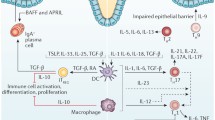
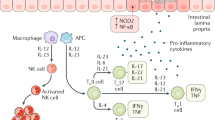
Abstract
Crohn’s disease is a chronic inflammatory disorder primarily affecting the gastrointestinal tract. Its clinical manifestations arise from a substantial infiltration of the intestinal mucosa by activated leukocytes and the downstream consequences of chronic inflammation. The underlying cause driving this immunological reaction remains poorly understood. A number of hypotheses have been proposed, most of which postulate a primary over-activation of the immune response, based on the pathological appearances of active Crohn’s lesions. Interestingly, none of these theories have been mechanistically proven. It is possible that the immunological events responsible for disease initiation are quite different from those contributing to its persistence and propagation. A substantial body of data has emerged in recent years to suggest that the primary defect in Crohn’s disease is actually one of relative immunodeficiency. This review considers the evidence for such a phenomenon in contrast to alternative prevailing hypotheses and attempts to address some of the potential paradoxes that it generates.

Similar content being viewed by others
References
Shanahan F (2002) Crohn’s disease. Lancet 359(9300):62–69
Podolsky DK (2002) Inflammatory bowel disease. N Engl J Med 347(6):417–429
Segal AW, Loewi G (1976) Neutrophil dysfunction in Crohn’s disease. Lancet 2(7979):219–221
Marks DJ, Segal AW (2008) Innate immunity in inflammatory bowel disease: a disease hypothesis. J Pathol 214(2):260–266
Barrett JC et al (2008) Genome-wide association defines more than 30 distinct susceptibility loci for Crohn’s disease. Nat Genet 40(8):955–962
Rahman FZ et al (2008) Phagocyte dysfunction and inflammatory bowel disease. Inflamm Bowel Dis 14(10):1443–1452
Rutgeerts P et al (1991) Effect of faecal stream diversion on recurrence of Crohn’s disease in the neoterminal ileum. Lancet 338(8770):771–774
Harper PH et al (1985) Role of the faecal stream in the maintenance of Crohn’s colitis. Gut 26(3):279–284
Dalziel TK (1913) Chronic intestinal enteritis. BMJ ii:1068–1070
Chiodini RJ et al (1984) Possible role of mycobacteria in inflammatory bowel disease. I. An unclassified Mycobacterium species isolated from patients with Crohn’s disease. Dig Dis Sci 29(12):1073–1079
Feller M et al (2007) Mycobacterium avium subspecies paratuberculosis and Crohn’s disease: a systematic review and meta-analysis. Lancet Infect Dis 7(9):607–613
Quirke P (2001) Antagonist. Mycobacterium avium subspecies paratuberculosis is a cause of Crohn’s disease. Gut 49(6):757–760
Van Kruiningen HJ (1999) Lack of support for a common etiology in Johne’s disease of animals and Crohn’s disease in humans. Inflamm Bowel Dis 5(3):183–191
Selby W et al (2007) Two-year combination antibiotic therapy with clarithromycin, rifabutin, and clofazimine for Crohn’s disease. Gastroenterology 132(7):2313–2319
Darfeuille-Michaud A et al (2004) High prevalence of adherent-invasive Escherichia coli associated with ileal mucosa in Crohn’s disease. Gastroenterology 127(2):412–421
Glasser AL et al (2001) Adherent invasive Escherichia coli strains from patients with Crohn’s disease survive and replicate within macrophages without inducing host cell death. Infect Immun 69(9):5529–5537
Berin MC et al (2002) Role of EHEC O157:H7 virulence factors in the activation of intestinal epithelial cell NF-kappaB and MAP kinase pathways and the upregulated expression of interleukin 8. Cell Microbiol 4(10):635–648
