Abstract
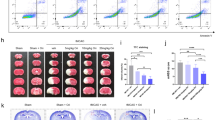
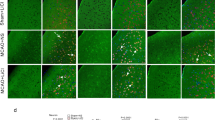
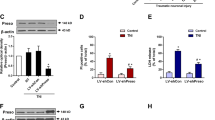
Stroke is a major public health problem leading to high rates of death and disability in adults1,2. Excessive stimulation of N-methyl-D-aspartate receptors (NMDARs) and the resulting neuronal nitric oxide synthase (nNOS) activation are crucial for neuronal injury after stroke insult3,4,5,6,7. However, directly inhibiting NMDARs or nNOS can cause severe side effects because they have key physiological functions in the CNS5,8,9,10,11,12. Here we show that cerebral ischemia induces the interaction of nNOS with postsynaptic density protein-95 (PSD-95). Disrupting nNOS–PSD-95 interaction via overexpressing the N-terminal amino acid residues 1–133 of nNOS (nNOS-N1–133) prevented glutamate-induced excitotoxicity and cerebral ischemic damage. Given the mechanism of nNOS–PSD-95 interaction, we developed a series of compounds and discovered a small-molecular inhibitor of the nNOS-PSD-95 interaction, ZL006. This drug blocked the ischemia-induced nNOS–PSD-95 association selectively, had potent neuroprotective activity in vitro and ameliorated focal cerebral ischemic damage in mice and rats subjected to middle cerebral artery occlusion (MCAO) and reperfusion. Moreover, it readily crossed the blood-brain barrier, did not inhibit NMDAR function, catalytic activity of nNOS or spatial memory, and had no effect on aggressive behaviors. Thus, this new drug may serve as a treatment for stroke, perhaps without major side effects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
07 September 2011
In the version of this article initially published, one of the structural details for the compound ZL010 in Figure 3f was incorrect. The R1 of ZL010 should be OCH3, not OH. This error does not affect the interpretation of the data or the conclusions of the paper. The error has been corrected in the HTML and PDF versions of the article.
References
Flynn, R.W., MacWalter, R.S. & Doney, A.S. The cost of cerebral ischaemia. Neuropharmacology 55, 250–256 (2008).
Gállego, J., Muñoz, R. & Martínez-Vila, E. Emergent cerebrovascular disease risk factor weighting: is transient ischemic attack an imminent threat? Cerebrovasc. Dis. 27 Suppl 1, 88–96 (2009).
Lee, J.M., Zipfel, G.J. & Choi, D.W. The changing landscape of ischaemic brain injury mechanisms. Nature 399, A7–A14 (1999).
Arundine, M. & Tymianski, M. Molecular mechanisms of glutamate-dependent neurodegeneration in ischemia and traumatic brain injury. Cell. Mol. Life Sci. 61, 657–668 (2004).
Lipton, S.A. Pathologically activated therapeutics for neuroprotection. Nat. Rev. Neurosci. 8, 803–808 (2007).
Sattler, R. et al. Specific coupling of NMDA receptor activation to nitric oxide neurotoxicity by PSD-95 protein. Science 284, 1845–1848 (1999).
Dawson, V.L., Kizushi, V.M., Huang, P.L., Snyder, S.H. & Dawson, T.M. Resistance to neurotoxicity in cortical cultures from neuronal nitric oxide synthase-deficient mice. J. Neurosci. 16, 2479–2487 (1996).
Smith, P.F. Therapeutic N-methyl-d-aspartate receptor antagonists: will reality meet expectation? Curr. Opin. Investig. Drugs 4, 826–832 (2003).
Muir, K.W. Glutamate-based therapeutic approaches: clinical trials with NMDA antagonists. Curr. Opin. Pharmacol. 6, 53–60 (2006).
Lipton, S.A. Paradigm shift in neuroprotection by NMDA receptor blockade: memantine and beyond. Nat. Rev. Drug Discov. 5, 160–170 (2006).
Kelley, J.B., Balda, M.A., Anderson, K.L. & Itzhak, Y. Impairments in fear conditioning in mice lacking the nNOS gene. Learn. Mem. 16, 371–378 (2009).
Zhou, L. & Zhu, D.Y. Neuronal nitric oxide synthase: structure, subcellular localization, regulation, and clinical implications. Nitric Oxide 20, 223–230 (2009).
Aarts, M. et al. Treatment of ischemic brain damage by perturbing NMDA receptor–PSD-95 protein interactions. Science 298, 846–850 (2002).
Cao, J. et al. The PSD95-nNOS interface: a target for inhibition of excitotoxic p38 stress-activated protein kinase activation and cell death. J. Cell Biol. 168, 117–126 (2005).
Hecker, M., Mülsch, A. & Busse, R. Subcellular localization and characterization of neuronal nitric oxide synthase. J. Neurochem. 62, 1524–1529 (1994).
Rothe, F., Canzler, U. & Wolf, G. Subcellular localization of the neuronal isoform of nitric oxide synthase in the rat brain: a critical evaluation. Neuroscience 83, 259–269 (1998).
Brenman, J.E. et al. Interaction of nitric oxide synthase with the postsynaptic density protein PSD-95 and α1-syntrophin mediated by PDZ domains. Cell 84, 757–767 (1996).
Cui, H. et al. PDZ protein interactions underlying NMDA receptor-mediated excitotoxicity and neuroprotection by PSD-95 inhibitors. J. Neurosci. 27, 9901–9915 (2007).
