Abstract
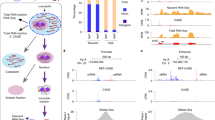
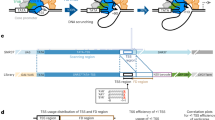
Transcriptional initiation of each gene is assumed to be independently controlled in mammals. On the other hand, recent large-scale transcriptome analyses have shown that the genome is pervasively transcribed, such that the most of its DNA gives rise to RNAs1,2,3,4. This raises the question of whether it is possible to pinpoint and activate a particular locus without perturbing numerous neighbouring transcripts. Here we show that intensive transcription at one locus frequently spills over into its physical neighbouring loci. Rapid induction of immediate-early genes (IEGs) in response to growth factor stimulation5 is accompanied by co-upregulation of their neighbouring genes. Profiling the primary transcripts in the nucleus with whole-genome tiling arrays delineated simultaneous activation of transcription centred on IEGs. Even in surrounding intergenic regions, transcriptional activation took place at the same time. Acetylation levels of histone H3 and H4 are elevated along with the IEG induction and neighbouring co-upregulation. Inhibition of the mitogen-activated protein kinase (MAPK) pathway or the transcription factor SRF suppresses all transcriptional upregulation. These results suggest that transcriptional activation has a ripple effect, which may be advantageous for coordinated expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bertone, P. et al. Global identification of human transcribed sequences with genome tiling arrays. Science 306, 2242–2246 (2004).
Carninci, P. et al. The transcriptional landscape of the mammalian genome. Science 309, 1559–1563 (2005).
Kapranov, P. et al. RNA maps reveal new RNA classes and a possible function for pervasive transcription. Science 316, 1484–1488 (2007).
The ENCODE project Consortium. Identification and analysis of functional elements in 1% of the human genome by the ENCODE pilot project. Nature 447, 799–816 (2007).
Herschman, H. Primary response genes induced by growth factors and tumor promoters. Annu. Rev. Biochem. 60, 281–319 (1991).
Cho, R. et al. A genome-wide transcriptional analysis of the mitotic cell cycle. Mol. Cell 2, 65–73 (1998).
Kruglyak, S. & Tang, H. Regulation of adjacent yeast genes. Trends Genet. 16, 109–111 (2000).
Cohen, B., Mitra, R., Hughes, J. & Church, G. A computational analysis of whole-genome expression data reveals chromosomal domains of gene expression. Nature Genet. 26, 183–186 (2000).
Yamamoto, T. et al. Continuous ERK activation downregulates antiproliferative genes throughout G1 phase to allow cell-cycle progression. Curr. Biol. 16, 1171–1182 (2006).
Trinklein, N. et al. An abundance of bidirectional promoters in the human genome. Genome Res. 14, 62–66 (2004).
Lipson, K. & Baserga, R. Transcriptional activity of the human thymidine kinase gene determined by a method using the polymerase chain reaction and an intron-specific probe. Proc. Natl Acad. Sci. USA 86, 9774–9777 (1989).
Mattick, J. & Makunin, I. Non-coding RNA. Hum. Mol. Genet. 15, R17–R29 (2006).
Katayama, S. et al. Antisense transcription in the mammalian transcriptome. Science 309, 1564–1566 (2005).
Ge, X., Wu, Q., Jung, Y., Chen, J. & Wang, S. A large quantity of novel human antisense transcripts detected by LongSAGE. Bioinformatics 22, 2475–2479 (2006).
Carninci, P. et al. Genome-wide analysis of mammalian promoter architecture and evolution. Nature Genet. 38, 626–635 (2006).
Shiraki, T. et al. Cap analysis gene expression for high-throughput analysis of transcriptional starting point and identification of promoter usage. Proc. Natl Acad. Sci. USA 100, 15776–15781 (2003).
Martens, J., Laprade, L. & Winston, F. Intergenic transcription is required to repress the Saccharomyces cerevisiae SER3 gene. Nature 429, 571–574 (2004).