Meconi S et al (2007) Adherent-invasive Escherichia coli isolated from Crohn’s disease patients induce granulomas in vitro. Cell Microbiol 9(5):1252–1261
Marks DJ et al (2006) Can unresolved infection precipitate autoimmune disease? Curr Top Microbiol Immunol 305:105–125
Frank DN, Pace NR (2008) Gastrointestinal microbiology enters the metagenomics era. Curr Opin Gastroenterol 24(1):4–10
Ainsworth M et al (1989) Intestinal permeability of 51Cr-labelled ethylenediaminetetraacetic acid in patients with Crohn’s disease and their healthy relatives. Scand J Gastroenterol 24(8):993–998
Hollander D et al (1986) Increased intestinal permeability in patients with Crohn’s disease and their relatives. A possible etiologic factor. Ann Intern Med 105(6):883–885
Marin ML et al (1983) A freeze fracture study of Crohn’s disease of the terminal ileum: changes in epithelial tight junction organization. Am J Gastroenterol 78(9):537–547
Kyo K et al (2001) Associations of distinct variants of the intestinal mucin gene MUC3A with ulcerative colitis and Crohn’s disease. J Hum Genet 46(1):5–20
Buisine MP et al (2001) Mucin gene expression in intestinal epithelial cells in Crohn’s disease. Gut 49(4):544–551
Clamp JR, Fraser G, Read AE (1981) Study of the carbohydrate content of mucus glycoproteins from normal and diseased colons. Clin Sci (Lond) 61(2):229–234
Pullan RD et al (1994) Thickness of adherent mucus gel on colonic mucosa in humans and its relevance to colitis. Gut 35(3):353–359
Hugot JP et al (1996) Mapping of a susceptibility locus for Crohn’s disease on chromosome 16. Nature 379(6568):821–823
Hugot JP et al (2001) Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature 411(6837):599–603
Ogura Y et al (2001) A frameshift mutation in NOD2 associated with susceptibility to Crohn’s disease. Nature 411(6837):603–606
Cuthbert AP et al (2002) The contribution of NOD2 gene mutations to the risk and site of disease in inflammatory bowel disease. Gastroenterology 122(4):867–874
Ogura Y et al (2001) Nod2, a Nod1/Apaf-1 family member that is restricted to monocytes and activates NF-kappaB. J Biol Chem 276(7):4812–4818
Gutierrez O et al (2002) Induction of Nod2 in myelomonocytic and intestinal epithelial cells via nuclear factor-kappa B activation. J Biol Chem 277(44):41701–41705
Lala S et al (2003) Crohn’s disease and the NOD2 gene: a role for paneth cells. Gastroenterology 125(1):47–57
Girardin SE et al (2003) Nod2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection. J Biol Chem 278(11):8869–8872
Inohara N et al (2003) Host recognition of bacterial muramyl dipeptide mediated through NOD2. Implications for Crohn’s disease. J Biol Chem 278(8):5509–5512
Li J et al (2004) Regulation of IL-8 and IL-1beta expression in Crohn’s disease associated NOD2/CARD15 mutations. Hum Mol Genet 13(16):1715–1725
Marks DJ et al (2006) Defective acute inflammation in Crohn’s disease: a clinical investigation. Lancet 367(9511):668–678
Rioux JD et al (2007) Genome-wide association study identifies new susceptibility loci for Crohn disease and implicates autophagy in disease pathogenesis. Nat Genet 39(5):596–604
Borgiani P et al (2007) Interleukin-23R Arg381Gln is associated with susceptibility to Crohn’s disease but not with phenotype in an Italian population. Gastroenterology 133(3):1049–1051
Weersma RK et al (2009) Confirmation of multiple Crohn's Disease susceptibility loci in a large Dutch-Belgian cohort. Am J Gastroenterol 104(3):630–638
Maloy KJ (2008) The interleukin-23/interleukin-17 axis in intestinal inflammation. J Intern Med 263(6):584–590
