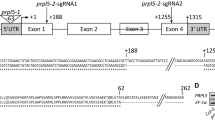
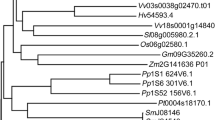
Abstract
The defective chloroplast and leaf-mutable (dcl-m) mutation of tomato blocks chloroplast differentiation in leaf mesophyll cells and a signaling system that appears to be required for morphogenesis of palisade cells during leaf growth. To dissect the function of DCL, mutants with stable dcl alleles (dcl-s) were generated and examined for their phenotype. DCL/dcl-s plant produce dcl-s/dcl-s seeds with embryos arrested at the globular stage of development. The levels of several chloroplast- and nuclear-encoded proteins are strongly reduced in dcl-m mutant leaf sectors without significant changes in their corresponding mRNAs. The 4.5S rRNA fails to be processed efficiently, however, suggesting that DCL has a direct or indirect function in rRNA processing or correct ribosome assembly. Accordingly, chloroplasts in dcl-m sectors are impaired in polysome assembly, which can explain the reduced accumulation of chloroplast-encoded proteins. These results suggest that DCL is required for chloroplast rRNA processing, and emphasize the importance of plastid function during embryogenesis.
Article PDF
Similar content being viewed by others
References
Albert, S., Despres, B., Guilleminot, J., Bechtold, N., Pelletier, G., Delseny, M. and Devic, M. 1999. The EMB 506 gene encodes a novel ankyrin repeat containing protein that is essential for the normal development of Arabidopsis embryos. Plant J. 17: 169–179.
Aluru, M.R., Bae, B., Wu, D. and Rodermel, S.R. 2001. The Arabidopsis immutans mutation affects plastid differentiation and the morphogenesis of white and green sectors in variegated plants. Plant Physiol. 127: 67–77.
Apuya, N.R., Yadegari, R., Fischer, R.L., Harada, J.J., Zimmerman, J.L. and Goldberg, R.B. 2001. The Arabidopsis embryo mutant schlepperless has a defect in the chaperonin-60α gene. Plant Physiol. 126: 717–730.
Arabidopsis Genome Initiative. 2000. Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408: 796–815.
Arimura, S.I., Hirai, A. and Tsutsumi, N. 2001. Numerous and highly developed tubular projections from plastids observed in tobacco epidermal cells. Plant Sci. 160: 449–454.
Ausubel, M., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A. and Struhl, K. 1990. Current Protocols in Molecular Biology. Wiley, New York.
Babiychuk, E., Fuangthong, M., Van Montagu, M., Inzé, D. and Kushnir, S. 1997. Efficient gene tagging in Arabidopsis thaliana using a gene trap approach. Proc. Natl. Acad. Sci. USA 94: 12722–12727.
Barkan, A. 1993. Nuclear mutants of maize with defects in chloroplast polysome assembly have altered chloroplast RNA metabolism. Plant Cell 5: 389–402.
Barkan, A. 1998. Approaches to investigating nuclear genes that function in chloroplast biogenesis in land plants. Meth. Enzymol. 297: 38–57.
Bisanz, C., Begot, L., Carol, P., Perez, P., Bligny, M., Pesey, H., Gallois, J.L., Lerbs-Mache, S. and Mache, R. 2003. The Arabidopsis nuclear DAL gene encodes a chloroplast protein which is required for the maturation of the plastid ribosomal RNAs and is essential for chloroplast differentiation. Plant Mol. Biol. 51: 651–663.
Bonhomme, S., Budar, F., Férault, M. and Pelletier, G. 1991. A NcoI fragment of Ogura radish mitochondrial DNA is correlated with cytoplasmic male-sterility in Brassica cybrids. Curr. Genet. 19: 121–127.
Bonnema, A.B., Castillo, C., Reiter, N., Cunningham, M., Adams, H.P. and O'Connell, M. 1995. Molecular and ultrastructural analysis of a nonchromosomal variegated mutant. Tomato mitochondrial mutants that cause abnormal leaf development. Plant Physiol. 109: 385–392.
