Abstract
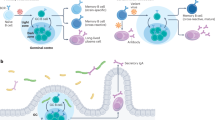
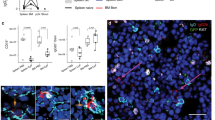
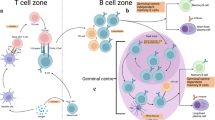
Immunological memory in the antibody system is generated in T-cell-dependent responses and carried by long-lived memory B cells that recognize antigen by high-affinity antibodies1,2. But it remains controversial1 whether these B cells represent true ‘memory’ cells (that is, their maintenance is independent of the immunizing antigen), or whether they are a product of a chronic immune response driven by the immunizing antigen, which can be retained in the organism for extended time periods on the surface of specialized antigen-presenting cells (follicular dendritic cells)3. Cell transfer experiments provided evidence in favour of a role of the immunizing antigen4,5; however, analysis of memory cells in intact animals, which showed that these cells are mostly resting6 and can persist in the absence of detectable T-cell help7 or follicular dendritic cells8, argued against it. Here we show, by using a genetic switch mediated by Cre recombinase, that memory B cells switching their antibody specificity away from the immunizing antigen are indeed maintained in the animal over long periods of time, similar to cells retaining their original antigen-binding specificity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sprent, J. T and B memory cells. Cell 76, 315– 322 (1994).
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272 , 54–60 (1996).
Mandel, T. E., Phipps, R. P., Abbot, A. & Tew, J. G. The follicular dendritic cell: long term antigen retention during immunity. Immunol. Rev. 53, 29–59 ( 1980).
Askonas, B. A., Cunningham, A. J., Kreth, H. W., Roelants, G. E. & Williamson, A. R. Amplification of B cell clones forming antibody to the 2,4-dinitrophenyl group. Eur. J. Immunol. 2, 494–498 (1972).
Gray, D. & Skarvall, H. B-cell memory is short-lived in the absence of antigen. Nature 336, 70– 73 (1988).
Schittek, B. & Rajewsky, K. Maintenance of B-cell memory by long-lived cells generated from proliferating precursors. Nature 346, 749–751 ( 1990).
Vieira, P. & Rajewsky, K. Persistence of memory B cells in mice deprived of T cell help. Int. Immunol. 2, 487–494 (1990).
Karrer, U. et al. Antiviral B cell memory in the absence of mature follicular dendritic cell networks and classical germinal centers in TNFR1-/- mice. J. Immunol. 164, 768–778 (2000).
Lam, K. P. & Rajewsky, K. Rapid elimination of mature autoreactive B cells demonstrated by Cre- induced change in B cell antigen receptor specificity in vivo. Proc. Natl Acad. Sci. USA 95, 13171–13175 (1998).
Lam, K. P., Kühn, R. & Rajewsky, K. In vivo ablation of surface immunoglobulin on mature B cells by inducible gene targeting results in rapid cell death. Cell 90, 1073–1083 ( 1997).
Hayakawa, K., Ishii, R., Yamasaki, K., Kishimoto, T. & Hardy, R. R. Isolation of high-affinity memory B cells: phycoerythrin as a probe for antigen-binding cells. Proc. Natl Acad. Sci. USA 84, 1379–1383 ( 1987).
Hoess, R. H., Wierzbicki, A. & Abremski, K. The role of the loxP spacer region in P1 site-specific recombination. Nucleic Acids Res. 14, 2287 –2300 (1986).
Kühn, R., Schwenk, F., Aguet, M. & Rajewsky, K. Inducible gene targeting in mice. Science 269, 1427– 1429 (1995).
Okumura, K., Julius, M. H., Tsu, T., Herzenberg, L. A. & Herzenberg, L. A. Demonstration that IgG memory is carried by IgG-bearing cells. Eur. J. Immunol. 6, 467– 472 (1976).
