Abstract
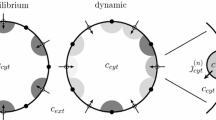
Although there is consensus that localized Ca2+ elevations known as Ca2+ puffs and sparks arise from the cooperative activity of intracellular Ca2+ channels, the precise relationship between single-channel kinetics and the collective phenomena of stochastic Ca2+ excitability is not well understood. Here we present a formalism by which mathematical models for Ca2+-regulated Ca2+ release sites are derived from stochastic models of single-channel gating that include Ca2+ activation, Ca2+ inactivation, or both. Such models are stochastic automata networks (SANs) that involve a large number of functional transitions, that is, the transition probabilities of the infinitesimal generator matrix of one of the automata (i.e., an individual channel) may depend on the local [Ca2+] and thus the state of the other channels. Simulation and analysis of the SAN descriptors representing homogeneous clusters of intracellular Ca2+ channels show that (1) release site density can modify both the steady-state open probability and stochastic excitability of Ca2+ release sites, (2) Ca2+ inactivation is not a requirement for Ca2+ puffs or sparks, and (3) a single-channel model with a bell-shaped open probability curve does not lead to release site activity that is a biphasic function of release site density. These findings are obtained using iterative, memory-efficient methods (novel in this biophysical context and distinct from Monte Carlo simulation) that leverage the highly structured SAN descriptor to unambiguously calculate the steady-state probability of each release site configuration and puff statistics such as puff duration and inter-puff interval. The validity of a mean field approximation that neglects the spatial organization of Ca2+ release sites is also discussed.
Similar content being viewed by others
References
Allbritton, N., Meyer, T., Stryer, L., 1992. Range of messenger action of Ca2+ ion and inositol 1,4,5-trisphosphate. Science 258, 1812–1815.
Asmussen, S., Bladt, M., 1997. Renewal theory and queueing algorithms for matrix-exponential distributions. In: Chakravarthy, S., Alfa, A. (Eds.), Matrix-Analytic Methods in Stochastic Models. Marcel Dekker, Inc., New York, pp. 313–341.
Atri, A., Amundson, J., Clapham, D., Sneyd, J., 1993. A single-pool model for intracellular Ca2+ oscillations and waves in the Xenopus laevis oocyte. Biophys. J. 65, 1727–1739.
Berridge, M., 1993. Inositol trisphosphate and Ca2+ signaling. Nature 361, 315–325.
Berridge, M., 1997. Elementary and global aspects of Ca2+ signalling. J. Physiol. (London) 499, 291–306.
Berridge, M., 1998. Neuronal Ca2+ signaling. Neuron 21, 13–26.
Bers, D., 1992. Excitation-Contraction Coupling and Cardiac Contractile Force, 2nd edition. Kluwer Academic Publishers.
Bertram, R., Smith, G., Sherman, A., 1999. Modeling study of the effects of overlapping Ca2+ microdomains on neurotransmitter release. Biophys. J. 76, 735–750.
Bezprozvanny, I., Ehrlich, B., 1994. Inositol (1,4,5)-trisphosphate (IP3)-gated Ca2+ channels from cerebellum: conduction properties for divalent cations and regulation by intraluminal Ca2+. J. Gen. Physiol. 104, 821–856.
Bootman, M., Lipp, P., 1999. Ringing changes to the ‘bell-shaped curve’. Curr. Biol. 9, R876–R878.
Bugrim, A., Zhabotinsky, A., Epstein, I., 1997. Ca2+ waves in a model with a random spatially discrete distribution of Ca2+ release sites. Biophys. J. 73, 2897–2906.
Cannell, M., Cheng, H., Lederer, W., 1995. The control of calcium release in heart muscle. Science 268, 1045–1049.
Cheng, H., Lederer, M., Lederer, W., Cannell, M., 1993a. Ca2+ sparks and [Ca2+]i waves in cardiac myocytes. Am J. Physiol. 270, C148–C159.
Cheng, H., Lederer, W., Cannell, M., 1993b. Ca2+ sparks: elementary events underlying excitation-contraction coupling in heart muscle. Science 262, 740–744.
Chow, C., White, J., 1996. Spontaneous action potentials due to channel fluctuations. Biophys. J. 71, 3013–3021.
Clay, J., DeFelice, L., 1983. Relationship between membrane excitability and single channel open-close kinetics. Biophys. J. 42, 151–157.
