Abstract
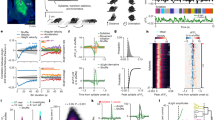
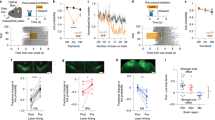
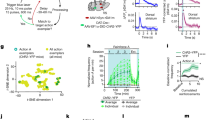
In changing environments, animals must adaptively select actions to achieve their goals. In tasks involving goal-directed action selection, striatal neural activity has been shown to represent the value of competing actions. Striatal representations of action value could potentially bias responses toward actions of higher value. However, no study to date has demonstrated the direct effect of distinct striatal pathways in goal-directed action selection. We found that transient optogenetic stimulation of dorsal striatal dopamine D1 and D2 receptor–expressing neurons during decision-making in mice introduced opposing biases in the distribution of choices. The effect of stimulation on choice was dependent on recent reward history and mimicked an additive change in the action value. Although stimulation before and during movement initiation produced a robust bias in choice behavior, this bias was substantially diminished when stimulation was delayed after response initiation. Together, our data suggest that striatal activity is involved in goal-directed action selection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
30 August 2012
In the version of this article initially published, a reference was not included in Online Methods for the BAC transgenic mice expressing Cre recombinase. The reference is Gong, S. et al. Targeting Cre recombinase to specific neuron populations with bacterial artificial chromosome constructs. J. Neurosci. 27, 9817–9823 (2007). The error has been corrected in the HTML and PDF versions of the article.
References
Lauwereyns, J., Watanabe, K., Coe, B. & Hikosaka, O. A neural correlate of response bias in monkey caudate nucleus. Nature 418, 413–417 (2002).
Cromwell, H.C. & Schultz, W. Effects of expectations for different reward magnitudes on neuronal activity in primate striatum. J. Neurophysiol. 89, 2823–2838 (2003).
Samejima, K., Ueda, Y., Doya, K. & Kimura, M. Representation of action-specific reward values in the striatum. Science 310, 1337–1340 (2005).
Lau, B. & Glimcher, P.W. Value representations in the primate striatum during matching behavior. Neuron 58, 451–463 (2008).
Barnes, T.D., Kubota, Y., Hu, D., Jin, D.Z. & Graybiel, A.M. Activity of striatal neurons reflects dynamic encoding and recoding of procedural memories. Nature 437, 1158–1161 (2005).
Pasupathy, A. & Miller, E.K. Different time courses of learning-related activity in the prefrontal cortex and striatum. Nature 433, 873–876 (2005).
Balleine, B.W., Delgado, M.R. & Hikosaka, O. The role of the dorsal striatum in reward and decision-making. J. Neurosci. 27, 8161–8165 (2007).
Albin, R.L., Young, A.B. & Penney, J.B. The functional anatomy of basal ganglia disorders. Trends Neurosci. 12, 366–375 (1989).
Gerfen, C.R. The neostriatal mosaic: multiple levels of compartmental organization. Trends Neurosci. 15, 133–139 (1992).
DeLong, M.R. Primate models of movement disorders of basal ganglia origin. Trends Neurosci. 13, 281–285 (1990).
Kravitz, A.V. et al. Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature 466, 622–626 (2010).
Lobo, M.K. & Nestler, E.J. The striatal balancing act in drug addiction: distinct roles of direct and indirect pathway medium spiny neurons. Front. Neuroanat. 5, 41 (2011).
Kreitzer, A.C. & Berke, J.D. Investigating striatal function through cell type–specific manipulations. Neuroscience 198, 19–26 (2011).
Kreitzer, A.C. & Malenka, R.C. Striatal plasticity and basal ganglia circuit function. Neuron 60, 543–554 (2008).
Shuen, J.A., Chen, M., Gloss, B. & Calakos, N. Drd1a-tdTomato BAC transgenic mice for simultaneous visualization of medium spiny neurons in the direct and indirect pathways of the basal ganglia. J. Neurosci. 28, 2681–2685 (2008).
Hikosaka, O., Nakamura, K. & Nakahara, H. Basal ganglia orient eyes to reward. J. Neurophysiol. 95, 567–584 (2006).
