Abstract
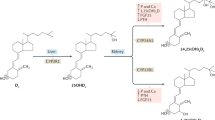
Synthesis of vitamin D in the skin in response to ultraviolet light is the main determinant of vitamin D status in man1 and it is therefore surprising that rickets and osteomalacia, clinical signs of vitamin D deficiency, remain common in tropical and subtropical countries2. Skin pigmentation can reduce vitamin D formation3 but this is a negligible limitation in people exposed to abundant ultraviolet light4,5. Earlier studies in animals6 and man7 suggested that another environmental factor, the low calcium/high cereal diet typical of susceptible populations2, might affect the efficiency of vitamin D utilization. We show here in rats that the rate of inactivation of vitamin D in the liver is increased by calcium deprivation. The effect is mediated by 1,25-dihydroxyvitamin D, produced in response to secondary hyperparathyroidism8,9, which promotes hepatic conversion of vitamin D to polar inactivation products that are excreted in bile. This finding has widespread implications both for understanding the pathogenesis of endemic rickets and in that it provides a unifying mechanism for the development of vitamin D deficiency in many clinical disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Haddad, J. G. & Hahn, T. J. Nature 244, 515–517 (1973).
DHSS Report on Health and Social Subjects, 19 (HMSO, London, 1980).
Clemens, T. L., Adams, I. S., Henderson, S. L. & Holick, M. F. Lancet i, 74–76 (1982).
Stamp, T. C. B. Proc Nutr. Soc. 34, 119–130 (1975).
Davie, M. & Lawson, D. E. M. Clin. Sci. 58, 235–242 (1980).
Ewer, T. K. Nature 166, 732–733 (1950).
Robertson, I, Ford, J. A., McIntosh, W. B. & Dunnigan, M. G. Br. J. Nutr. 45, 17–22 (1981).
Garabedian, M., Holick, M. F., DeLuca, H. F. & Boyle, I. T. Proc natn. Acad. Sci. U.S.A. 69, 1673–1676 (1972).
Fraser, D. R. & Kodicek, E. Nature new Biol. 241, 163–166 (1973).
Gray, R. W., Weber, H. P., Dominguez, J. H. & Lemann, J. J. clin. Endocr. Metab. 39, 1045–1056 (1974).
Ribovich, M. L. & DeLuca, H. F. Archs Biochem. Biophys. 188, 145–156 (1978).
Fox, J. & Ross, R. J. Endocr. 105, 169–173 (1985).
Paulson, S. K. & Kenny, A. D. Biopharmaceutics and Drug Disposition 6, 359–372 (1985).
Lawson, D. E. M., Wilson, P. W. & Kodicek, E. Nature 222, 171–172 (1969).
Bell, P. A. & Kodicek, E. Biochem. J. 116, 755–757 (1970).
Anderson, G. H. & Draper, H. H. J. Nutr. 102, 1123–1132 (1972).
Siu, G. M. & Draper, H. H. Calc Tiss. Int. 33, 667–672 (1981).
Charbonne, G. A. & Hulstaert, P. E. Endocrinology 95, 621–626 (1974).
Groszmann, R. G., Vorobioff, J. & Riley, E. Am. J. Phystol. 242, G156–G160 (1982).
Ketterer, S. G., Wiegand, B. D. & Rapaport, E. Am. J. Physiol. 199, 481–484 (1960).
Bell, P. A. & Kodicek, E. Biochem. J. 115, 663–669 (1969).
Hahn, T. J., Hendin, B. A., Scharp, C. R. & Haddad; J. G. New Engl. J. Med. 287, 900–904 (1972).
Omura, T. & Sato, R. J. biol. Chem. 239, 2370–2378 (1964).
Braidman, I. P. & Anderson, D. C. Clin. Endocr. 23, 445–460 (1985).
Baran, D. T. & Milne, M. L. J. clin. Invest. 77, 1622–1626 (1986).
Sikorska, M. & Whitfield, J. F. Biochem. biophys. Res. Commun. 129, 766–772 (1985).
Woodhouse, N. J. Y., Doyle, F. H. & Joplin, G. H. Lancet ii, 283–287 (1971).
Stanbury, S. W. J. R. Coll. Phys. Lond. 15, 205–217 (1981).
Driscoll, R. H., Meredith, S. C., Sitrin, M. & Rosenberg, I. H. Gastroenterology 83, 1252–1258 (1982).
Dibble, J. B., Sheridan, P. & Losowsky, M. S. Q. Jl Med. 209, 119–134 (1984).
Morgan, S. B., Hunt, G. & Patterson, C. R. Q. Jl Med. 39, 395–410 (1970).
Teitelbaum, S. L., Halverson, J. D., Bates, M., Wise, L. & Haddad, J. G. Ann. intern. Med. 86, 289–293 (1977).
Dibble, J. B., Sheridan, P., Hampshire, R., Hardy, G. J. & Losowsky, M. S. Clin. Endocr. 15, 373–383 (1981).
Bell, N. H., Epstein, S., Greend, A., Shary, J., Oexmann, M. J. & Shaw, S. J. clin. Invest. 76, 370–373 (1985).
DeLuca, H. F., Guroff, E., Steenbock, H., Reiser, F. & Mannatt, M. R. J. Nutr. 75, 175–180 (1961).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Clements, M., Johnson, L. & Fraser, D. A new mechanism for induced vitamin D deficiency in calcium deprivation . Nature 325, 62–65 (1987). https://doi.org/10.1038/325062a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/325062a0
This article is cited by
-
Predicting the acute-phase response fever risk in bisphosphonate-naive osteoporotic patients receiving their first dose of zoledronate
Osteoporosis International (2022)
-
Increased calcium intake is associated lower serum 25-hydroxyvitamin D levels in subjects with adequate vitamin D intake: a population-based observational study
BMC Nutrition (2020)
-
Vitamin D status among the elderly Chinese population: a cross-sectional analysis of the 2010–2013 China national nutrition and health survey (CNNHS)
Nutrition Journal (2017)
-
Letter to the Editor on “Vitamin D-binding protein, vitamin D status and serum bioavailable 25(OH)D of young Asian Indian males working in outdoor and indoor environments”
Journal of Bone and Mineral Metabolism (2017)
-
Vitamin D expenditure is not altered in pregnancy and lactation despite changes in vitamin D metabolite concentrations
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.