Abstract
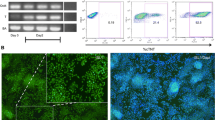
Induced pluripotent stem cell (iPS) technology has launched a new platform in regenerative medicine aimed at deriving unlimited replacement tissue from autologous sources through somatic cell reprogramming using stemness factor sets. In this way, authentic cardiomyocytes have been obtained from iPS and recently demonstrated in proof-of-principle studies to repair infarcted heart. Optimizing the cardiogenic potential of iPS progeny would ensure a maximized yield of bioengineered cardiac tissue. Here, we reprogrammed fibroblasts in the presence or absence of c-MYC to determine if the acquired cardiogenicity is sensitive to the method of nuclear reprogramming. Using lentiviral constructs that expressed stemness factors SOX2, OCT4, and KLF4 with or without c-MYC, iPS clones generated through fibroblast reprogramming demonstrated indistinguishable characteristics for 5 days of differentiation with similar cell morphology, growth rates, and chimeric embryo integration. However, four-factor c-MYC-dependent nuclear reprogramming produced iPS progeny that consistently prolonged the expression of pluripotent Oct4 and Fgf4 genes and repressed cardiac differentiation. In contrast, three-factor c-MYC-less iPS clones efficiently upregulated precardiac (CXCR4, Flk1, and Mesp1/2) and cardiac (Nkx2.5, Mef2c, and myocardin) gene expression patterns. In fact, three-factor iPS progeny demonstrated early and robust cardiogenesis during in vitro differentiation with consistent beating activity, sarcomere maturation, and rhythmical intracellular calcium dynamics. Thus, nuclear reprogramming independent of c-MYC enhances production of pluripotent stem cells with innate cardiogenic potential.






Similar content being viewed by others
References
Yamanaka, S. (2007). Strategies and new developments in the generation of patient-specific pluripotent stem cells. Cell Stem Cell, 1(1), 39–49.
Takahashi, K., Okita, K., Nakagawa, M., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126(4), 663–676.
Okita, K., Ichisaka, T., & Yamanaka, S. (2007). Generation of germ line-competent induced pluripotent stem cells. Nature, 448(7151), 313–317.
Nelson, T. J., Martinez-Fernandez, A. J., Yamada, S., Mael, A. A., Terzic, A., & Ikeda, Y. (2009). Induced pluripotent reprogramming from promiscuous human stemness-related factors. Clinical and Translational Science, 2(2), 118–126.
Wernig, M., Meissner, A., Foreman, R., Brambrink, T., Ku, M., Hochedlinger, K., et al. (2007). In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature, 448(7151), 318–324.
Yu, J., Vodyanik, M. A., Smuga-Otto, K., Antosiewicz-Bourget, J., Frane, J. L., Tian, S., et al. (2007). Induced pluripotent stem cell lines derived from human somatic cells. Science, 318(5858), 1917–1920.
Hochedlinger, K., & Plath, K. (2009). Epigenetic reprogramming and induced pluripotency. Development, 136(4), 509–523.
Nishikawa, S., Goldstein, R. A., & Nierras, C. R. (2008). The promise of human induced pluripotent stem cells for research and therapy. Nature Reviews Molecular Cell Biology, 9(9), 725–729.
Park, I. H., Arora, N., Huo, H., Maherali, N., Ahfeldt, T., Shimamura, A., et al. (2008). Disease-specific induced pluripotent stem cells. Cell, 134(5), 877–886.
Hanna, J., Wernig, M., Markoulaki, S., Sun, C. W., Meissner, A., Cassady, J. P., et al. (2007). Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science, 318(5858), 1920–1923.
Wernig, M., Zhao, J. P., Pruszak, J., Hedlund, E., Fu, D., Soldner, F., et al. (2008). Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson's disease. Proceedings of the National Academy of Sciences of the United States of America, 105(14), 5856–5861.
Xu, D., Alipio, Z., Fink, L. M., Adcock, D. M., Yang, J., Ward, D. C., et al. (2009). Phenotypic correction of murine hemophilia A using an iPS cell-based therapy. Proceedings of the National Academy of Sciences of the United States of America, 106(3), 808–813.
Nelson, T. J., Martinez-Fernandez, A., Yamada, S., Perez-Terzic, C., Ikeda, Y., & Terzic, A. (2009). Repair of acute myocardial infarction with human stemness factors induced pluripotent stem cells. Circulation, 120(5), 408–416.