Isoda, M. & Hikosaka, O. Cortico-basal ganglia mechanisms for overcoming innate, habitual and motivational behaviors. Eur. J. Neurosci. 33, 2058–2069 (2011).
Brainard, M.S. & Doupe, A.J. Interruption of a basal ganglia-forebrain circuit prevents plasticity of learned vocalizations. Nature 404, 762–766 (2000).
Turner, R.S. & Desmurget, M. Basal ganglia contributions to motor control: a vigorous tutor. Curr. Opin. Neurobiol. 20, 704–716 (2010).
Desmurget, M. & Turner, R.S. Motor sequences and the basal ganglia: kinematics, not habits. J. Neurosci. 30, 7685–7690 (2010).
Schwarting, R.K. & Huston, J.P. The unilateral 6-hydroxydopamine lesion model in behavioral brain research. Analysis of functional deficits, recovery and treatments. Prog. Neurobiol. 50, 275–331 (1996).
Thorn, C.A., Atallah, H., Howe, M. & Graybiel, A.M. Differential dynamics of activity changes in dorsolateral and dorsomedial striatal loops during learning. Neuron 66, 781–795 (2010).
Kubota, Y. et al. Stable encoding of task structure coexists with flexible coding of task events in sensorimotor striatum. J. Neurophysiol. 102, 2142–2160 (2009).
Kim, H., Sul, J.H., Huh, N., Lee, D. & Jung, M.W. Role of striatum in updating values of chosen actions. J. Neurosci. 29, 14701–14712 (2009).
Felsen, G. & Mainen, Z.F. Neural substrates of sensory-guided locomotor decisions in the rat superior colliculus. Neuron 60, 137–148 (2008).
Grillner, S., Hellgren, J., Menard, A., Saitoh, K. & Wikstrom, M.A. Mechanisms for selection of basic motor programs–roles for the striatum and pallidum. Trends Neurosci. 28, 364–370 (2005).
Grillner, S., Wallen, P., Saitoh, K., Kozlov, A. & Robertson, B. Neural bases of goal-directed locomotion in vertebrates–an overview. Brain Res. Rev. 57, 2–12 (2008).
Sugrue, L.P., Corrado, G.S. & Newsome, W.T. Choosing the greater of two goods: neural currencies for valuation and decision making. Nat. Rev. Neurosci. 6, 363–375 (2005).
Gold, J.I. & Shadlen, M.N. The neural basis of decision making. Annu. Rev. Neurosci. 30, 535–574 (2007).
Lau, B. & Glimcher, P.W. Dynamic response-by-response models of matching behavior in rhesus monkeys. J. Exp. Anal. Behav. 84, 555–579 (2005).
Sutton, R.S. & Barto, A.G. Reinforcement Learning: an Introduction (MIT Press, Cambridge, Massachusetts, 1998).
Kawaguchi, Y., Wilson, C.J., Augood, S.J. & Emson, P.C. Striatal interneurones: chemical, physiological and morphological characterization. Trends Neurosci. 18, 527–535 (1995).
Rymar, V.V., Sasseville, R., Luk, K.C. & Sadikot, A.F. Neurogenesis and stereological morphometry of calretinin-immunoreactive GABAergic interneurons of the neostriatum. J. Comp. Neurol. 469, 325–339 (2004).
Salzman, C.D., Britten, K.H. & Newsome, W.T. Cortical microstimulation influences perceptual judgements of motion direction. Nature 346, 174–177 (1990).
Jin, X. & Costa, R.M. Start/stop signals emerge in nigrostriatal circuits during sequence learning. Nature 466, 457–462 (2010).
Watanabe, K. & Hikosaka, O. Immediate changes in anticipatory activity of caudate neurons associated with reversal of position-reward contingency. J. Neurophysiol. 94, 1879–1887 (2005).
Redgrave, P., Prescott, T.J. & Gurney, K. The basal ganglia: a vertebrate solution to the selection problem? Neuroscience 89, 1009–1023 (1999).
Mink, J.W. The basal ganglia: focused selection and inhibition of competing motor programs. Prog. Neurobiol. 50, 381–425 (1996).
Jiang, H., Stein, B.E. & McHaffie, J.G. Opposing basal ganglia processes shape midbrain visuomotor activity bilaterally. Nature 423, 982–986 (2003).
