Abstract
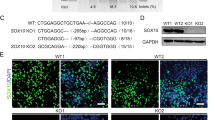
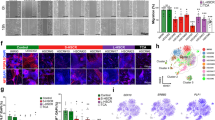
Hirschsprung disease (HSCR, MIM ♯142623) is a multigenic neuocristopathy (neural crest disorder) characterized by absence of enteric ganglia in a variable portion of the distal colon. Subsets of HSCR individuals also present with neural crest-derived melanocyte deficiencies (Hirschsprung-Waardenburg, HSCR-WS, MIM ♯277580). Murine models have been instrumental in the identification and analysis of HSCR disease genes. These include mice with deficiencies of endothelin B receptor (Ednrbs–l refs 1,2) endothelin 3 (Edn3ls; refs 1,3) the tyrosine kinase receptor cftet4 and glial-derived neurotrophic factor5–7. Another mouse model of HSCR disease, Dom, arose spontaneously at the Jackson Laboratory8. While Dom/+ heterozygous mice display regional deficiencies of neural crest–derived enteric ganglia in the distal colon, Dom/Dom homozygous animals are embryonic lethal8. We have determined that premature termination of SOX10, a member of the SflV–like HMG box family of transcription factors, is responsible for absence of the neural crest derivatives in Dom mice. We demonstrate expression of SOX10 in normal neural crest cells, disrupted expression of both SOX10 and the HSCR disease gene Ednrb in Dom mutant embryos, and loss of neural crest derivatives due to apoptosis. Our studies suggest that SOX10 is essential for proper peripheral nervous system development. We propose SOX10 as a candidate disease gene for individuals with HSCR whose disease does not have an identified genetic origin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lane, P.W. Association of megacolon with two recessive spotting genes in the mouse. J. Hered. 57, 29–31 (1966).
Hosoda, K. et al. Targeted and natural (piebald-lethal) mutations of endothelin-B receptor gene produce megacolon associated with spotted coat color in mice. Cell 79, 1267–1276 (1994)
Baynash, A.G. et al. Interaction of endothelin-3 with endothelin-B receptor is essential for development of epidermal melanocytes and enteric neurons. Cell 79, 1277–1285 (1994).
Schuchardt, A., D'Agati, V., Larsson-Blomberg, L., Costantini, F. & Pachnis, V. Defects in the kidney and enteric nervous system of mice lacking the tyrosine kinase receptor Ret. Nature 367, 380–383 (1994).
Sanchez, M.P. et al. Renal agenesis and the absence of enteric neurons in mice lacking GDNF. Nature 382, 70–73 (1996).
Pichel, J.G. et al. Defects in enteric innervation and kidney development in mice lacking GDNF. Nature 382, 73–76 (1996).
Moore, M.W. et al. Renal and neuronal abnormalities in mice lacking GDNF. Nature 382, 76–79 (1996).
Lane, P.W. & Liu, H.M. Association of megacolon with a new dominant spotting gene (Dom) in the mouse. J. Hered. 75, 435–439 (1984).
Pingault, V. et al. Human homology and candidate genes for the Dominant megacolon locus, a mouse model of Hirschsprung disease. Genomics 39, 86–89 (1997).
Puliti, A. et al. A high-resolution genetic map of mouse chromosome 15 encompassing the Dominant megacolon (Dom) locus. Mamm. Genome 6, 763–768 (1995).
Stock, D.W., Buchanan, A.V., Zhao, Z. & Weiss, K.M. Numerous members of the Sox family of HMG box-containing genes are expressed in developing mouse teeth. Genomics 37, 234–237 (1996).
Wright, E.M., Snopek, B. & Koopman, P. Seven new memebers of the Sox gene family of HMG box-containing genes are expressed during mouse development Nucleic Acids Res. 21, 744 (1993).
Tani, M. et al. Isolation of a novel Sry-related gene that is expressed in high-metastatic K-1735 murine melanoma cells. Genomics 39, 30–37 (1997).
Hashimoto, Y. et al. Identification of genes differentially expressed in association with metastatic potential of K-1735 murine melanoma by messenger RNA differential display. Cancer Res. 56, 5266–5271 (1996).
Bennett, D.C., Cooper, P.J. & Hart, I.R. A line of non-tumorigenic mouse melanocytes, syngeneic with the B16 melanoma and requiring a tumour promoter for growth. Int. J. Cancer 39, 414–418 (1987).
Gershon, M.D. Neural crest development: do developing enteric neurons need endothelins? Curr. Biol. 5, 601–604 (1995).
Kapur, R.P., Sweetser, D.A., Doggett, B., Siebert, J.R. & Palmiter, R.D. Intercellular signals downstream of endothelin receptor-B mediate colonization of the large intestine by enteric neuroblasts. Development 121, 3787–3795 (1995).
