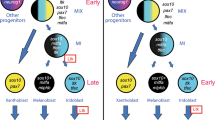
Abstract
Colour patterns are a striking feature of animals; they evolve rapidly and play an important role in natural as well as sexual selection. It has been proposed that colour pattern formation in adult vertebrates depends on Turing-type interactions between pigment cells1; however, little is known about the actual developmental mechanisms underlying the complex and prolonged ontogeny of this important adult feature. Zebrafish (Danio rerio) owe their name to a repetitive pattern of dark stripes and light interstripes parallel to the anteroposterior body axis that develop during juvenile stages2,3. By inducible Cre/loxP-mediated recombination in neural-crest-derived progenitors, we created labelled clones of skin pigment cells that were imaged over several weeks in juvenile and adult fish. Metamorphic iridophores arise from postembryonic stem cells located at the dorsal root ganglia of the peripheral nervous system. They emerge in the skin at the horizontal myoseptum to form the first interstripe and proliferate while spreading bidirectionally along the dorsoventral axis. Patterned aggregation of iridophores during their dispersal generates a series of interstripes that define the stripe regions. Melanophore progenitors appear in situ in the presumptive stripe region where they melanize and expand in size to form compact stripes. Thus, although depending on mutual interactions between different pigment cells, stripes and interstripes are formed by a completely different cellular route.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kondo, S. & Miura, T. Reaction-diffusion model as a framework for understanding biological pattern formation. Science 329, 1616–1620 (2010).
Parichy, D. M., Elizondo, M. R., Mills, M. G., Gordon, T. N. & Engeszer, R. E. Normal table of postembryonic zebrafish development: staging by externally visible anatomy of the living fish. Dev. Dynam. 238, 2975–3015 (2009).
Johnson, S. L., Africa, D., Walker, C. & Weston, J. A. Genetic control of adult pigment stripe development in zebrafish. Dev. Biol. 167, 27–33 (1995).
Hirata, M., Nakamura, K., Kanemaru, T., Shibata, Y. & Kondo, S. Pigment cell organization in the hypodermis of zebrafish. Dev. Dynam. 227, 497–503 (2003).
Frohnhöfer, H. G., Krauss, J., Maischein, H. M. & Nüsslein-Volhard, C. Iridophores and their interactions with other chromatophores are required for stripe formation in zebrafish. Development 140, 2997–3007 (2013).
Maderspacher, F. & Nüsslein-Volhard, C. Formation of the adult pigment pattern in zebrafish requires leopard and obelix dependent cell interactions. Development 130, 3447–3457 (2003).
Patterson, L. B. & Parichy, D. M. Interactions with iridophores and the tissue environment required for patterning melanophores and xanthophores during zebrafish adult pigment stripe formation. PLoS Genet. 9, e1003561 (2013).
Nakamasu, A., Takahashi, G., Kanbe, A. & Kondo, S. Interactions between zebrafish pigment cells responsible for the generation of Turing patterns. Proc. Natl Acad. Sci. USA 106, 8429–8434 (2009).
Krauss, J., Astrinides, P., Frohnhofer, H. G., Walderich, B. & Nüsslein-Volhard, C. transparent, a gene affecting stripe formation in Zebrafish, encodes the mitochondrial protein Mpv17 that is required for iridophore survival. Biol. Open 2, 703–710 (2013).
Takahashi, G. & Kondo, S. Melanophores in the stripes of adult zebrafish do not have the nature to gather, but disperse when they have the space to move. Pigment Cell Melanoma Res. 21, 677–686 (2008).
Inaba, M., Yamanaka, H. & Kondo, S. Pigment pattern formation by contact-dependent depolarization. Science 335, 677 (2012).
Painter, K. J., Maini, P. K. & Othmer, H. G. Stripe formation in juvenile Pomacanthus explained by a generalized Turing mechanism with chemotaxis. Proc. Natl Acad. Sci. USA 96, 5549–5554 (1999).
Lang, M. R., Patterson, L. B., Gordon, T. N., Johnson, S. L. & Parichy, D. M. Basonuclin-2 requirements for zebrafish adult pigment pattern development and female fertility. PLoS Genet. 5, e1000744 (2009).