Rangel, A., Camerer, C. & Montague, P.R. A framework for studying the neurobiology of value-based decision making. Nat. Rev. Neurosci. 9, 545–556 (2008).
Schultz, W., Dayan, P. & Montague, P.R. A neural substrate of prediction and reward. Science 275, 1593–1599 (1997).
Kimchi, E.Y. & Laubach, M. The dorsomedial striatum reflects response bias during learning. J. Neurosci. 29, 14891–14902 (2009).
Castane, A., Theobald, D.E. & Robbins, T.W. Selective lesions of the dorsomedial striatum impair serial spatial reversal learning in rats. Behav. Brain Res. 210, 74–83 (2010).
Ragozzino, M.E. The contribution of the medial prefrontal cortex, orbitofrontal cortex, and dorsomedial striatum to behavioral flexibility. Ann. NY Acad. Sci. 1121, 355–375 (2007).
Kravitz, A.V., Tye, L.D. & Kreitzer, A.C. Distinct roles for direct and indirect pathway striatal neurons in reinforcement. Nat. Neurosci. 15, 816–818 (2012).
Ikeda, T. & Hikosaka, O. Reward-dependent gain and bias of visual responses in primate superior colliculus. Neuron 39, 693–700 (2003).
Lo, C.C. & Wang, X.J. Cortico-basal ganglia circuit mechanism for a decision threshold in reaction time tasks. Nat. Neurosci. 9, 956–963 (2006).
Kable, J.W. & Glimcher, P.W. The neurobiology of decision: consensus and controversy. Neuron 63, 733–745 (2009).
Redgrave, P., Prescott, T.J. & Gurney, K. Is the short-latency dopamine response too short to signal reward error? Trends Neurosci. 22, 146–151 (1999).
Hikosaka, O. & Isoda, M. Switching from automatic to controlled behavior: cortico-basal ganglia mechanisms. Trends Cogn. Sci. 14, 154–161 (2010).
Sparta, D.R. et al. Construction of implantable optical fibers for long-term optogenetic manipulation of neural circuits. Nat. Protoc. 7, 12–23 (2012).
Ariano, M.A. et al. Striatal potassium channel dysfunction in Huntington's disease transgenic mice. J. Neurophysiol. 93, 2565–2574 (2005).
Gong, S. et al. Targeting Cre recombinase to specific neuron populations with bacterial artificial chromosome constructs. J. Neurosci. 27, 9817–9823 (2007).
Acknowledgements
We thank G. Stuber, F. Ambroggi, D. Niell, K. Berger, C. Wu and H. Peckler for assistance with experiments, V. Kharazia for immunostaining and histological quantification, K. Deisseroth (Stanford University) for advice and reagents, and C. Niell for comments on the manuscript. This work was supported by the State of California, the US National Institutes of Health (1RC2NS069350; R01MH087542 and R01DA029150), and the P. Royer and K. Clayton Family.
Author information
Authors and Affiliations
Contributions
L.-H.T., A.M.L. and L.W. designed the study. L.-H.T., A.M.L. and N.B. collected behavioral data. L.-H.T. and A.M.L. analyzed and modeled data. A.M.L. and N.B. processed tissue. L.-H.T., A.M.L., L.W. and A.B. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 (PDF 4701 kb)
Rights and permissions
About this article
Cite this article
Tai, LH., Lee, A., Benavidez, N. et al. Transient stimulation of distinct subpopulations of striatal neurons mimics changes in action value. Nat Neurosci 15, 1281–1289 (2012). https://doi.org/10.1038/nn.3188
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3188
This article is cited by
-
Reward expectation enhances action-related activity of nigral dopaminergic and two striatal output pathways
Communications Biology (2023)
-
Error-related signaling in nucleus accumbens D2 receptor-expressing neurons guides inhibition-based choice behavior in mice
Nature Communications (2023)
-
Dopamine and glutamate regulate striatal acetylcholine in decision-making
Nature (2023)
-
Neuronal and astrocytic CB1R signaling differentially modulates goal-directed behavior and working memory by distinct temporal mechanisms
Neuropsychopharmacology (2023)
-
Orbitofrontal cortex control of striatum leads economic decision-making
Nature Neuroscience (2023)