Colquhoun, D., Hawkes, A., 1995. A Q-matrix cookbook: how to write only one program to calculate the single-channel and macroscopic predictions for any kinetic mechanism. In: Sakmann, B., Neher, E. (Eds.), Single-Channel Recording. Plenum Press, New York, pp. 589–633.
Dargan, S., Parker, I., 2003. Buffer kinetics shape the spatiotemporal patterns of IP3-evoked Ca2+ signals. J. Physiol. 553, 775–788.
Durrett, R., 1999. Essentials of Stochastic Processes. Springer, New York.
de Souza e Silva, E., Gail, H., 2000. Transient solutions for Markov chains. In: Grassmann, W. (Ed.), Computational Probability. Kluwer Academic Publishers, Boston, pp. 43–79.
De Young, G., Keizer, J., 1992. A single-pool inositol 1,4,5-trisphosphate-receptor-based model for agonist-stimulated oscillations in Ca2+ concentration. Proc. Natl. Acad. Sci. USA 89, 9895–9899.
DeFelice, L., 1981. Introduction to Membrane Noise. Plenum Press, New York.
Falcke, M., Tsimring, L., Levine, H., 2000. Stochastic spreading of intracellular Ca2+ release. Phys. Rev. E Stat. Phys. Plasmas Fluids Relat. Interdiscip. Topics 62, 2636–2643.
Fernandes, P., Plateau, B., Stewart, W., 1998. Efficient descriptor-vector multiplications in stochastic automata networks. J. ACM 45, 381–414.
Fox, R., Lu, Y., 1994. Emergent collective behavior in large numbers of globally coupled independently stochastic ion channels. Phys. Rev. E 49, 3421–3431.
Gabso, M., Neher, E., Spira, M., 1997. Low mobility of the Ca2+ buffers in axons of cultured Aplysia neurons. Neuron 18, 473–481.
Gillespie, D., 1977. Exact stochastic simulation of coupled chemical reactions. J. Phys. Chem. 81, 2340–2361.
Golub, G., Loan, C.V., 1996. Matrix Computations. Johns Hopkins University Press.
Hagar, R., Burgstahler, A., Nathanson, M., Ehrlich, B., 1998. Type III IP3 receptor channel stays open in the presence of increased Ca2+. Nature 396, 81–84.
Hill, T., 1977. Free Energy Transduction in Biology: The Steady-State Kinetic and Thermodynamic Formalism. Academic Press, New York.
Horn, R., Johnson, C., 1991. Topics in Matrix Analysis. Cambridge University Press.
Joseph, S., 1996. The inositol triphosphate receptor family. Cell Signal 8, 1–7.
Kaftan, E., Ehrlich, B., Watras, J., 1997. Inositol 1,4,5-trisphosphate (IP3) and Ca2+ interact to increase the dynamic range of IP3 receptor-dependent Ca2+ signaling. J. Gen. Physiol. 110, 529–538.
Keizer, J., 1987. Statistical Thermodynamics of Nonequilibrium Processes. Springer, Berlin.
Kemeny, J., Snell, J., 1976. Finite Markov Chains. Springer.
Langville, A., Stewart, W., 2003. A Kronecker product approximate preconditioner for SANs. Numer. Linear Algebra Appl. 10, 1–29.
Langville, A., Stewart, W., 2004. The Kronecker product and stochastic automata networks. J. Comput. Appl. Math. 167(2), 429–447.
Latouche, G., Ramaswami, V., 1999. Introduction to Matrix Analytic Methods in Stochastic Modeling. Society for Industrial and Applied Mathematics, Philadelphia, PA.
LeBeau, A., Yule, D., Groblewski, G., Sneyd, J., 1999. Agonist-dependent phosphorylation of the inositol 1,4,5-trisphosphate receptor: a possible mechanism for agonist-specific calcium oscillations in pancreatic acinar cells. J. Gen. Physiol. 113, 851–872.
Li, Y., Rinzel, J., 1994. Equations for IP3R-mediated [Ca2+]i oscillations derived from a detailed kinetic model: a Hodgkin-Huxley like formalism. J. Theor. Biol. 166, 461–473.
Mak, D., Foskett, J., 1997. Single-channel kinetics, inactivation, and spatial distribution of inositol trisphosphate (IP3) receptors in Xenopus oocyte nucleus. J. Gen. Physiol. 109, 571–587.
Mak, D., McBride, S., Raghuram, V., Yue, Y., Joseph, S., Foskett, J., 2000. Single-channel properties in endoplasmic reticulum membrane of recombinant type 3 inositol trisphosphate receptor. J. Gen. Physiol. 115, 241–256.