Mongera, A. et al. Genetic lineage labeling in zebrafish uncovers novel neural crest contributions to the head, including gill pillar cells. Development 140, 916–925 (2013).
Bertrand, J. Y. et al. Haematopoietic stem cells derive directly from aortic endothelium during development. Nature 464, 108–111 (2010).
Levesque, M. P., Krauss, J., Koehler, C., Boden, C. & Harris, M. P. New tools for the identification of developmentally regulated enhancer regions in embryonic and adult zebrafish. Zebrafish 10, 21–29 (2013).
Lopes, S. S. et al. Leukocyte tyrosine kinase functions in pigment cell development. PLoS Genet. 4, e1000026 (2008).
Parichy, D. M. et al. Mutational analysis of endothelin receptor b1 (rose) during neural crest and pigment pattern development in the zebrafish Danio rerio. Dev. Biol. 227, 294–306 (2000).
Dooley, C. M., Mongera, A., Walderich, B. & Nüsslein-Volhard, C. On the embryonic origin of adult melanophores: the role of ErbB and Kit signalling in establishing melanophore stem cells in zebrafish. Development 140, 1003–1013 (2013).
Parichy, D. M. & Turner, J. M. Temporal and cellular requirements for Fms signaling during zebrafish adult pigment pattern development. Development 130, 817–833 (2003).
Odenthal, J. et al. Mutations affecting xanthophore pigmentation in the zebrafish, Danio rerio. Development 123, 391–398 (1996).
Lister, J. A., Robertson, C. P., Lepage, T., Johnson, S. L. & Raible, D. W. nacre encodes a zebrafish microphthalmia-related protein that regulates neural-crest-derived pigment cell fate. Development 126, 3757–3767 (1999).
Brand, M., Granato, M. & Nüsslein-Volhard, C. Zebrafish, Practical Approach 7–37 (Oxford Univ. Press, 2002).
Walker, C. & Streisinger, G. Induction of mutations by gamma-rays in pregonial germ cells of zebrafish embryos. Genetics 103, 125–136 (1983).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Preibisch, S., Saalfeld, S. & Tomancak, P. Globally optimal stitching of tiled 3D microscopic image acquisitions. Bioinformatics 25, 1463–1465 (2009).
Acknowledgements
We thank A. Mongera and M. Levesque for providing transgenic lines before publication; C. Dooley for initial help; and C.N-V. laboratory members, P. Müller, D. Gilmour and F. Chan for discussions and comments. We thank C. Liebig and the light microscopy facility, B. Walderich and the fish facility for great support. This work was financially supported by the Max-Planck Society for the Advancement of Science. A.P.S. received a long-term postdoctoral fellowship (EMBO-LTF) from the European Molecular Biology Organization.
Author information
Authors and Affiliations
Contributions
A.P.S. and C.N-V. conceived and designed experiments; A.P.S. and U.S. performed experiments; A.P.S. and C.N-V. analysed data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Pigment cell clones vary in size and cellular composition.
(a–o) Cre-lox induced clones (red) in the juvenile skin (¿14 mmSL) of Tg(sox10:ERT2-Cre); Tg(actin2:loxP-STOP-loxP-DsRed-express) animals label various cell types. (a–b) Examples of small clones with only iridophore labelling, (c–g, k–l) typical mixed clones with iridophores and variable numbers of melanophores, and (i–j) small clones with only melanophore labelling. Iridophore clones in (a,b) are restricted to the first interstripe along the D-V axis, clones in (d–g,l) extend both dorsally and ventrally, clones in (h, k) cover the skin predominantly ventrally whereas clone in (c) predominantly cover the skin dorsally of the first interstripe. Labelled pigment cells of the scales can be seen in (d, e, l). (n—r”) Different kinds of pigment cells can be identified by their distinct shape, location, and pigment content. (n–n”) a single labelled melanophore in the stripe of a juvenile shows characteristic morphology and black pigment (arrow in n’). (o–o”) Dense iridophores are located in the interstripe (arrowheads). (p–p”) loose iridophores and (q–q”) L-iridophores are located in the stripe region and are close to the melanophores. (r–r”) Xanthophores have distinctly different morphology and packaging in the interstripe compared to the dense S-iridophores. Scale bar: 100 m. (s) The majority of the iridophore clones span two to four segments along the anterior-posterior axis (79%, n = 136 clones) and (t) more than one stripe along the dorso-ventral axis (87%, n = 163 clones). (u) Labeling of iridophores in the new interstripes is strongly associated with labeling of iridophores in the first interstripe (93.6%, n = 142 clones). (v) Frequency of association of iridophores with the DRGs.
