Abstract
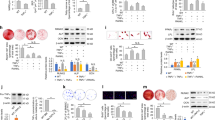
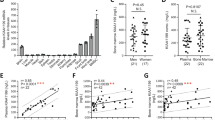
Insulin-like growth factor 1 (IGF-1), the most abundant growth factor in the bone matrix, maintains bone mass in adulthood. We now report that IGF-1 released from the bone matrix during bone remodeling stimulates osteoblastic differentiation of recruited mesenchymal stem cells (MSCs) by activation of mammalian target of rapamycin (mTOR), thus maintaining proper bone microarchitecture and mass. Mice with knockout of the IGF-1 receptor (Igf1r) in their pre-osteoblastic cells showed lower bone mass and mineral deposition rates than wild-type mice. Further, MSCs from Igf1rflox/flox mice with Igf1r deleted by a Cre adenovirus in vitro, although recruited to the bone surface after implantation, were unable to differentiate into osteoblasts. We also found that the concentrations of IGF-1 in the bone matrix and marrow of aged rats were lower than in those of young rats and directly correlated with the age-related decrease in bone mass. Likewise, in age-related osteoporosis in humans, we found that bone marrow IGF-1 concentrations were 40% lower in individuals with osteoporosis than in individuals without osteoporosis. Notably, injection of IGF-1 plus IGF binding protein 3 (IGFBP3), but not injection of IGF-1 alone, increased the concentration of IGF-1 in the bone matrix and stimulated new bone formation in aged rats. Together, these results provide mechanistic insight into how IGF-1 maintains adult bone mass, while also providing a further rationale for its therapeutic targeting to treat age-related osteoporosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rizzoli, R., Bianchi, M.L., Garabedian, M., McKay, H.A. & Moreno, L.A. Maximizing bone mineral mass gain during growth for the prevention of fractures in the adolescents and the elderly. Bone 46, 294–305 (2010).
Schettler, A.E. & Gustafson, E.M. Osteoporosis prevention starts in adolescence. J. Am. Acad. Nurse Pract. 16, 274–282 (2004).
Teitelbaum, S.L. Bone resorption by osteoclasts. Science 289, 1504–1508 (2000).
Zaidi, M. Skeletal remodeling in health and disease. Nat. Med. 13, 791–801 (2007).
Agnusdei, D. & Gentilella, R. GH and IGF-I as therapeutic agents for osteoporosis. J. Endocrinol. Invest. 28, 32–36 (2005).
Belfiore, A., Frasca, F., Pandini, G., Sciacca, L. & Vigneri, R. Insulin receptor isoforms and insulin receptor/insulin-like growth factor receptor hybrids in physiology and disease. Endocr. Rev. 30, 586–623 (2009).
Giustina, A., Mazziotti, G. & Canalis, E. Growth hormone, insulin-like growth factors, and the skeleton. Endocr. Rev. 29, 535–559 (2008).
Yakar, S. & Rosen, C.J. From mouse to man: redefining the role of insulin-like growth factor-I in the acquisition of bone mass. Exp. Biol. Med. (Maywood) 228, 245–252 (2003).
Yakar, S., Pennisi, P., Wu, Y., Zhao, H. & LeRoith, D. Clinical relevance of systemic and local IGF-I. Endocr. Dev. 9, 11–16 (2005).
Amin, S. et al. High serum IGFBP-2 is predictive of increased bone turnover in aging men and women. J. Bone Miner. Res. 22, 799–807 (2007).
Ohlsson, C. et al. The role of liver-derived insulin-like growth factor-I. Endocr. Rev. 30, 494–535 (2009).
Seck, T. et al. Concentration of insulin-like growth factor (IGF)-I and -II in iliac crest bone matrix from pre- and postmenopausal women: relationship to age, menopause, bone turnover, bone volume, and circulating IGFs. J. Clin. Endocrinol. Metab. 83, 2331–2337 (1998).
Yamaguchi, T. et al. Serum levels of insulin-like growth factor (IGF); IGF-binding proteins-3, -4, and -5; and their relationships to bone mineral density and the risk of vertebral fractures in postmenopausal women. Calcif. Tissue Int. 78, 18–24 (2006).
Hill, P.A. Bone remodelling. Br. J. Orthod. 25, 101–107 (1998).
Abe, E. et al. TSH is a negative regulator of skeletal remodeling. Cell 115, 151–162 (2003).
Mundy, G.R. & Elefteriou, F. Boning up on ephrin signaling. Cell 126, 441–443 (2006).
Tang, Y. et al. TGF-β1–induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat. Med. 15, 757–765 (2009).
Bautista, C.M., Mohan, S. & Baylink, D.J. Insulin-like growth factors I and II are present in the skeletal tissues of ten vertebrates. Metabolism 39, 96–100 (1990).
Canalis, E., Pash, J., Gabbitas, B., Rydziel, S. & Varghese, S. Growth factors regulate the synthesis of insulin-like growth factor-I in bone cell cultures. Endocrinology 133, 33–38 (1993).
Pfeilschifter, J. et al. Parathyroid hormone increases the concentration of insulin-like growth factor-I and transforming growth factor β 1 in rat bone. J. Clin. Invest. 96, 767–774 (1995).
Hayden, J.M., Mohan, S. & Baylink, D.J. The insulin-like growth factor system and the coupling of formation to resorption. Bone 17, 93S–98S (1995).