Martianov, I., Ramadass, A., Serra Barros, A., Chow, N. & Akoulitchev, A. Repression of the human dihydrofolate reductase gene by a non-coding interfering transcript. Nature 445, 666–670 (2007).
Hurst, L., Pál, C. & Lercher, M. The evolutionary dynamics of eukaryotic gene order. Nature Rev. Genet. 5, 299–310 (2004).
Sproul, D., Gilbert, N. & Bickmore, W. The role of chromatin structure in regulating the expression of clustered genes. Nature Rev. Genet. 6, 775–781 (2005).
David, L. et al. A high-resolution map of transcription in the yeast genome. Proc. Natl Acad. Sci. USA 103, 5320–5325 (2006).
Treisman, R. Regulation of transcription by MAP kinase cascades. Curr. Opin. Cell Biol. 8, 205–215 (1996).
Tullai, J., Schaffer, M., Mullenbrock, S., Kasif, S. & Cooper, G. Identification of transcription factor binding sites upstream of human genes regulated by the phosphatidylinositol 3-kinase and MEK/ERK signaling pathways. J. Biol. Chem. 279, 20167–20177 (2004).
Pritchard, C., Samuels, M., Bosch, E. & McMahon, M. Conditionally oncogenic forms of the A-Raf and B-Raf protein kinases display different biological and biochemical properties in NIH 3T3 cells. Mol. Cell. Biol. 15, 6430–6442 (1995).
Kel, A. et al. MATCH: A tool for searching transcription factor binding sites in DNA sequences. Nucleic Acids Res. 31, 3576–3579 (2003).
Norman, C., Runswick, M., Pollock, R. & Treisman, R. Isolation and properties of cDNA clones encoding SRF, a transcription factor that binds to the c-fos serum response element. Cell 55, 989–1003 (1988).
Suzuki, Y., Yamashita, R., Sugano, S. & Nakai, K. DBTSS, DataBase of Transcriptional Start Sites: progress report 2004. Nucleic Acids Res. 32, D78–81 (2004).
Gaszner, M. & Felsenfeld, G. Insulators: exploiting transcriptional and epigenetic mechanisms. Nature Rev. Genet. 7, 703–713 (2006).
Kim, T. et al. Analysis of the vertebrate insulator protein CTCF-binding sites in the human genome. Cell 128, 1231–1245 (2007).
Karolchik, D. et al. The UCSC Genome Browser Database. Nucleic Acids Res. 31, 51–54 (2003).
Acknowledgements
We thank I. Smith for helpful discussion and M. McMahon for ΔB-Raf–ER cells. This work was supported by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan (to E.N.).
Author information
Authors and Affiliations
Contributions
M.E. conceived the study and wrote the manuscript with the help of E.N.; M.E. and T.Y. designed and performed the experiments and analysed the data; M.N. conducted histone acetylation ChIP experiments; E.N. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4, S5, Supplementary Tables 1, 2, 3, 4 and 5 (PDF 2835 kb)
Rights and permissions
About this article
Cite this article
Ebisuya, M., Yamamoto, T., Nakajima, M. et al. Ripples from neighbouring transcription. Nat Cell Biol 10, 1106–1113 (2008). https://doi.org/10.1038/ncb1771
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1771
This article is cited by
-
Extensive fragmentation and re-organization of transcription in Systemic Lupus Erythematosus
Scientific Reports (2020)
-
BLNCR is a long non-coding RNA adjacent to integrin beta-1 that is rapidly lost during epidermal progenitor cell differentiation
Scientific Reports (2019)
-
Co-regulation map of the human proteome enables identification of protein functions
Nature Biotechnology (2019)
-
Important cardiac transcription factor genes are accompanied by bidirectional long non-coding RNAs
BMC Genomics (2018)
-
Intrachromosomal colocalization strengthens co-expression, co-modification and evolutionary conservation of neighboring genes
BMC Genomics (2018)