Schenke-Layland, K., Rhodes, K. E., Angelis, E., Butylkova, Y., Heydarkhan-Hagvall, S., Gekas, C., et al. (2008). Reprogrammed mouse fibroblasts differentiate into cells of the cardiovascular and hematopoietic lineages. Stem Cells, 26(6), 1537–1546.
Mauritz, C., Schwanke, K., Reppel, M., Neef, S., Katsirntaki, K., Maier, L. S., et al. (2008). Generation of functional murine cardiac myocytes from induced pluripotent stem cells. Circulation, 118(5), 507–517.
Narazaki, G., Uosaki, H., Teranishi, M., Okita, K., Kim, B., Matsuoka, S., et al. (2008). Directed and systematic differentiation of cardiovascular cells from mouse induced pluripotent stem cells. Circulation, 118(5), 498–506.
Zhang, J., Wilson, G., Soerens, A., Koonce, C., Yu, J., Palecek, S., et al. (2009). Functional cardiomyocytes derived from human induced pluripotent stem cells. Circulation Research, 104(4), e30–e41.
Martinez-Fernandez, A., Nelson, T. J., Yamada, S., Reyes, S., Alekseev, A. E., Perez-Terzic, C., et al. (2009). iPS programmed without c-MYC yield proficient cardiogenesis for functional heart chimerism. Circulation Research, 105(7), 648–656.
Dimos, J. T., Rodolfa, K. T., Niakan, K. K., Weisenthal, L. M., Mitsumoto, H., Chung, W., et al. (2008). Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science, 321(5893), 1218–1221.
Raya, A., Rodríguez-Pizà, I., Guenechea, G., Vassena, R., Navarro, S., Barrero, M. J., et al. (2009). Disease-corrected haematopoietic progenitors from Fanconi anaemia induced pluripotent stem cells. Nature, 460(7251), 53–59.
Maehr, R., Chen, S., Snitow, M., Ludwig, T., Yagasaki, L., Goland, R., et al. (2009). Generation of pluripotent stem cells from patients with type 1 diabetes. Proceedings of the National Academy of Sciences of the United States of America, 106(37), 15768–15773.
Lee, G., Papapetrou, E. P., Kim, H., Chambers, S. M., Tomishima, M. J., Fasano, C. A., et al. (2009). Modelling pathogenesis and treatment of familial dysautonomia using patient-specific iPSCs. Nature, 461(7262), 402–406.
Chien, K. R., Domian, I. J., & Parker, K. K. (2008). Cardiogenesis and the complex biology of regenerative cardiovascular medicine. Science, 322(5907), 1494–1497.
Perez-Terzic, C., Faustino, R. S., Boorsma, B. J., Arrell, D. K., Niederländer, N. J., Behfar, A., et al. (2007). Stem cells transform into a cardiac phenotype with remodeling of the nuclear transport machinery. Nature Clinical Practice Cardiovascular Medicine, 4(Suppl 1), S68–S76.
Perez-Terzic, C., Behfar, A., Méry, A., van Deursen, J. M., Terzic, A., & Pucéat, M. (2003). Structural adaptation of the nuclear pore complex in stem cell-derived cardiomyocytes. Circulation Research, 92(4), 444–452.
Nelson, T. J., Martinez-Fernandez, A., & Terzic, A. (2009). KCNJ11 knockout morula reengineered by stem cell diploid aggregation. Philosophical Transactions of the Royal Society London B: Biological Sciences, 364(1514), 269–276.
Behfar, A., Perez-Terzic, C., Faustino, R. S., Arrell, D. K., Hodgson, D. M., Yamada, S., et al. (2007). Cardiopoietic programming of embryonic stem cells for tumor-free heart repair. Journal of Experimental Medicine, 204(2), 405–420.
Nelson, T. J., Chiriac, A., Faustino, R. S., Crespo-Diaz, R. J., Behfar, A., & Terzic, A. (2009). Lineage specification of Flk-1+ progenitors is associated with divergent Sox7 expression in cardiopoiesis. Differentiation, 77(3), 248–255.
Hodgson, D. M., Behfar, A., Zingman, L. V., Kane, G. C., Perez-Terzic, C., Alekseev, A. E., et al. (2004). Stable benefit of embryonic stem cell therapy in myocardial infarction. American Journal of Physiology Heart and Circulatory Physiology, 287(2), H471–H479.
Maherali, N., & Hochedlinger, K. (2008). Guidelines and techniques for the generation of induced pluripotent stem cells. Cell Stem Cell, 3(6), 595–605.
Yamanaka, S. (2008). Pluripotency and nuclear reprogramming. Philosophical Transactions of the Royal Society London B: Biological Science, 363(1500), 2079–2087.