Takagi, N., Logan, R., Teves, L., Wallace, M.C. & Gurd, J.W. Altered interaction between PSD-95 and the NMDA receptor following transient global ischemia. J. Neurochem. 74, 169–178 (2000).
Cappuccio, J.A. et al. Cell-free expression for nanolipoprotein particles: building a high-throughput membrane protein solubility platform. Methods Mol. Biol. 498, 273–296 (2009).
Tochio, H., Hung, F., Li, M., Bredt, D.S. & Zhang, M. Solution structure and backbone dynamics of the second PDZ domain of postsynaptic density-95. J. Mol. Biol. 295, 225–237 (2000a).
Tochio, H. et al. Formation of nNOS/PSD-95 PDZ dimer requires a preformed beta-finger structure from the nNOS PDZ domain. J. Mol. Biol. 303, 359–370 (2000b).
Chiavegatto, S. et al. Brain serotonin dysfunction accounts for aggression in male mice lacking neuronal nitric oxide synthase. Proc. Natl. Acad. Sci. USA 98, 1277–1281 (2001).
Huang, Z. et al. Effects of cerebral ischemia in mice deficient in neuronal nitric oxide synthase. Science 265, 1883–1885 (1994).
Moghaddam, B. Bringing order to the glutamate chaos in schizophrenia. Neuron 40, 881–884 (2003).
Gielen, M., Siegler Retchless, B., Mony, L., Johnson, J.W. & Paoletti, P. Mechanism of differential control of NMDA receptor activity by NR2 subunits. Nature 459, 703–707 (2009).
Koroshetz, W.J. & Moskowitz, M.A. Emerging treatments for stroke in humans. Trends Pharmacol. Sci. 17, 227–233 (1996).
Kemp, J.A. & McKernan, R.M. NMDA receptor pathways as drug targets. Nat. Neurosci. 5, 1039–1042 (2002).
Nelson, R.J. et al. Behavioural abnormalities in male mice lacking neuronal nitric oxide synthase. Nature 378, 383–386 (1995).
Tanda, K. et al. Abnormal social behavior, hyperactivity, impaired remote spatial memory, and increased D1-mediated dopaminergic signaling in neuronal nitric oxide synthase knockout mice. Mol. Brain 2, 19 (2009).
Lea, P.M., Movsesyan, V.A. & Faden, A.I. Neuroprotective activity of the mGluR5 antagonists MPEP and MTEP against acute excitotoxicity differs and does not reflect actions at mGluR5 receptors. Br. J. Pharmacol. 145, 527–534 (2005).
Quintana, E., Hernández, C., Alvarez-Barrientos, A., Esplugues, J.V. & Barrachina, M.D. Synthesis of nitric oxide in postganglionic myenteric neurons during endotoxemia: implications for gastric motor function in rats. FASEB J. 18, 531–533 (2004).
Qoronfleh, M.W., Benton, B., Ignacio, R. & Kaboord, B. Selective enrichment of membrane proteins by partition phase separation for proteomic studies. J. Biomed. Biotechnol. 2003, 249–255 (2003).
Longa, E.Z., Weinstein, P.R., Carlson, S. & Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20, 84–91 (1989).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China ((30971021, 81030023) (D.-Y.Z.) and (30901550) (C.-X.L.)). We thank X. Jin, Q.-P. Li, C.-C. Cao, H.-H. Zhou, W.-X. Sun, D.-L. Wu, Y. Hu and J. Zhang for technical assistance.
Author information
Authors and Affiliations
Contributions
L.Z. contributed to the design of the study and performed the cell culture studies, coimmunoprecipitation, western blotting, imaging of NO synthesis and surgical preparation. F.L. performed the design and synthesis of target compounds. H.-B.X. contributed to coimmunoprecipitation, structure-activity relationship analyses and behavioral analyses. C.-X.L. contributed to the design of the study and performed cell culture and morphological analysis of cortical neurons. H.-Y.W. participated in surgical preparation, infarct volume measurement, neuroscore assessment, physiological parameter determination, platelet aggregation and bleeding time measurements. M.-M.Z. performed behavioral analyses, lentivirus production and stereotaxic injection. W.L. performed electrophysiological experiments. X.J. participated in synthesis of target compounds. Q.-G.Z. participated in western blotting analysis. D.-Y.Z. initiated the project and participated in the design of the studies. All authors contributed to data analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 845 kb)
Rights and permissions
About this article
Cite this article
Zhou, L., Li, F., Xu, HB. et al. Treatment of cerebral ischemia by disrupting ischemia-induced interaction of nNOS with PSD-95. Nat Med 16, 1439–1443 (2010). https://doi.org/10.1038/nm.2245
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2245
This article is cited by
-
Surface-Tailored Nanoplatform for the Diagnosis and Management of Stroke: Current Strategies and Future Outlook
Molecular Neurobiology (2024)
-
Cerebral organoids transplantation repairs infarcted cortex and restores impaired function after stroke
npj Regenerative Medicine (2023)
-
Humanized cerebral organoids-based ischemic stroke model for discovering of potential anti-stroke agents
Acta Pharmacologica Sinica (2023)
-
Enhanced AMPAR-dependent synaptic transmission by S-nitrosylation in the vmPFC contributes to chronic inflammatory pain-induced persistent anxiety in mice
Acta Pharmacologica Sinica (2023)
-
Upregulation of HDAC9 in hippocampal neurons mediates depression-like behaviours by inhibiting ANXA2 degradation
Cellular and Molecular Life Sciences (2023)