Supplementary Figure 2 Metamorphic iridophores are clonally associated with the neurons and glia of the DRGs.
(a1–a3’) In a premetamorphic Tg(sox10:ERT2-Cre); Tg(β-actin2:loxP-STOP-loxP-DsRed-express) larva with Cre-induced clones, a couple of labelled cells (red asterisk) can be seen. At the onset of metamorphosis, these cells start to proliferate and differentiate into the neurons and glia of the DRGs (red asterisk in a2-a3) and into iridophores (yellow arrowheads in a3). (b1–h2) Several other cell types were also observed before and after metamorphosis which did not make iridophores. (i1–i2’) shows another example of clonal and developmental association between the iridophores (yellow arrowheads) and the cells of the DRG (asterisk). Scale bar: 100 μm.
Supplementary Figure 3 nterstripe formation precedes stripe formation and is necessary for subsequent stripe formation.
(a–c) At the onset of metamorphosis, clusters of iridophores appear along the horizontal myoseptum (arrowheads in b). While the clusters of iridophores form the first interstripe (X0 in c), melanophores (arrows in c) begin to appear in the skin. Stripe 1D and 1V become clear at later stages as more melanophores appear in the stripe region (e–f). (d–h) The initial fuzzy boundary between stripes and interstripe gets sharpened during metamorphosis. New interstripe (X1V) starts to appear at subsequent stages (40-50 dpf) and is prominent in (h-i) while dark stripe are yet to develop. Scale bar: 100 m. (j) Compared with wild-type controls, iridophore mutants add fewer stripes beyond the basic metamorphic stripes 1D and 1V. (k-l) In animals homozygous for a weak allele of rose (genotype: rosetAN17X;Tg(sox10 : ERT2 − Cre)/+;Tg(β − actin2 : loxP − STOP − loxP − DsRed − express)/+), iridophores appear scattered (arrowheads in c indicate a few scattered loose iridophores) and disorganized unlike the iridophores in the (b) control sibling. Arrow: a small cluster of iridophores in the interstripe X0.
Supplementary Figure 4 Development of metamorphic melanophores which appear near the ventral myotome.
(A-C’) Melanophores (arrows) and unpigmented cells (arrowheads) are seen close to the ventral myotome in a clone. The melanophores contribute to the ventral stripe (1V). Scale bar: 100 μm
Supplementary information
Supplementary Information
Supplementary Information (PDF 640 kb)
Iridophore behaviour in the skin during metamorphosis.
Time-lapse images from the Figure 2c-x are compiled in this video after manual alignment of the stacks in Adobe Photoshop. The stacks encompass metamoprphic stages from 28 dpf to 45 dpf. (AVI 4179 kb)
Rights and permissions
About this article
Cite this article
Singh, A., Schach, U. & Nüsslein-Volhard, C. Proliferation, dispersal and patterned aggregation of iridophores in the skin prefigure striped colouration of zebrafish. Nat Cell Biol 16, 604–611 (2014). https://doi.org/10.1038/ncb2955
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2955
This article is cited by
-
Reaction-diffusion in a growing 3D domain of skin scales generates a discrete cellular automaton
Nature Communications (2021)
-
Influence of survival, promotion, and growth on pattern formation in zebrafish skin
Scientific Reports (2021)
-
Nerve-associated Schwann cell precursors contribute extracutaneous melanocytes to the heart, inner ear, supraorbital locations and brain meninges
Cellular and Molecular Life Sciences (2021)
-
In situ differentiation of iridophore crystallotypes underlies zebrafish stripe patterning
Nature Communications (2020)
-
Evolution of the potassium channel gene Kcnj13 underlies colour pattern diversification in Danio fish
Nature Communications (2020)