Bonnett, H.T., Djurberg, I., Fajardo, M. and Glimelius, K. 1993. A mutation causing variegation and abnormal development in tobacco is associated with an altered mitochondrial DNA. Plant J. 3: 519–525.
Chatterjee, M., Sparvoli, S., Edmunds, C., Garosi, P., Findlay, K. and Martin, C. 1996. DAG, a gene required for chloroplast differentiation and palisade development in Antirrhinum majus.EMBO J. 15: 4194–4207.
Despres, B., Delseny, M. and Devic, M. 2001. Partial complementation of embryo defective mutations: a general strategy to elucidate gene function. Plant J. 27: 149–159.
Edwards, K., Bedbrook, J., Dyer, T. and Kössel, H. 1981. 4.5S rRNA from Zea mays chloroplasts shows structural homology with the 3′ end of prokaryotic 23S rRNA. Biochem. Int. 2: 533–538.
Grelon, M., Budar, F., Bonhomme, S. and Pelletier, G. 1994. Ogura cytolplasmique male-sterility (CMS)-associated orf138 is translated into a mitochondrial membrane polypetide in male-sterile Brassica cybrids. Mol. Gen. Genet. 243: 540–547.
Keddie, J.S., Carroll, B., Jones, J.D. and Gruissem, W. 1996. The DCL gene of tomato is required for chloroplast development and palisade cell morphogenesis in leaves. EMBO J. 15: 4208–4217.
Keus, R.J.A., Dekker, A.F., Kreuk, K.C.J. and Groot, G.S.P. 1984. Transcription of ribosomal DNA in chloroplast of Spirodela oligorhiza. Curr. Genet. 9: 91–97.
Klimyuk, V.I., Carroll, B.J., Thomas, C.M. and Jones, J.D. 1993. Alkali treatment for rapid preparation of plant material for reliable PCR analysis. Plant J. 3: 493–494.
Kohler, R.H. and Hanson, M.R. 2000. Plastid tubules of higher plants are tissue-specific and developmentally regulated. J. Cell Sci. 113: 81–89.
Kössel, H., Edwards, K., Koch, W., Langridge, P., Schiefermayr, E., Schwarz, Z., Strittmatter, G. and Zenke G. 1982. Structural and functional analysis of an rRNA operon and its flanking tRNA genes from Zea mays chloroplasts. Nucl. Acids Res. Symp. Series 11: 117–120.
Kössel, H., Natt, E., Strittmatter, G., Fritzsche, E., Gozdzicka-Jozefiak, A. and Przybyl, D. 1985. Structure and expression of rRNA operons from plastids of higher plants. In: L. van Vloten-Doting, G.S.P. Groot and T.C. Hall (Eds.) Molecular Form and Function of the Plant Genome, Plenum, New York, pp. 183–198.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Leal-Klevezas, D.S., Martinez-Soriano, J.P. and Nazar, R.N. 2000. Transcription and processing map of the 4.5-5S rRNA intergenic regions (ITS3) from rapeseed (Brassica napus) chloroplasts. Plant Cell Rep. 19: 667–673.
Li, H., Culligan, K., Dixon, R.A. and Chory, J. 1995. CUE1: a mesophyll cell-specific positive regulator of light-controlled gene expression in Arabidopsis. Plant Cell 7: 1599–1610.
Mandel, M.A., Feldmann, K.A., Herrera-Estrella, L., Rocha-Sosa, M. and Leon, P. 1996. CLA1, a novel gene required for chloroplast development, is highly conserved in evolution. Plant J. 9: 649–658.
Martin, W, Stoebe, B, Goremykin, V, Hapsmann, S, Hasegawa, M. and Kowallik, K.V. 1998. Gene transfer to the nucleus and the evolution of chloroplasts. Nature 393: 162–165.
Meurer, J., Grevelding, C., Westhoff, P. and Reiss, B. 1998. The PAC protein affects the maturation of specific chloroplast mRNAs in Arabidopsis thaliana. Mol. Gen. Genet. 258: 342–351.
