup, N.,.. . Ptito, M. (2014). Neural correlates of taste perception in congenital olfactory impairment. Neuropsychologia, 62, 297–305. https://doi.org/10.1016/j.neuropsychologia.2014.07.018.
Galliot, E., Comte, A., Magnin, E., Tatu, L., Moulin, T., & Millot, J. L. (2013). Effects of an ambient odor on brain activations during episodic retrieval of objects. Brain Imaging and Behavior, 7(2), 213–219. https://doi.org/10.1007/s11682-012-9218-8.
Gottfried, J. A. (2010). Central mechanisms of odour object perception. Nature Reviews Neuroscience, 11(9), 628–641. https://doi.org/10.1038/nrn2883.
Gottfried, J. A., & Dolan, R. J. (2003). The nose smells what the eye sees: crossmodal visual facilitation of human olfactory perception. Neuron, 39(2), 375–386.
Gottfried, J. A., & Dolan, R. J. (2004). Human orbitofrontal cortex mediates extinction learning while accessing conditioned representations of value. Nature Neuroscience, 7(10), 1144–1152. https://doi.org/10.1038/nn1314.
Gottfried, J. A., & Zald, D. H. (2005). On the scent of human olfactory orbitofrontal cortex: meta-analysis and comparison to non-human primates. Brain Research. Brain Research Reviews, 50(2), 287–304. https://doi.org/10.1016/j.brainresrev.2005.08.004.
Gyllensten, L., Malmfors, T., & Norrlin, M. L. (1966). Growth alteration in the auditory cortex of visually deprived mice. The Journal of Comparative Neurology, 126(3), 463–469. https://doi.org/10.1002/cne.901260308.
Howard, J. D., Plailly, J., Grueschow, M., Haynes, J. D., & Gottfried, J. A. (2009). Odor quality coding and categorization in human posterior piriform cortex. Nature Neuroscience, 12(7), 932–938. https://doi.org/10.1038/nn.2324.
Huart, C., Meusel, T., Gerber, J., Duprez, T., Rombaux, P., & Hummel, T. (2011). The depth of the olfactory sulcus is an indicator of congenital anosmia. AJNR. American Journal of Neuroradiology, 32(10), 1911–1914. https://doi.org/10.3174/ajnr.A2632.
Hummel, T., Damm, M., Vent, J., Schmidt, M., Theissen, P., Larsson, M., & Klussmann, J. P. (2003). Depth of olfactory sulcus and olfactory function. Brain Research, 975(1–2), 85–89.
Hummel, T., Kobal, G., Gudziol, H., & Mackay-Sim, A. (2007). Normative data for the “Sniffin’ Sticks” including tests of odor identification, odor discrimination, and olfactory thresholds: an upgrade based on a group of more than 3,000 subjects. European Archives of Oto-Rhino-Laryngology, 264(3), 237–243. https://doi.org/10.1007/s00405-006-0173-0.
Hummel, T., Smitka, M., Puschmann, S., Gerber, J. C., Schaal, B., & Buschhuter, D. (2011). Correlation between olfactory bulb volume and olfactory function in children and adolescents. Experimental Brain Research, 214(2), 285–291. https://doi.org/10.1007/s00221-011-2832-7.
Hummel, T., Urbig, A., Huart, C., Duprez, T., & Rombaux, P. (2015). Volume of olfactory bulb and depth of olfactory sulcus in 378 consecutive patients with olfactory loss. Journal of Neurology, 262(4), 1046–1051. https://doi.org/10.1007/s00415-015-7691-x.
Illig, K. R. (2005). Projections from orbitofrontal cortex to anterior piriform cortex in the rat suggest a role in olfactory information processing. Journal of Comparative Neurology, 488(2), 224–231. https://doi.org/10.1002/cne.20595.
Jiang, J., Zhu, W., Shi, F., Liu, Y., Li, J., Qin, W.,.. . Jiang, T. (2009). Thick visual cortex in the early blind. The Journal of Neuroscience, 29(7), 2205–2211. https://doi.org/10.1523/JNEUROSCI.5451-08.2009.