Rodda, S.J. & McMahon, A.P. Distinct roles for Hedgehog and canonical Wnt signaling in specification, differentiation and maintenance of osteoblast progenitors. Development 133, 3231–3244 (2006).
Callewaert, F., Sinnesael, M., Gielen, E., Boonen, S. & Vanderschueren, D. Skeletal sexual dimorphism: relative contribution of sex steroids, GH-IGF1, and mechanical loading. J. Endocrinol. 207, 127–134 (2010).
Russell, R.C., Fang, C. & Guan, K.L. An emerging role for TOR signaling in mammalian tissue and stem cell physiology. Development 138, 3343–3356 (2011).
Harrison, D.E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
Selman, C. et al. Ribosomal protein S6 kinase 1 signaling regulates mammalian life span. Science 326, 140–144 (2009).
Liu, L. & Parent, C.A. Review series: TOR kinase complexes and cell migration. J. Cell Biol. 194, 815–824 (2011).
Blüher, M., Kahn, B.B. & Kahn, C.R. Extended longevity in mice lacking the insulin receptor in adipose tissue. Science 299, 572–574 (2003).
Bonjour, J.P., Chevalley, T., Ferrari, S. & Rizzoli, R. The importance and relevance of peak bone mass in the prevalence of osteoporosis. Salud Publica Mex. 51 (suppl. 1), S5–S17 (2009).
Liu, J.M. et al. IGF-1 as an early marker for low bone mass or osteoporosis in premenopausal and postmenopausal women. J. Bone Miner. Metab. 26, 159–164 (2008).
Ohlsson, C. et al. Older men with low serum IGF-1 have an increased risk of incident fractures: the MrOS Sweden study. J. Bone Miner. Res. 26, 865–872 (2011).
Hauschka, P.V., Mavrakos, A.E., Iafrati, M.D., Doleman, S.E. & Klagsbrun, M. Growth factors in bone matrix. Isolation of multiple types by affinity chromatography on heparin-Sepharose. J. Biol. Chem. 261, 12665–12674 (1986).
Lamberts, S.W., van den Beld, A.W. & van der Lely, A.J. The endocrinology of aging. Science 278, 419–424 (1997).
Tatar, M., Bartke, A. & Antebi, A. The endocrine regulation of aging by insulin-like signals. Science 299, 1346–1351 (2003).
Ziv, E. & Hu, D. Genetic variation in insulin/IGF-1 signaling pathways and longevity. Ageing Res. Rev. 10, 201–204 (2011).
Mohan, S. & Baylink, D.J. Serum insulin-like growth factor binding protein (IGFBP)-4 and IGFBP-5 levels in aging and age-associated diseases. Endocrine 7, 87–91 (1997).
Boonen, S. et al. Musculoskeletal effects of the recombinant human IGF-I/IGF binding protein-3 complex in osteoporotic patients with proximal femoral fracture: a double-blind, placebo-controlled pilot study. J. Clin. Endocrinol. Metab. 87, 1593–1599 (2002).
Yakar, S. et al. Normal growth and development in the absence of hepatic insulin-like growth factor I. Proc. Natl. Acad. Sci. USA 96, 7324–7329 (1999).
Kuznetsov, S. & Gehron, R.P. Species differences in growth requirements for bone marrow stromal fibroblast colony formation in vitro. Calcif. Tissue Int. 59, 265–270 (1996).
Di Gregorio, G.B. et al. Attenuation of the self-renewal of transit-amplifying osteoblast progenitors in the murine bone marrow by 17 β-estradiol. J. Clin. Invest. 107, 803–812 (2001).
Acknowledgements
The project described was supported by grant AR 053973 (X.C.) from NIAMS/NIH. J.C. is supported by grant T32DK007751v from the US National Institutes of Health. The authors thank B.J. Canning (Asthma and Allergy Center, Johns Hopkins University) for providing guinea pig marrow cells.
Author information
Authors and Affiliations
Contributions
L.X., X.W., M.L. and L.P. performed the majority of the experiments, analyzed data and prepared the manuscript. T.Q. maintained mice, collected tissue samples and helped with micro-CT analyses. L.P. helped with the in vitro transwell migration assay. J.P.R. finished the human sample detection. X.J. and L.Z. assisted with rat in vivo experiments. J.C., F.F., C.J.R., S.Y., S.X., A.E. and M.W. provided suggestions for the project and critically reviewed the manuscript. X.C. supervised the project and wrote most of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Tables 1–3 and Supplementary Methods (PDF 543 kb)
Rights and permissions
About this article
Cite this article
Xian, L., Wu, X., Pang, L. et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat Med 18, 1095–1101 (2012). https://doi.org/10.1038/nm.2793
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2793
This article is cited by
-
The emerging studies on mesenchymal progenitors in the long bone
Cell & Bioscience (2023)
-
Identification of key ferroptosis-related biomarkers in steroid-induced osteonecrosis of the femoral head based on machine learning
Journal of Orthopaedic Surgery and Research (2023)
-
CD301b+ macrophage: the new booster for activating bone regeneration in periodontitis treatment
International Journal of Oral Science (2023)
-
Lumbar ossification of the ligamentum flavum reflects a strong ossification tendency of the entire spinal ligament
Scientific Reports (2023)
-
Physiology and diseases of tissue-resident macrophages
Nature (2023)