Stuart LM, Ezekowitz RA (2005) Phagocytosis: elegant complexity. Immunity 22(5):539–550
Kuballa P et al (2008) Impaired autophagy of an intracellular pathogen induced by a Crohn’s disease associated ATG16L1 variant. PLoS ONE 3(10):e3391
Singh SB et al (2006) Human IRGM induces autophagy to eliminate intracellular mycobacteria. Science 313(5792):1438–1441
Torok HP et al (2005) Polymorphisms in the DLG5 and OCTN cation transporter genes in Crohn’s disease. Gut 54(10):1421–1427
Rioux JD et al (2001) Genetic variation in the 5q31 cytokine gene cluster confers susceptibility to Crohn disease. Nat Genet 29(2):223–228
Peltekova VD et al (2004) Functional variants of OCTN cation transporter genes are associated with Crohn disease. Nat Genet 36(5):471–475
Nakamura H et al (1998) Identification of a novel human homolog of the Drosophila dlg, P-dlg, specifically expressed in the gland tissues and interacting with p55. FEBS Lett 433(1–2):63–67
Maher B (2008) Personal genomes: the case of the missing heritability. Nature 456(7218):18–21
Kuramoto S et al (1987) Granulomas of the gut in Crohn’s disease. A step sectioning study. Dis Colon Rectum 30(1):6–11
James DG (2000) A clinicopathological classification of granulomatous disorders. Postgrad Med J 76(898):457–465
Reeves EP et al (2002) Killing activity of neutrophils is mediated through activation of proteases by K+ flux. Nature 416(6878):291–297
Thrasher AJ et al (1994) Chronic granulomatous disease. Biochim Biophys Acta 1227(1–2):1–24
Marks DJ et al (2009) Inflammatory bowel disease in CGD reproduces the clinicopathological features of Crohn’s disease. Am J Gastroenterol 104(1):117–124
Ishii E et al (1987) Chediak-Higashi syndrome with intestinal complication. Report of a case. J Clin Gastroenterol 9(5):556–558
Grucela AL et al (2006) Granulomatous enterocolitis associated with Hermansky-Pudlak syndrome. Am J Gastroenterol 101(9):2090–2095
O’Morain C, Segal AW, Levi AJ (1984) Elemental diet as primary treatment of acute Crohn’s disease: a controlled trial. Br Med J (Clin Res Ed) 288(6434):1859–1862
Rhodes JM et al (1982) Serum inhibitors of leukocyte chemotaxis in Crohn’s disease and ulcerative colitis. Gastroenterology 82(6):1327–1334
Farrell RJ, Peppercorn MA (2002) Ulcerative colitis. Lancet 359(9303):331–340
Harbord MW et al (2006) Impaired neutrophil chemotaxis in Crohn’s disease relates to reduced production of chemokines and can be augmented by granulocyte-colony stimulating factor. Aliment Pharmacol Ther 24(4):651–660
Strober W, Fuss IJ, Blumberg RS (2002) The immunology of mucosal models of inflammation. Annu Rev Immunol 20:495–549
Vavricka SR et al (2004) hPepT1 transports muramyl dipeptide, activating NF-kappaB and stimulating IL-8 secretion in human colonic Caco2/bbe cells. Gastroenterology 127(5):1401–1409
Doyle SL, O’Neill LA (2006) Toll-like receptors: from the discovery of NFkappaB to new insights into transcriptional regulations in innate immunity. Biochem Pharmacol 72(9):1102–1113
Marks DJ et al (2006) An exuberant inflammatory response to E. coli: implications for the pathogenesis of ulcerative colitis and pyoderma gangrenosum. Gut 55(11):1662–1663
Castell JV et al (1989) Interleukin-6 is the major regulator of acute phase protein synthesis in adult human hepatocytes. FEBS Lett 242(2):237–239
Bridger S et al (2002) In siblings with similar genetic susceptibility for inflammatory bowel disease, smokers tend to develop Crohn’s disease and non-smokers develop ulcerative colitis. Gut 51(1):21–25
Sopori M (2002) Effects of cigarette smoke on the immune system. Nat Rev Immunol 2(5):372–377