Jovicich, J., Czanner, S., Greve, D., Haley, E., van der Kouwe, A., Gollub, R.,.. . Dale, A. (2006). Reliability in multi-site structural MRI studies: effects of gradient non-linearity correction on phantom and human data. Neuroimage, 30(2), 436–443. https://doi.org/10.1016/j.neuroimage.2005.09.046.
Karstensen, H. G., & Tommerup, N. (2012). Isolated and syndromic forms of congenital anosmia. Clinical Genetics, 81(3), 210–215. https://doi.org/10.1111/j.1399-0004.2011.01776.x.
Kjelvik, G., Evensmoen, H. R., Brezova, V., & Haberg, A. K. (2012). The human brain representation of odor identification. Journal of Neurophysiology, 108(2), 645–657. https://doi.org/10.1152/jn.01036.2010.
Kobal, G., Klimek, L., Wolfensberger, M., Gudziol, H., Temmel, A., Owen, C. M.,.. . Hummel, T. (2000). Multicenter investigation of 1,036 subjects using a standardized method for the assessment of olfactory function combining tests of odor identification, odor discrimination, and olfactory thresholds. European Archives of Oto-Rhino-Laryngology, 257(4), 205–211.
Lenroot, R. K., & Giedd, J. N. (2006). Brain development in children and adolescents: insights from anatomical magnetic resonance imaging. Neuroscience and Biobehavioral Reviews, 30(6), 718–729. https://doi.org/10.1016/j.neubiorev.2006.06.001.
Levy, L. M., Degnan, A. J., Sethi, I., & Henkin, R. I. (2013). Anatomic olfactory structural abnormalities in congenital smell loss: magnetic resonance imaging evaluation of olfactory bulb, groove, sulcal, and hippocampal morphology. Journal of Computer Assisted Tomography, 37(5), 650–657. https://doi.org/10.1097/RCT.0b013e31829bfa3b.
Li, W., Howard, J. D., Parrish, T. B., & Gottfried, J. A. (2008). Aversive learning enhances perceptual and cortical discrimination of indiscriminable odor cues. Science, 319(5871), 1842–1845. https://doi.org/10.1126/science.1152837.
Lygonis, C. S. (1969). Familiar absence of olfaction. Hereditas, 61(3), 413–416.
Mai, J., Paxinos, G., & Voss, T. (1997). Atlas of the Human Brain (Third edition). London: Elsevier Academic Press.
Matsunaga, M., Bai, Y., Yamakawa, K., Toyama, A., Kashiwagi, M., Fukuda, K.,.. . Ohira, H. (2013). Brain-immune interaction accompanying odor-evoked autobiographic memory. PLoS One, 8(8), e72523. https://doi.org/10.1371/journal.pone.0072523.
Mueller, A., Rodewald, A., Reden, J., Gerber, J., von Kummer, R., & Hummel, T. (2005). Reduced olfactory bulb volume in post-traumatic and post-infectious olfactory dysfunction. Neuroreport, 16(5), 475–478.
Nasreddine, Z. S., Phillips, N. A., Bedirian, V., Charbonneau, S., Whitehead, V., Collin, I.,.. . Chertkow, H. (2005). The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. Journal of the American Geriatrics Society, 53(4), 695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x.
Oldfield, R. C. (1971). The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia, 9(1), 97–113.
Osterbauer, R. A., Matthews, P. M., Jenkinson, M., Beckmann, C. F., Hansen, P. C., & Calvert, G. A. (2005). Color of scents: chromatic stimuli modulate odor responses in the human brain. Journal of Neurophysiology, 93(6), 3434–3441. https://doi.org/10.1152/jn.00555.2004.
Park, H. J., Lee, J. D., Kim, E. Y., Park, B., Oh, M. K., Lee, S., & Kim, J. J. (2009). Morphological alterations in the congenital blind based on the analysis of cortical thickness and surface area. Neuroimage, 47(1), 98–106. https://doi.org/10.1016/j.neuroimage.2009.03.076.