Mochizuki, N., Brusslan, J.A., Larkin, R., Nagatani, A. and Chory, J. 2001. Arabidopsis genomes uncoupled 5 (GUN5) mutant reveals the involvement of Mg-chelatase H subunit in plastidto-nucleus signal transduction. Proc. Natl. Acad. Sci. USA 98: 2053–2058.
Møller, S.G., Kunkel, T. and Chua, N.H. 2001. A plastidic ABC protein involved in intercompartmental communication of light signaling. Genes Dev. 15: 90–103.
Oelmüller, R., Levitan, I., Bergfeld, R., Rajasekhar, V.K. and Mohr, H. 1986. Expression of nuclear genes are affected by treatments acting on the plastids. Planta 168: 482–492.
Reiter, R.S., Coomber, S.A., Bourett, T.M., Bartley, G.E. and Scolnik, P.A. 1994. Control of leaf and chloroplast development by the Arabidopsis gene pale cress. Plant Cell 6: 1253–1264.
Rodriguez-Concepcion, M., Yalovsky, S., Zik, M., Fromm, H. and Gruissem, W. 1999. The prenylation status of a novel plant calmodulin directs plasma membrane or nuclear localization of the protein. EMBO J. 18: 1996–2007.
Rujan, T. and Martin, W. 2001. How many genes in Arabidopsis come from cyanobacteria? An estimate from 386 protein phylogenies. Trends Genet. 17: 113–120.
Sato, S, Nakamura, Y, Kaneko, T, Asamizu, E. and Tabata, S. 1999. Complete structure of the chloroplast genome of Arabidopsis thaliana. DNA Res. 6: 283–290.
Scott, A., Wyatt, S., Tsou, P.L., Robertson, D. and Allen, N.S. 1999. Model system for plant cell biology: GFP imaging in living onion epidermal cells. Biotechniques 26: 1125–1132.
Srivastava, A.K. and Schlessinger, D. 1990. Mechanism and regulation of bacterial ribosomal RNA processing. Annu. Rev. Microbiol. 44: 105–129.
Streatfield, S.J., Weber, A., Kinsman, E.A., Hausler, R.E., Li, J., Post-Beittenmiller, D., Kaiser, W.M., Pyke, K.A., Flugge, U.I. and Chory, J. 1999. The phosphoenolpyruvate/phosphate translocator is required for phenolic metabolism, palisade cell development, and plastid-dependent nuclear gene expression. Plant Cell 11: 1609–1622.
Strittmatter, G. and Kössel, H. 1984. Cotranscription and processing of 23S, 4.5S and 5S rRNA in chloroplasts from Zea mays. Nucl. Acids Res. 12: 7633–7647.
Thomas, C.M., Jones, D.A., English, J.J., Carroll, B.J., Bennetzen, J.L., Harrison, K., Burbidge, A., Bishop, G.J. and Jones, J.D. 1994. Analysis of the chromosomal distribution of transposoncarrying T-DNAs in tomato using the inverse polymerase chain reaction. Mol. Gen. Genet. 242: 573–585.
Tsugeki, R., Kochieva, E.Z. and Fedoroff, N.V. 1996. A transposon insertion in the Arabidopsis SSR16 gene causes an embryodefective lethal mutation. Plant J. 10: 479–489.
Uwer, U., Willmitzer, L. and Altmann, T. 1998. Inactivation of a glycyl-tRNA synthetase leads to an arrest in plant embryo development. Plant Cell 10: 1277–1294.
Wang, Y., Duby, G., Purnelle, B. and Boutry, M. 2000. Tobacco VDL gene encodes a plastid DEAD box RNA helicase and is involved in chloroplast differentiation and plant morphogenesis. Plant Cell 12: 2129–2142.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bellaoui, M., Keddie, J.S. & Gruissem, W. DCL is a plant-specific protein required for plastid ribosomal RNA processing and embryo development. Plant Mol Biol 53, 531–543 (2003). https://doi.org/10.1023/B:PLAN.0000019061.79773.06
Published:
Issue Date:
DOI: https://doi.org/10.1023/B:PLAN.0000019061.79773.06