Sher ME et al (1999) The influence of cigarette smoking on cytokine levels in patients with inflammatory bowel disease. Inflamm Bowel Dis 5(2):73–78
Hugot JP et al (2003) Crohn’s disease: the cold chain hypothesis. Lancet 362(9400):2012–2015
Liu AH, Murphy JR (2003) Hygiene hypothesis: fact or fiction? J Allergy Clin Immunol 111(3):471–478
Elliott DE et al (2000) Does the failure to acquire helminthic parasites predispose to Crohn’s disease? FASEB J 14(12):1848–1855
Summers RW et al (2005) Trichuris suis therapy in Crohn’s disease. Gut 54(1):87–90
Farthing MJ (2004) Bugs and the gut: an unstable marriage. Best Pract Res Clin Gastroenterol 18(2):233–239
Porter CK et al (2008) Infectious gastroenteritis and risk of developing inflammatory bowel disease. Gastroenterology 135(3):781–786
Bednarz W et al (2008) Analysis of results of surgical treatment in Crohn’s disease. Hepatogastroenterology 55(84):998–1001
Scammell BE, Keighley MR (1986) Delayed perineal wound healing after proctectomy for Crohn’s colitis. Br J Surg 73(2):150–152
Kyle J (1980) Urinary complications of Crohn’s disease. World J Surg 4(2):153–160
Ambrose NS, Alexander-Williams J, Keighley MR (1984) Audit of sepsis in operations for inflammatory bowel disease. Dis Colon Rectum 27(9):602–604
Hutfless SM et al (2007) Mortality by medication use among patients with inflammatory bowel disease, 1996–2003. Gastroenterology 133(6):1779–1786
Gilroy DW et al (2004) Inflammatory resolution: new opportunities for drug discovery. Nat Rev Drug Discov 3(5):401–416
Nahar IK et al (2003) Infliximab treatment of rheumatoid arthritis and Crohn’s disease. Ann Pharmacother 37(9):1256–1265
Cui G et al (2006) Improvement of real-time polymerase chain reaction for quantifying TNF-alpha mRNA expression in inflamed colorectal mucosa: an approach to optimize procedures for clinical use. Scand J Clin Lab Invest 66(3):249–259
Dionne S et al (1997) Quantitative PCR analysis of TNF-alpha and IL-1 beta mRNA levels in pediatric IBD mucosal biopsies. Dig Dis Sci 42(7):1557–1566
Stallmach A et al (2004) Cytokine/chemokine transcript profiles reflect mucosal inflammation in Crohn’s disease. Int J Colorectal Dis 19(4):308–315
Plevy SE et al (1997) A role for TNF-alpha and mucosal T helper-1 cytokines in the pathogenesis of Crohn’s disease. J Immunol 159(12):6276–6282
Sandborn WJ et al (2001) Etanercept for active Crohn’s disease: a randomized, double-blind, placebo-controlled trial. Gastroenterology 121(5):1088–1094
Van den Brande JM et al (2003) Infliximab but not etanercept induces apoptosis in lamina propria T-lymphocytes from patients with Crohn’s disease. Gastroenterology 124(7):1774–1785
Naito Y et al (2003) Enhanced intestinal inflammation induced by dextran sulfate sodium in tumor necrosis factor-alpha deficient mice. J Gastroenterol Hepatol 18(5):560–569
Oh J, Arkfeld DG, Horwitz DA (2005) Development of Crohn’s disease in a patient taking etanercept. J Rheumatol 32(4):752–753
Charach G, Grosskopf I, Weintraub M (2008) Development of Crohn’s disease in a patient with multiple sclerosis treated with copaxone. Digestion 77(3–4):198–200
Saverymuttu S, Hodgson HJ, Chadwick VS (1985) Controlled trial comparing prednisolone with an elemental diet plus non-absorbable antibiotics in active Crohn’s disease. Gut 26(10):994–998
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marks, D.J.B., Rahman, F.Z., Sewell, G.W. et al. Crohn’s Disease: an Immune Deficiency State. Clinic Rev Allerg Immunol 38, 20–31 (2010). https://doi.org/10.1007/s12016-009-8133-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-009-8133-2