Abstract
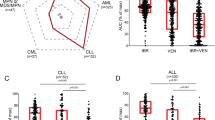
Although the BTK inhibitor ibrutinib has transformed the management of patients with chronic lymphocytic leukemia (CLL), it does not induce substantial apoptosis in vitro, and as such the mechanisms underlying its ability to kill CLL cells are not well understood. Acalabrutinib, a more specific BTK inhibitor now in development, also appears to be highly effective in CLL, but the connection of its mechanism with CLL cell death is also unclear. Using dynamic BH3 profiling, we analyzed alterations in the function of the mitochondrial apoptotic pathway induced by ibrutinib and acalabrutinib. We studied CLL patient samples treated ex vivo with both drugs, as well as primary samples from CLL patients on clinical trials of both drugs. We found that BTK inhibition enhances mitochondrial BCL-2 dependence without significantly altering overall mitochondrial priming. Enhancement of BCL-2 dependence was accompanied by an increase in the pro-apoptotic protein BIM. In contrast, treatment with the selective BCL-2 inhibitor venetoclax enhanced overall mitochondrial priming without increasing BCL-2 dependence. Pre-treatment of CLL cells with either BTK inhibitor, whether ex vivo or in vivo in patients, enhanced killing by venetoclax. Our data suggest that BTK inhibition enhances mitochondrial BCL-2 dependence, supporting the ongoing development of clinical trials combining BTK and BCL-2 inhibition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Wiestner A . BCR pathway inhibition as therapy for chronic lymphocytic leukemia and lymphoplasmacytic lymphoma. Hematology Am Soc Hematol Educ Program 2014; 2014: 125–134.
Davids MS, Brown JR . Ibrutinib: a first in class covalent inhibitor of Bruton's tyrosine kinase. Future Oncol 2014; 10: 957–967.
Byrd JC, Furman RR, Coutre SE, Flinn IW, Burger JA, Blum KA et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N Engl J Med 2013; 369: 32–42.
Ponader S, Chen SS, Buggy JJ, Balakrishnan K, Gandhi V, Wierda WG et al. The Bruton tyrosine kinase inhibitor PCI-32765 thwarts chronic lymphocytic leukemia cell survival and tissue homing in vitro and in vivo. Blood 2012; 119: 1182–1189.
Burger JA, Styles L, Kipps TJ . Ibrutinib for chronic lymphocytic leukemia. N Engl J Med 2016; 374: 1594–1595.
O'Brien S, Furman RR, Coutre SE, Sharman JP, Burger JA, Blum KA et al. Ibrutinib as initial therapy for elderly patients with chronic lymphocytic leukaemia or small lymphocytic lymphoma: an open-label, multicentre, phase 1b/2 trial. Lancet Oncol 2014; 15: 48–58.
Byrd JC, Furman RR, Coutre SE, Burger JA, Blum KA, Coleman M et al. Three-year follow-up of treatment-naive and previously treated patients with CLL and SLL receiving single-agent ibrutinib. Blood 2015; 125: 2497–2506.
Byrd JC, Harrington B, O'Brien S, Jones JA, Schuh A, Devereux S et al. Acalabrutinib (ACP-196) in relapsed chronic lymphocytic leukemia. N Engl J Med 2016; 374: 323–332.
Del Gaizo Moore V, Brown JR, Certo M, Love TM, Novina CD, Letai A . Chronic lymphocytic leukemia requires BCL2 to sequester prodeath BIM, explaining sensitivity to BCL2 antagonist ABT-737. J Clin Invest 2007; 117: 112–121.
Souers AJ, Leverson JD, Boghaert ER, Ackler SL, Catron ND, Chen J et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med 2013; 19: 202–208.
Roberts AW, Davids MS, Pagel JM, Kahl BS, Puvvada SD, Gerecitano JF et al. Targeting BCL2 with venetoclax in relapsed chronic lymphocytic leukemia. N Engl J Med 2016; 374: 311–322.
Anderson MA, Deng J, Seymour JF, Tam C, Kim SY, Fein J et al. The BCL2 selective inhibitor venetoclax induces rapid onset apoptosis of CLL cells in patients via a TP53-independent mechanism. Blood 2016; 127: 3215–3224.
Stilgenbauer S, Eichhorst B, Schetelig J, Coutre S, Seymour JF, Munir T et al. Venetoclax in relapsed or refractory chronic lymphocytic leukaemia with 17p deletion: a multicentre, open-label, phase 2 study. Lancet Oncol 2016; 17: 768–778.
Cervantes-Gomez F, Lamothe B, Woyach JA, Wierda WG, Keating MJ, Balakrishnan K et al. Pharmacological and protein profiling suggests venetoclax (ABT-199) as optimal partner with ibrutinib in chronic lymphocytic leukemia. Clin Cancer Res 2015; 21: 3705–3715.