Plailly, J., Bensafi, M., Pachot-Clouard, M., Delon-Martin, C., Kareken, D. A., Rouby, C.,.. . Royet, J. P. (2005). Involvement of right piriform cortex in olfactory familiarity judgments. Neuroimage, 24(4), 1032–1041. https://doi.org/10.1016/j.neuroimage.2004.10.028.
Plailly, J., Tillmann, B., & Royet, J. P. (2007). The feeling of familiarity of music and odors: the same neural signature? Cereb Cortex, 17(11), 2650–2658. https://doi.org/10.1093/cercor/bhl173.
Qureshy, A., Kawashima, R., Imran, M. B., Sugiura, M., Goto, R., Okada, K.,.. . Fukuda, H. (2000). Functional mapping of human brain in olfactory processing: a PET study. Journal of Neurophysiology, 84(3), 1656–1666.
Ray, J. P., & Price, J. L. (1992). The organization of the thalamocortical connections of the mediodorsal thalamic nucleus in the rat, related to the ventral forebrain-prefrontal cortex topography. The Journal of Comparative Neurology, 323(2), 167–197. https://doi.org/10.1002/cne.903230204.
Roesch, M. R., Stalnaker, T. A., & Schoenbaum, G. (2007). Associative encoding in anterior piriform cortex versus orbitofrontal cortex during odor discrimination and reversal learning. Cereb Cortex, 17(3), 643–652. https://doi.org/10.1093/cercor/bhk009.
Rolls, E. T. (2004). The functions of the orbitofrontal cortex. Brain Cogn, 55(1), 11–29. https://doi.org/10.1016/S0278-2626(03)00277-X.
Rolls, E. T., & Baylis, L. L. (1994). Gustatory, olfactory, and visual convergence within the primate orbitofrontal cortex. The Journal of Neuroscience, 14(9), 5437–5452.
Rombaux, P., Duprez, T., & Hummel, T. (2009). Olfactory bulb volume in the clinical assessment of olfactory dysfunction. Rhinology, 47(1), 3–9.
Rosser, D. A., Laidlaw, D. A., & Murdoch, I. E. (2001). The development of a “reduced logMAR” visual acuity chart for use in routine clinical practice. The British Journal of Ophthalmology, 85(4), 432–436.
Royet, J. P., Hudry, J., Zald, D. H., Godinot, D., Gregoire, M. C., Lavenne, F.,.. . Holley, A. (2001). Functional neuroanatomy of different olfactory judgments. Neuroimage, 13(3), 506–519. https://doi.org/10.1006/nimg.2000.0704.
Royet, J. P., Koenig, O., Gregoire, M. C., Cinotti, L., Lavenne, F., Le Bars, D.,.. . Froment, J. C. (1999). Functional anatomy of perceptual and semantic processing for odors. Journal of Cognitive Neuroscience, 11(1), 94–109.
Ruiz-Marcos, A., & Valverde, F. (1969). The temporal evolution of the distribution of dendritic spines in the visual cortex of normal and dark raised mice. Experimental Brain Research, 8(3), 284–294.
Ryugo, D. K., Ryugo, R., Globus, A., & Killackey, H. P. (1975). Increased spine density in auditory cortex following visual or somatic deafferentation. Brain Research, 90(1), 143–146.
Savic, I., Gulyas, B., Larsson, M., & Roland, P. (2000). Olfactory functions are mediated by parallel and hierarchical processing. Neuron, 26(3), 735–745.
Schmahmann, J. D., Pandya, D. N., Wang, R., Dai, G., D’Arceuil, H. E., de Crespigny, A. J., & Wedeen, V. J. (2007). Association fibre pathways of the brain: parallel observations from diffusion spectrum imaging and autoradiography. Brain, 130(Pt 3), 630–653. https://doi.org/10.1093/brain/awl359.
Seubert, J., Freiherr, J., Djordjevic, J., & Lundstrom, J. N. (2013). Statistical localization of human olfactory cortex. Neuroimage, 66, 333–342. https://doi.org/10.1016/j.neuroimage.2012.10.030.
