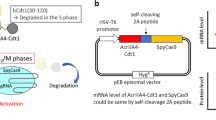
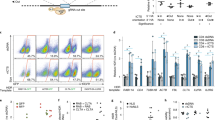
Abstract
CRISPR-Cas-mediated genome editing relies on guide RNAs that direct site-specific DNA cleavage facilitated by the Cas endonuclease. Here we report that chemical alterations to synthesized single guide RNAs (sgRNAs) enhance genome editing efficiency in human primary T cells and CD34+ hematopoietic stem and progenitor cells. Co-delivering chemically modified sgRNAs with Cas9 mRNA or protein is an efficient RNA- or ribonucleoprotein (RNP)-based delivery method for the CRISPR-Cas system, without the toxicity associated with DNA delivery. This approach is a simple and effective way to streamline the development of genome editing with the potential to accelerate a wide array of biotechnological and therapeutic applications of the CRISPR-Cas technology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Jinek, M. et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816–821 (2012).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Cong, L. et al. Multiplex genome engineering using CRISPR-Cas systems. Science 339, 819–823 (2013).
Hsu, P.D., Lander, E.S. & Zhang, F. Development and applications of CRISPR-Cas9 for genome engineering. Cell 157, 1262–1278 (2014).
Hendel, A., Fine, E.J., Bao, G. & Porteus, M.H. Quantifying on- and off-target genome editing. Trends Biotechnol. 33, 132–140 (2015).
Dellinger, D.J. et al. Streamlined process for the chemical synthesis of RNA using 2′-O-thionocarbamate-protected nucleoside phosphoramidites in the solid phase. J. Am. Chem. Soc. 133, 11540–11556 (2011).
Deleavey, G.F. & Damha, M.J. Designing chemically modified oligonucleotides for targeted gene silencing. Chem. Biol. 19, 937–954 (2012).
Eckstein, F. Phosphorothioates, essential components of therapeutic oligonucleotides. Nucleic Acid Ther. 24, 374–387 (2014).
Dellinger, D.J., Sheehan, D.M., Christensen, N.K., Lindberg, J.G. & Caruthers, M.H. Solid-phase chemical synthesis of phosphonoacetate and thiophosphonoacetate oligodeoxynucleotides. J. Am. Chem. Soc. 125, 940–950 (2003).
Hendel, A. et al. Quantifying genome-editing outcomes at endogenous loci with SMRT sequencing. Cell Reports 7, 293–305 (2014).
Cradick, T.J., Fine, E.J., Antico, C.J. & Bao, G. CRISPR-Cas9 systems targeting beta-globin and CCR5 genes have substantial off-target activity. Nucleic Acids Res. 41, 9584–9592 (2013).
Tebas, P. et al. Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV. N. Engl. J. Med. 370, 901–910 (2014).
Lombardo, A. et al. Site-specific integration and tailoring of cassette design for sustainable gene transfer. Nat. Methods 8, 861–869 (2011).
Voit, R.A., Hendel, A., Pruett-Miller, S.M. & Porteus, M.H. Nuclease-mediated gene editing by homologous recombination of the human globin locus. Nucleic Acids Res. 42, 1365–1378 (2014).
Hsu, P.D. et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 31, 827–832 (2013).
Cradick, T.J., Qiu, P., Lee, C.M., Fine, E.J. & Bao, G. COSMID: a web-based tool for identifying and validating CRISPR-Cas off-target sites. Mol. Ther. Nucleic Acids 3, e214 (2014).
Yi, G. et al. CCR5 gene editing of resting CD4(+) T cells by transient ZFN expression from HIV envelope pseudotyped nonintegrating lentivirus confers HIV-1 resistance in humanized mice. Mol. Ther. Nucleic Acids 3, e198 (2014).
Mandal, P.K. et al. Efficient ablation of genes in human hematopoietic stem and effector cells using CRISPR-Cas9. Cell Stem Cell 15, 643–652 (2014).
Gilbert, L.A. et al. Genome-scale CRISPR-mediated control of gene repression and activation. Cell 159, 647–661 (2014).
Chen, B. et al. Dynamic imaging of genomic loci in living human cells by an optimized CRISPR-Cas system. Cell 155, 1479–1491 (2013).
O'Connell, M.R. et al. Programmable RNA recognition and cleavage by CRISPR-Cas9. Nature 516, 263–266 (2014).
Threlfall, R.N., Torres, A.G., Krivenko, A., Gait, M.J. & Caruthers, M.H. Synthesis and biological activity of phosphonoacetate- and thiophosphonoacetate-modified 2′-O-methyl oligoribonucleotides. Org. Biomol. Chem. 10, 746–754 (2012).
Brinkman, E.K., Chen, T., Amendola, M. & van Steensel, B. Easy quantitative assessment of genome editing by sequence trace decomposition. Nucleic Acids Res. 42, e168 (2014).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595 (2010).
Acknowledgements
A.H. was supported by the Myotonic Dystrophy Foundation. R.O.B. was supported through an Individual Postdoctoral grant (DFF–1333-00106B) and a Sapere Aude, Research Talent grant (DFF–1331-00735B), both from the Danish Council for Independent Research, Medical Sciences. M.H.P. gratefully acknowledges the support of the Amon Carter Foundation, the Laurie Krauss Lacob Faculty Scholar Award in Pediatric Translational Research and US National Institutes of Health grant support PN2EY018244 and R01-AI097320. We thank R. Perriman, and Porteus laboratory members and C. Carstens, S. Laderman and Agilent laboratories members for helpful input, comments and discussion.
Author information
Authors and Affiliations
Contributions
A.H. and R.O.B. contributed equally to this work. A.H., R.O.B., J.T.C., A.B.W., R.B., A.B.K. and D.E.R. performed and designed experiments. D.D., D.E.R. and R.J.K. chose the specific modification types and the positions for their incorporation into the sgRNAs. S.R., B.D.L. and R.J.K. performed synthesis and purification of all the sgRNAs. I.S. and A.T. developed and applied the sequencing data analysis pipeline used to analyze the deep sequencing data. L.B. and M.H.P. directed the research. A.H. and R.O.B. wrote the manuscript with help from all authors.
Corresponding authors
Ethics declarations
Competing interests
A.B.K., D.E.R., I.S., B.D.L., R.J.K., A.T., D.D. and L.B. are employees of Agilent Technologies. M.H.P. is a consultant and has equity interest in CRISPR Tx, but CRISPR Tx had no input into the design, execution, interpretation or publication of the results herein.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–16, Supplementary Tables 1–5 and Supplementary Notes 1 and 2 (PDF 999 kb)
Rights and permissions
About this article
Cite this article
Hendel, A., Bak, R., Clark, J. et al. Chemically modified guide RNAs enhance CRISPR-Cas genome editing in human primary cells. Nat Biotechnol 33, 985–989 (2015). https://doi.org/10.1038/nbt.3290
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3290
This article is cited by
-
High throughput intracellular delivery by viscoelastic mechanoporation
Nature Communications (2024)
-
Transient inhibition of 53BP1 increases the frequency of targeted integration in human hematopoietic stem and progenitor cells
Nature Communications (2024)
-
Improving prime editing with an endogenous small RNA-binding protein
Nature (2024)
-
Nuclear morphology is shaped by loop-extrusion programs
Nature (2024)
-
A potential paradigm in CRISPR/Cas systems delivery: at the crossroad of microalgal gene editing and algal-mediated nanoparticles
Journal of Nanobiotechnology (2023)