Certo M, Del Gaizo Moore V, Nishino M, Wei G, Korsmeyer S, Armstrong SA et al. Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell 2006; 9: 351–365.
Montero J, Sarosiek KA, DeAngelo JD, Maertens O, Ryan J, Ercan D et al. Drug-induced death signaling strategy rapidly predicts cancer response to chemotherapy. Cell 2015; 160: 977–989.
Byrd JC, Brown JR, O'Brien S, Barrientos JC, Kay NE, Reddy NM et al. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N Engl J Med 2014; 371: 213–223.
Davids MS, Deng J, Wiestner A, Lannutti BJ, Wang L, Wu CJ et al. Decreased mitochondrial apoptotic priming underlies stroma-mediated treatment resistance in chronic lymphocytic leukemia. Blood 2012; 120: 3501–3509.
Ryan JA, Brunelle JK, Letai A . Heightened mitochondrial priming is the basis for apoptotic hypersensitivity of CD4+ CD8+ thymocytes. Proc Natl Acad Sci USA 2010; 107: 12895–12900.
Foight GW, Ryan JA, Gulla SV, Letai A, Keating AE . Designed BH3 peptides with high affinity and specificity for targeting Mcl-1 in cells. ACS Chem Biol 2014; 9: 1962–1968.
Wodarz D, Garg N, Komarova NL, Benjamini O, Keating MJ, Wierda WG et al. Kinetics of CLL cells in tissues and blood during therapy with the BTK inhibitor ibrutinib. Blood 2014; 123: 4132–4135.
Thompson PA, O'Brien SM, Wierda WG, Ferrajoli A, Stingo F, Smith SC et al. Complex karyotype is a stronger predictor than del(17p) for an inferior outcome in relapsed or refractory chronic lymphocytic leukemia patients treated with ibrutinib-based regimens. Cancer 2015; 121: 3612–3621.
Woyach JA, Furman RR, Liu TM, Ozer HG, Zapatka M, Ruppert AS et al. Resistance mechanisms for the Bruton's tyrosine kinase inhibitor ibrutinib. N Engl J Med 2014; 370: 2286–2294.
Bojarczuk K, Sasi BK, Gobessi S, Innocenti I, Pozzato G, Laurenti L et al. BCR signaling inhibitors differ in their ability to overcome Mcl-1-mediated resistance of CLL B cells to ABT-199. Blood 2016; 127: 3192–3201.
Thomas LW, Lam C, Edwards SW . Mcl-1; the molecular regulation of protein function. FEBS Lett 2010; 584: 2981–2989.
Davids MS . Boldly Targeting Kinases without mutations. Blood 2014; 123: 1119–1121.
Acknowledgements
We thank the patients who generously donated their samples for this study; John F Daley II and Suzan Lazo-Kallanian for technical support with flow cytometry; Abbvie for providing venetoclax, and Acerta for providing acalabrutinib. This work was funded by the National Institutes of Health (R01CA129974) and a Dana-Farber Gloria Spivak Faculty Advancement Award. MSD is an American Society of Clinical Oncology Career Development Award recipient and a recipient of a National Institutes of Health Loan Repayment Program award.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
JD, EI and SMF have no relevant conflicts of interest. JRB has received consulting fees from Abbvie, Genentech, Pharmacyclics and Janssen. AL is a paid advisor to and his laboratory receives research sponsorship from AbbVie, Astra-Zeneca and Tetralogic. MSD has received research sponsorship from Pharmacyclics and Genentech, and has received consulting fees from Genentech, Abbvie, Pharmacyclics and Janssen.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Deng, J., Isik, E., Fernandes, S. et al. Bruton’s tyrosine kinase inhibition increases BCL-2 dependence and enhances sensitivity to venetoclax in chronic lymphocytic leukemia. Leukemia 31, 2075–2084 (2017). https://doi.org/10.1038/leu.2017.32
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2017.32
This article is cited by
-
Unresolved questions in selection of therapies for treatment-naïve chronic lymphocytic leukemia
Journal of Hematology & Oncology (2023)
-
Ibrutinib sensitizes CLL cells to venetoclax by interrupting TLR9-induced CD40 upregulation and protein translation
Leukemia (2023)
-
A phase 1b study of ibrutinib in combination with obinutuzumab in patients with relapsed or refractory chronic lymphocytic leukemia
Leukemia (2023)
-
Combining BTK inhibitors with BCL2 inhibitors for treating chronic lymphocytic leukemia and mantle cell lymphoma
Biomarker Research (2022)
-
Progress in understanding the mechanisms of resistance to BCL-2 inhibitors
Experimental Hematology & Oncology (2022)