Abstract
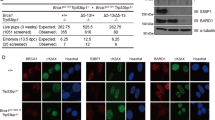
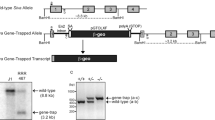
Mutations in the mouse Brca1 gene cause lethality at different embryonic stages1–3. We have shown that Brca15–6 mutant embryos, in which the fifth and sixth exons of Brcal are deleted, die before E7.5 and show decreased cellular proliferation2. Brca15–6 mutants also show decreased expression of mdm2, a gene encoding an inhibitor ofp53 activity4. Thus, we have proposed that the reduction in mdm2 expression in Brca15–6 mutants might lead to increased p53 activity. Consistent with this finding, the expression of p21, which encodes a G1 cell cycle inhibitor5–10 and is a target for p53 transcriptional activation, was dramatically increased in the Brca15–6 mutants, suggesting that impaired cellular proliferation could be due to a G1 cell-cycle arrest, caused by increased p21 levels. To test this hypothesis, we generated mice double mutant for Brca15–6 and p53, or Brca15–6 and p21. Mutation in either p53 or p21 prolonged the survival of Brca15–6 mutant embryos from E7.5 to E9.5. The development of most Brca15–6: p21 double-mutant embryos was comparable to that of their wild-type littermates, although no mutant survived past E10.5. The fact that mutation of neither p53 nor p21 completely rescued Brca15–6 embryos suggests that their lethality is likely due to a multi-factorial process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gowen, L.C., Johnson, B.L., Latour, A.M., Sulik, K.K. & Koller, B.H. Brca1 deficiency results in early embryonic lethality characterized by neuroepithelial abnormalities. Nature Genet. 12, 191–194 (1996).
Hakem, R. et al. The tumor suppressor gene Brcal is required for embryonic cellular proliferation in the mouse. Cell 85, 1009–1023 (1996).
Liu, C.Y., Fleskennikitin, A., Li, S., Zeng, Y.Y. & Lee, W.H. Inactivation of the mouse Brcal gene leads to failure in the morphogenesis of the egg cylinder in early postimplantation development. Genes Dev. 10, 1835–1843 (1996).
Momand, J., Zambetti, G.P., Olson, D.C., George, D. & Levine, A.J. The mdm-2 oncogene product forms a complex with the p53 protein and inhibits p53 mediated transaction. Cell 69, 1237–1245 (1992).
Gu, Y., Turek, C.W. & Morgan, D.O. Inhibition of CDK2 activity in vivo by an associated 20k regulatory subunit. Nature 366, 707–710 (1993).
Harper, J.W., Adami, G.R., Wei, N., Keyomarsi, K. & Elledge, S.J. The p21-cdk interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 75, 805–316 (1993).
EL-Deiry, W.S. et al. WAF1, a potential mediator of p53 tumor suppression. Cell 75, 817–625 (1993).
Xiong, Y. et al. p21 is a universal inhibitor of cyclin kinases. Nature 366, 701–704 (1993).
EI-Deiry, W.S. et al. WAF/CIP1 is induced in p53-mediated G1 arrest and apoptosis. Cancer Res. 54, 1169–1174 (1994).
Dulic, V. et al. p53-dependent inhibition of cyclin-dependent kinase-activities in human fibroblasts during radiation-induced G1 arrest. Cell 76, 1013–1023 (1994).
Jacks, T. et al. Tumor spectrum analysis in p53-mutant mice. Curr. Biol. 4, 1–7 (1994).
Brugarolas, J. et al. Radiation-induced cell cycle arrest compromised by p21 deficiency. Nature 377, 552–557 (1995).
Kaufman, M.H. Morphological stages of postimplantation embryonic development. In Postimplantation Mammalian Embryos.(eds Copp, A.J. & Cockroft, D.L.) IRL Press, Oxford,81–91 ( 1990).
Scully, R. et al. Association of BRCA1 with Rad51 in mitotic and meiotic cells. Cell 88, 265–275 (1997).
Sharan, S.K. et al. Embryonic lethality and radiation hypersensitivity mediated by Rad51 in mice lacking Brca2. Nature 386, 804–810 (1997).
Radding, C.M. Helical interactions in homologous pairing and strand exchange driven by RecA protein. J. Biol. Chem. 266, 5355–5358 (1991).
Aboussekhra, A., Chanet, R., Adjiri, A. & Fabre, F. Semidominant suppressors of Srs2 helicase mutations of Saccharomyces cerevisiae map in the RAD51 gene, whose sequence predicts a protein with similarities to procaryotic RecA proteins. Mol. Cell. Biol. 12, 3224–3234 (1992).
Shinohara, A., Ogawa, H. & Ogawa, T. Rad51 protein involved in repair and recombination in S. cerevisiae is a RecA-like protein. Cell 69, 457–470 (1992).
Lim, D.-S. & Hasty, P.A. mutation in mouse rad51 results in a early embryonic lethal that is suppressed by a mutation in p53. Mol. Cell. Biol. 16, 7133–7143 (1996).
Tsuzuki, T. et al. Targeted disruption of the Rad51 gene leads to lethality in embryonic mice. Proc. Natl. Acad. Sci. USA 93, 6336–6240 (1996).
Levine, A.J. p53, the cellular gatekeeper for growth and division. Cell 88, 323–331 (1997).
Elledge, S.J. Cell cycle checkpoints: preventing an identity crisis. Science 274, 1664–1672 (1996).
Selivanova, G. & Wiman, K.G. p53: a cell cycle regulator activated by DMA damage. Adv. Cancer Res. 66, 143–180 (1995).
Guidos, C.J. et al. V(D)J recombination activates a p53-dependent DNA damage checkpoint in scid lymphocyte precursors. Genes Dev. 10, 2038–2054 (1996).
Huang, L.-C. & Prives, C. p53: puzzle and paradigm. Genes Dev. 10, 1054–1072 (1996).
Huang, L.-C., Clarkin, K.C. & Wahl, G.M Sensitivity and selectivity of the DNA damage sensor responsible for activating p53-dependent G1 arrest. Proc. Natl. Acad. Sci. USA 93, 4827–4832 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hakem, R., de la Pompa, J., Elia, A. et al. Partial rescue of Brca15–6 early embryonic lethality by p53 or p21 null mutation. Nat Genet 16, 298–302 (1997). https://doi.org/10.1038/ng0797-298
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0797-298
This article is cited by
-
BRCA1 and homologous recombination: implications from mouse embryonic development
Cell & Bioscience (2020)
-
Roles of BCCIP deficiency in mammary tumorigenesis
Breast Cancer Research (2017)
-
Selective mode of action of plumbagin through BRCA1 deficient breast cancer stem cells
BMC Cancer (2016)
-
The role of BRCA1 in homologous recombination repair in response to replication stress: significance in tumorigenesis and cancer therapy
Cell & Bioscience (2013)
-
Embryonic stem cells shed new light on the developmental roles of p53
Cell & Bioscience (2013)