Esser, C. & Radbruch, A. Immunoglobulin class switching: molecular and cellular analysis. Annu. Rev. Immunol. 8, 717–735 (1990).
MacLennan, I. C. M. Germinal centers. Annu. Rev. Immunol. 12, 117–139 (1994).
Rajewsky, K. Clonal selection and learning in the antibody system. Nature 381, 751–758 (1996).
Neuberger, M. S. et al. Monitoring and interpreting the intrinsic features of somatic hypermutation. Immunol. Rev. 162, 107– 116 (1998).
Nemazee, D. Antigen receptor ‘capacity’ and the sensitivity of self-tolerance. Immunol. Today 17, 25– 29 (1996).
Manz, R. A., Thiel, A. & Radbruch, A. Lifetime of plasma cells in the bone marrow. Nature 388, 133–134 ( 1997).
McHeyzer-Williams, M. G. & Ahmed, R. B cell memory and the long-lived plasma cell. Curr. Opin. Immunol. 11 , 172–179 (1999).
Bruno, L., Kirberg, J. & von Boehmer, H. On the cellular basis of immunological T cell memory. Immunity 2, 37–43 (1995).
Murali-Krishna, K. et al. Persistence of memory CD8 T cells in MHC class I-deficient mice. Science 286, 1377– 1381 (1999).
Swain, S. L., Hu, H. & Huston, G. Class II-independent generation of CD4 memory T cells from effectors. Science 286, 1381—1383 ( 1999).
Zou, Y. R., Takeda, S. & Rajewsky, K. Gene targeting in the Ig κ locus: efficient generation of λ chain-expressing B cells, independent of gene rearrangements in Ig κ. EMBO J. 12, 811– 820 (1993).
Torres, R. M. & Kühn, R. Laboratory Protocols for Conditional Gene Targeting (Oxford Univ. Press, Oxford, 1997).
Schwenk, F., Baron, U. & Rajewsky, K. A cre-transgenic mouse strain for the ubiquitous deletion of loxP- flanked gene segments including deletion in germ cells. Nucleic Acids Res. 23, 5080– 5081 (1995).
Kaisho, T., Schwenk, F. & Rajewsky, K. The roles of γ1 heavy chain membrane expression and cytoplasmic tail in IgG1 responses. Science 276 , 412–415 (1997).
Ehlich, A., Martin, V., Müller, W. & Rajewsky, K. Analysis of the B-cell progenitor compartment at the level of single cells. Curr. Biol. 4, 573–583 (1994).
Cumano, A. & Rajewsky, K. Structure of primary anti-(4-hydroxy-3-nitrophenyl)acetyl (NP) antibodies in normal and idiotypically suppressed C57BL/6 mice. Eur. J. Immunol. 15, 512–520 (1985).
Acknowledgements
We are grateful to B. Hampel, A. Roth and C. Göttlinger for expert technical help, and to T. Kaisho, S. Schwers, L. Pao, A. Ehlich, and R. Küppers for discussion. We also thank S. Casola, P. Oberdoerffer and W. Müller for comments on the manuscript and G. Schmall for help in its preparation. This work was supported by the Volkswagen Foundation, the European Union and the Deutsche Forschungsgemeinschaft. M.M. was recipient of a fellowship from the Alexander von Humboldt Foundation, and K.-P.L. was a fellow of the Human Frontier Science Organization.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maruyama, M., Lam, KP. & Rajewsky, K. Memory B-cell persistence is independent of persisting immunizing antigen . Nature 407, 636–642 (2000). https://doi.org/10.1038/35036600
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35036600
This article is cited by
-
On the Dynamics of Matrix Models for Immune Clonal Networks
Journal of Mathematical Sciences (2022)
-
Memory B cells are reactivated in subcapsular proliferative foci of lymph nodes
Nature Communications (2018)
-
Interconnected subsets of memory follicular helper T cells have different effector functions
Nature Communications (2017)
-
Memory B cells
Nature Reviews Immunology (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.