Maranto, A., 1994. Primary structure, ligand binding, and localization of the human type 3 inositol 1,4,5-trisphosphate receptor expressed in intestinal epithelium. J. Biol. Chem. 269, 1222–1230.
Marchant, J., Parker, I., 1998. Kinetics of elementary Ca2+ puffs evoked in Xenopus oocytes by different Ins(1,4,5)P3 receptor agonists. Biochem. J. 334, 505–509.
Marx, S., Gaburjakova, J., Gaburjakova, M., Henrikson, C., Ondrias, K., Marks, A., 2001. Coupled gating between cardiac calcium release channels (ryanodine receptors). Circ. Res. 88, 1151–1158.
Mazzanti, M., DeFelice, L., Liu, Y., 1991. Gating of L-type Ca2+ channels in embryonic chick ventricle cells: dependence on voltage, current and channel density. J. Physiol. 443, 307–334.
Meyer, C.D., 1989. Stochastic complementation, uncoupling Markov chains, and the theory of nearly reducible systems. SIAM Rev. 31, 240–272.
Moraru, I., Kaftan, E., Ehrlich, B., Watras, J., 1999. Regulation of type 1 inositol 1,4,5-trisphosphate-gated calcium channels by InsP3 and Ca2+: simulation of single channel kinetics based on ligand binding and electrophysiological analysis. J. Gen. Physiol. 113, 837–849.
Naraghi, M., Neher, E., 1997. Linearized buffered Ca2+ diffusion in microdomains and its implications for calculation of [Ca2+] at the mouth of a Ca2+ channel. J. Neurosci. 17, 6961.
Neher, E., 1986. Concentration profiles of intracellular Ca2+ in the presence of diffusible chelator. Exp. Brain Res. 14, 80–96.
Neher, E., 1998. Usefulness and limitations of linear approximations to the understanding of Ca2+ signals. Cell Calcium 24, 345.
Nicola, V., 1998. Lumping in Markov reward processes. Technical Report, RC14719, IBM Thomas Watson Research Centre, P.O. Box 704, Yorktown Heights, NY 10598.
Niggli, E., 1999. Localized intracellular Ca2+ signaling in muscle: Ca2+ sparks and Ca2+ quarks. Annu. Rev. Physiol. 61, 311–335.
Norris, J., 1997. Markov Chains. Cambridge University Press, Cambridge.
Parker, I., Choi, J., Yao, Y., 1996. Elementary events of IP3-induced Ca2+ liberation in Xenopus oocytes: hot spots, puffs and blips. Cell Calcium 20, 105–121.
Plateau, B., Atif, K., 1991. Stochastic automata network for modeling parallel systems. IEEE Trans. Softw. Eng. 17, 1093–1108.
Plateau, B., Stewart, W., 2000. Stochastic automata networks. In: Grassmann, W. (Ed.), Computational Probability. Kluwer Academic Publishers, Boston, pp. 113–151.
Ramos-Franco, J., Fill, M., Mignery, G., 1998. Isoform-specific function of single inositol 1,4,5-trisphosphate receptor channels. Biophys. J. 75, 834–839.
Rengifo, J., Rosales, R., Gonzalez, A., Cheng, H., Stern, M., Rios, E., 2002. Intracellular Ca2+ release as irreversible Markov process. Biophys. J. 83, 2511–2521.
Rios, E., Stern, M., 1997. Ca2+ in close quarters: microdomain feedback in excitation-contraction coupling and other cell biological phenomena. Annu. Rev. Biophys. Biomol. Struct. 26, 47–82.
Sala, F., Hernandez-Cruz, A., 1990. Ca2+ diffusion modeling in a spherical neuron. Relevance of buffering properties. Biophys. J. 57, 313–324.
Sham, J., Song, L., Chen, Y., Deng, L., Stern, M., Lakatta, E., Cheng, H., 1998. Termination of Ca2+ release by a local inactivation of ryanodine receptors in cardiac myocytes. Proc. Natl. Acad. Sci. USA 95, 15096–15101.
Sherman, A., Keizer, J., Rinzel, J., 1990. Domain model for Ca2+-inactivation of Ca2+ channels at low channel density. Biophys. J. 58, 985–995.
Shuai, J., Jung, P., 2002. Stochastic properties of Ca2+ release of inositol 1,4,5-trisphosphate receptor clusters. Biophys. J. 83, 87–97.