Lahav, R., Ziller, C, Dupin, E. & Le Douarin, N.M. Endothelin 3 promotes neural crest cell proliferation and mediates a vast increase in melanocyte number in culture. Proc. atal. Acad. Sci. USA 93, 3892–3897 (1996).
Opdecamp, K. et al. Melanocyte development in vivo and in neural crest cel cultures: crucial dependence on the Mitf basic-helix-loop-helix-zipper transcription factor. Development 124, 2377–2386 (1997).
Reid, K. et al. Multiple roles for endothelin in melanocyte development: regulation of progenitor number and stimulation of differentiation. Development 122, 3911–3919 (1996).
Steel, K.P., Davidson, D.R. & Jackson, I.J. TRP-2/DT, a new early melanoblast marker, shows that steel growth factor (c-kit ligand) is a survival factor. Development 115, 1111–1119 (1992).
Pavan, W.J. & Tilghman, S.M. Piebald lethal (s1) acts early to disrupt the development of neural crest-derived melanocytes. Proc. Natl. Acad. Sci. USA 91, 7159–7163 (1994).
Pevny, L.H. & Lovell-Badge, R. Sox genes find their feet. Curr. Opin. Genet. Dev. 7, 338–344 (1997).
Prior, H.M. & Walter, M.A. SOX genes: architects of development. Mol. Med. 2, 405–412 (1996).
Foster, J.W. et al. Campomelic dysplasia and autosomal sex reversal caused by mutations in an SKV-related gene. Nature 372, 525–530 (1994).
Schilham, M.W., Moerer, P., Cumano, A. & Clevers, H.C. Sox-4 facilitates thymocyte differentiation. Eur. J. Immunol. 27, 1292–1295 (1997).
Wagner, T. et al. Autosomal sex reversal and campomelic dysplasia are caused by mutations in and around the SRY-related gene SOX9. Cell 79, 1111–1120 (1994).
Bell, D.M. et al. SOX9 directly regulates the type-ll collagen gene. Nature Genet. 16, 174–178 (1997).
Sudbeck, P., Schmitz, M.L., Baeuerle, P.A. & Scherer, G. Sex reversal by loss of the C-terminal transactivation domain of human SOX9. Nature Genet. 13, 230–232 (1996).
Kapur, R.P. et al. Abnormal microenvironmental signals underlie intestinal aganglionosis in Dominant megacolon mutant mice. Dev. Biol. 174, 360–369 (1996).
Kapur, R.P., Yost, C. & Palmiter, R.D. Aggregation chimeras demonstrate that the primary defect responsible for aganglionic megacolon in lethal spotted mice is not neuroblast autonomous. Development 117, 993–999 (1993).
Rothman, T.P., Goldowitz, D. & Gershon, M.D. Inhibition of migration of neural crest-derived cells by the abnormal mesenchyme of the presumptive aganglionic bowel of Is/Is mice: analysis with aggregation and interspecies chimeras. Dev. Biol. 159, 559–573 (1993).
Dudov, K.P. & Perry, R.P. The gene family encoding the mouse ribosomal protein L32 contains a uniquely expressed intron-containing gene and an unmutated processed gene. Cell 37, 457–468 (1984).
Pavan, W.J., Mac, S., Cheng, M. & Tilghman, S.M. Quantitative trait loci that modify the severity of spotting in piebald mice. Genome Res. 5, 29–41 (1995).
Swift, G.H., Dagorn, J.C., Ashley, P.L, Cummings, S.W. & MacDonald, R.J. Rat pancreatic kallikrein mRNA: nucleotide sequence and amino acid sequence of the encoded preproenzyme. Proc. Natl. Acad. Sci. USA 79, 7263–7267 (1982).
Wilkinson, D.G. & Nieto, M.A. Detection of messenger RNA by in situ hybridization to tissue sections and whole mounts. Methods Enzymol. 225, 361–373 (1993).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Southard-Smith, E., Kos, L. & Pavan, W. SOX10 mutation disrupts neural crest development in Dom Hirschsprung mouse model. Nat Genet 18, 60–64 (1998). https://doi.org/10.1038/ng0198-60
Issue Date:
DOI: https://doi.org/10.1038/ng0198-60
This article is cited by
-
The interaction between the nervous system and the stomatognathic system: from development to diseases
International Journal of Oral Science (2023)
-
mRNA sequencing provides new insights into the pathogenesis of Hirschsprung’s disease in mice
Pediatric Surgery International (2023)
-
Similarities and differences between nigral and enteric dopaminergic neurons unravel distinctive involvement in Parkinson’s disease
npj Parkinson's Disease (2022)
-
Genetic insights, disease mechanisms, and biological therapeutics for Waardenburg syndrome
Gene Therapy (2022)
-
Epigenome erosion and SOX10 drive neural crest phenotypic mimicry in triple-negative breast cancer
npj Breast Cancer (2022)