Seubert, J., Freiherr, J., Frasnelli, J., Hummel, T., & Lundstrom, J. N. (2013). Orbitofrontal cortex and olfactory bulb volume predict distinct aspects of olfactory performance in healthy subjects. Cereb Cortex, 23(10), 2448–2456. https://doi.org/10.1093/cercor/bhs230.
Sowell, E. R., Thompson, P. M., Leonard, C. M., Welcome, S. E., Kan, E., & Toga, A. W. (2004). Longitudinal mapping of cortical thickness and brain growth in normal children. The Journal of Neuroscience, 24(38), 8223–8231. https://doi.org/10.1523/JNEUROSCI.1798-04.2004.
Stiles, J., & Jernigan, T. L. (2010). The basics of brain development. Neuropsychology Review, 20(4), 327–348. https://doi.org/10.1007/s11065-010-9148-4.
Valverde, F. (1971). Rate and extent of recovery from dark rearing in the visual cortex of the mouse. Brain Research, 33(1), 1–11.
Voss, P., Pike, B. G., & Zatorre, R. J. (2014). Evidence for both compensatory plastic and disuse atrophy-related neuroanatomical changes in the blind. Brain, 137(Pt 4), 1224–1240. https://doi.org/10.1093/brain/awu030.
Voss, P., & Zatorre, R. J. (2012). Organization and reorganization of sensory-deprived cortex. Current Biology, 22(5), R168–R173. https://doi.org/10.1016/j.cub.2012.01.030.
Weiss, T., & Sobel, N. (2012). What’s primary about primary olfactory cortex? Nature Neuroscience, 15(1), 10–12. https://doi.org/10.1038/nn.3009.
Wilson, D. A., Best, A. R., & Brunjes, P. C. (2000). Trans-neuronal modification of anterior piriform cortical circuitry in the rat. Brain Research, 853(2), 317–322.
Zatorre, R. J., Fields, R. D., & Johansen-Berg, H. (2012). Plasticity in gray and white: neuroimaging changes in brain structure during learning. Nature Neuroscience, 15(4), 528–536. https://doi.org/10.1038/nn.3045.
Zufferey, P. D., Jin, F., Nakamura, H., Tettoni, L., & Innocenti, G. M. (1999). The role of pattern vision in the development of cortico-cortical connections. The European Journal of Neuroscience, 11(8), 2669–2688.
Zulauf, M., LeBlanc, R. P., & Flammer, J. (1994). Normal visual fields measured with Octopus-Program G1. II. Global visual field indices. Graefes Archive for Clinical and Experimental Ophthalmology, 232(9), 516–522.
Acknowledgements
The authors thank the participants for their invaluable contribution. The authors wish to acknowledge the important contribution made by Léa Gagnon, PhD, Maurice Ptito, Professor, PhD and Ron Kupers, Professor, PhD in the assessment of olfaction and taste.
Funding
The study was supported by the Lundbeck Foundation (R32-A2947), Chevron Texaco, the Faroese Research Council (0313) and the Danish National Research Foundation (02-512-48), Gangstedfonden, Ville Heises Legat and Hvidovre Hospital’s Research Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Hartwig R. Siebner (HRS) has served on a scientific advisory board for Lundbeck A/S, Valby Denmark. HRS has received honoraria as speaker from Biogen Idec, Denmark A/S, Genzyme, Denmark and MerckSerono, Denmark. HRS has received honoraria as editor from Elsevier Publishers, Amsterdam, The Netherlands and Springer Publishing, Stuttgart, Germany. HRS has received travel support from MagVenture, Denmark, and a research fund from Biogen-idec. All other authors have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.

Rights and permissions
About this article
Cite this article
Karstensen, H.G., Vestergaard, M., Baaré, W.F.C. et al. Congenital olfactory impairment is linked to cortical changes in prefrontal and limbic brain regions. Brain Imaging and Behavior 12, 1569–1582 (2018). https://doi.org/10.1007/s11682-017-9817-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-017-9817-5