Yamanaka, S. (2009). Elite and stochastic models for induced pluripotent stem cell generation. Nature, 460(7251), 49–52.
Kim, J. B., Sebastiano, V., Wu, G., Araúzo-Bravo, M. J., Sasse, P., Gentile, L., et al. (2009). Oct4-induced pluripotency in adult neural stem cells. Cell, 136(3), 411–419.
Nakagawa, M., Koyanagi, M., Tanabe, K., Takahashi, K., Ichisaka, T., Aoi, T., et al. (2008). Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nature Biotechnology, 26(1), 101–106.
Wernig, M., Meissner, A., Cassady, J. P., & Jaenisch, R. (2008). c-Myc is dispensable for direct reprogramming of mouse fibroblasts. Cell Stem Cell, 2(1), 10–12.
Guo, Y., Niu, C., Breslin, P., Tang, M., Zhang, S., Wei, W., et al. (2009). c-Myc-mediated control of cell fate in megakaryocyte–erythrocyte progenitors. Blood, 114(10), 2097–2106.
Izumo, S., Nadal-Ginard, B., & Mahdavi, V. (1988). Protooncogene induction and reprogramming of cardiac gene expression produced by pressure overload. Proceedings of the National Academy of Sciences of the United States of America, 85(2), 339–343.
Baudino, T. A., McKay, C., Pendeville-Samain, H., Nilsson, J. A., Maclean, K. H., White, E. L., et al. (2002). c-Myc is essential for vasculogenesis and angiogenesis during development and tumor progression. Genes and Development, 16(19), 2530–2543.
Jackson, T., Allard, M. F., Sreenan, C. M., Doss, L. K., Bishop, S. P., & Swain, J. L. (1990). The c-myc proto-oncogene regulates cardiac development in transgenic mice. Molecular Cell Biology, 10(7), 3709–3716.
Xiao, G., Mao, S., Baumgarten, G., Serrano, J., Jordan, M. C., Roos, K. P., et al. (2001). Inducible activation of c-Myc in adult myocardium in vivo provokes cardiac myocyte hypertrophy and reactivation of DNA synthesis. Circulation Research, 89(12), 1122–1129.
Zhong, W., Mao, S., Tobis, S., Angelis, E., Jordan, M. C., Roos, K. P., et al. (2006). Hypertrophic growth in cardiac myocytes is mediated by Myc through a cyclin D2-dependent pathway. EMBO Journal, 25(16), 3869–3879.
Kidder, B. L., Yang, J., & Palmer, S. (2008). Stat3 and c-Myc genome-wide promoter occupancy in embryonic stem cells. PLoS ONE, 3(12), e3932.
Brambrink, T., Foreman, R., Welstead, G. G., Lengner, C. J., Wernig, M., Suh, H., et al. (2008). Sequential expression of pluripotency markers during direct reprogramming of mouse somatic cells. Cell Stem Cell, 2(2), 151–159.
Nelson, T. J., Behfar, A., & Terzic, A. (2008). Stem cells: biologics for regeneration. Clinical Pharmacology and Therapeutics, 84(5), 620–623.
Nelson, T. J., Behfar, A., & Terzic, A. (2008). Strategies for therapeutic repair: the “R3” regenerative medicine paradigm. Clinical and Translational Science, 1(2), 168–171.
Nelson, T. J., Behfar, A., Yamada, S., Martinez-Fernandez, A., & Terzic, A. (2009). Stem cell platforms for regenerative medicine. Clinical and Translational Science, 2(3), 222–227.
Byrne, J. A., Nguyen, H. N., & Reijo Pera, R. A. (2009). Enhanced generation of induced pluripotent stem cells from a subpopulation of human fibroblasts. PLoS ONE, 4(9), e7118.
Acknowledgements
The authors thank James E. Tarara and the Mayo Clinic Flow Cytometry and Optical Morphology Resource Core for their expertise. This work was supported by National Institutes of Health (R01HL083439, T32HL007111, R56AI074363), Caja Madrid Graduate Program, Marriott Individualized Medicine Program, Marriott Heart Disease Research Program, and Mayo Clinic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martinez-Fernandez, A., Nelson, T.J., Ikeda, Y. et al. c-MYC-Independent Nuclear Reprogramming Favors Cardiogenic Potential of Induced Pluripotent Stem Cells. J. of Cardiovasc. Trans. Res. 3, 13–23 (2010). https://doi.org/10.1007/s12265-009-9150-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-009-9150-5