Shuai, J., Jung, P., 2003a. Optimal ion channel clustering for intracellular calcium signaling. Proc. Natl. Acad. Sci. USA 100, 506–510.
Shuai, J., Jung, P., 2003b. Selection of intracellular calcium patterns in a model with clustered Ca2+ release channels. Phys. Rev. E. Stat. Nonlin. Soft Matter Phys. 67, 031905.
Smith, G., 1996. Analytical steady-state solution to the rapid buffering approximation near an open Ca2+ channel. Biophys. J. 71, 3064–3072.
Smith, G., 2002a. An extended DeYoung-Keizer-like IP3 receptor model that accounts for domain Ca2+-mediated inactivation. In: Condat, C., Baruzzi, A. (Eds.), Recent Research Developments in Biophysical Chemistry II. Research Signpost, pp. 37–55.
Smith, G., 2002b. Modeling the stochastic gating of ion channels. In: Fall, C., Marland, E., Wagner, J., Tyson, J. (Eds.), Computational Cell Biology. Springer, pp. 291–325.
Smith, G., Dai, L., Muira, R., Sherman, A., 2001. Asymptotic analysis of equations for the buffered diffusion of intracellular Ca2+. SIAM J. Appl. Math. 61, 1816–1838.
Smith, G., Keizer, J., 1998. Spark-to-wave transition: saltatory transmission of Ca2+ waves in cardiac myocytes. Biophys. Chem. 72, 87–100.
Sobie, E., Dilly, K., dos Santos Cruz, J., Lederer, W., Jafri, M., 2002. Termination of cardiac Ca2+ sparks: an investigative mathematical model of calcium-induced calcium release. Biophys. J. 83, 59–78.
Steeb, W.-H., 1997. Matrix Calculus and Kronecker Product with Applications and C++ Programs. World Scientific.
Stern, M., Song, L., Cheng, H., Sham, J., Yang, H., Boheler, K., Rios, E., 1999. Local control models of cardiac excitation-contraction coupling. A possible role for allosteric interactions between ryanodine receptors. J. Gen. Physiol. 113, 469–489.
Stewart, W., 1994. Introduction to the Numerical Solution of Markov Chains. Princeton University Press.
Sun, X., Callamaras, N., Marchant, J., Parker, I., 1998. A continuum of IP3-mediated elementary Ca2+ signalling events in Xenopus oocytes. J. Physiol. 509, 67–80.
Swillens, S., Champeil, P., Combettes, L., Dupont, G., 1998. Stochastic simulation of a single inositol 1,4,5-trisphosphate-sensitive Ca2+ channel reveals repetitive openings during ‘blip-like’ Ca2+ transients. Cell Calcium 23, 291–302.
Swillens, S., Dupont, G., Combettes, L., Champeil, P., 1999. From Ca2+ blips to Ca2+ puffs: theoretical analysis of the requirements for interchannel communication. Proc. Natl. Acad. Sci. USA 96, 13750–13755.
Tang, Y., Othmer, H., 1994. A model of calcium dynamics in cardiac myocytes based on the kinetics of ryanodine-sensitive calcium channels. Biophys. J. 67, 2223–2235.
Tang, Y., Stephenson, J., Othmer, H., 1995. Simplification and analysis of models of Ca2+ dynamics based on IP3-sensitive Ca2+ channel kinetics. Biophys. J. 70, 246–263.
Wagner, J., Keizer, J., 1994. Effects of rapid buffers on Ca2+ diffusion and Ca2+ oscillations. Biophys. J. 67, 447–456.
Wang, S., Song, L., Xu, L., Meissner, G., Lakatta, E., Rios, E., Stern, M., Cheng, H., 2002. Thermodynamically irreversible gating of ryanodine receptors in situ revealed by stereotyped duration of release in Ca2+ sparks. Biophys. J. 83, 242–251.
White, J., Rubinstein, J., Kay, A., 2000. Channel noise in neurons. Trends Neurosci. 23, 131–137.
Yao, Y., Choi, J., Parker, I., 1995. Quantal puffs of intracellular Ca2+ evoked by inositol trisphosphate in Xenopus oocytes. J. Physiol. 482, 533–553.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nguyen, V., Mathias, R. & Smith, G.D. A stochastic automata network descriptor for Markov chain models of instantaneously coupled intracellular Ca2+ channels. Bull. Math. Biol. 67, 393–432 (2005). https://doi.org/10.1016/j.bulm.2004.08.010
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/j.bulm.2004.08.010