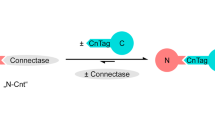
Abstract
Far western blotting (WB) was derived from the standard WB method to detect protein–protein interactions in vitro. In Far WB, proteins in a cell lysate containing prey proteins are firstly separated by SDS or native PAGE, and transferred to a membrane, as in a standard WB. The proteins in the membrane are then denatured and renatured. The membrane is then blocked and probed, usually with purified bait protein(s). The bait proteins are detected on spots in the membrane where a prey protein is located if the bait proteins and the prey protein together form a complex. Compared with other biochemical binding assays, Far WB allows prey proteins to be endogenously expressed without purification. Unlike most methods using cell lysates (e.g., co-immunoprecipitation (co-IP)) or living cells (e.g., fluorescent resonance energy transfer (FRET)), Far WB determines whether two proteins bind to each other directly. Furthermore, in cases where they bind to each other indirectly, Far WB allows the examination of candidate protein(s) that form a complex between them. Typically, 2–3 d are required to carry out the experiment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Fu, H. Protein-Protein Interactions: Method and Application. (Humana Press, Totowa, New Jersey, 2004).
Golemis, E.A. & Adams, P.D. Protein–Protein Interactions: A Molecular Cloning Manual 2nd edn. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2005).
Protein interaction technologies. In Molecular Cloning: A Laboratory Manual 3rd edn. Ed: Sambrook, J. & Russell, D.W. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2001).
Dmitrova, M. et al. A new LexA-based genetic system for monitoring and analyzing protein heterodimerization in Escherichia coli. Mol. Gen. Genet. 257, 205–212 (1998).
Vasavada, H.A., Ganguly, S., Germino, F.J., Wang, Z.X. & Weissman, S.M. A contingent replication assay for the detection of protein-protein interactions in animal cells. Proc. Natl. Acad. Sci. USA 88, 10686–10690 (1991).
Fields, S. & Song, O. A novel genetic system to detect protein-protein interactions. Nature 340, 245–246 (1989).
Gordon, G.W., Berry, G., Liang, X.H., Levine, B. & Herman, B. Quantitative fluorescence resonance energy transfer measurements using fluorescence microscopy. Biophys. J. 74, 2702–2713 (1998).
Pfleger, K.D., Seeber, R.M. & Eidne, K.A. Bioluminescence resonance energy transfer (BRET) for the real-time detection of protein-protein interactions. Nat. Protoc. 1, 337–345 (2006).
Ye, P. et al. Gene function prediction from congruent synthetic lethal interactions in yeast. Mol. Syst. Biol. 1, 2005 (2005).
Rigaut, G. et al. A generic protein purification method for protein complex characterization and proteome exploration. Nat. Biotechnol. 17, 1030–1032 (1999).
Smith, E.A. & Corn, R.M. Surface plasmon resonance imaging as a tool to monitor biomolecular interactions in an array based format. Appl. Spectrosc. 57, 320A–332A (2003).
Jones, R.B., Gordus, A., Krall, J.A. & MacBeath, G. A quantitative protein interaction network for the ErbB receptors using protein microarrays. Nature 439, 168–174 (2006).
Hall, R.A. Studying protein-protein interactions via blot overlay or far western blot. In Protein–Protein Interactions: Methods and Applications. p. 167–174 (ed. Fu, H.) (Humana Press, Totowa, New Jersey, 2004).
Vikis, H.G. & Guan, K.L. Glutathione-S-transferase-fusion based assays for studying protein-protein interactions. In Protein–Protein Interactions: Methods and Applications. p. 175–186 (ed. Fu, H.) (Humana Press, Totowa, New Jersey, 2004).
Wang, B., Pelletier, J., Massaad, M.J., Herscovics, A. & Shore, G.C. The yeast split-ubiquitin membrane protein two-hybrid screen identifies BAP31 as a regulator of the turnover of endoplasmic reticulum-associated protein tyrosine phosphatase-like B. Mol. Cell Biol. 24, 2767–2778 (2004).
Kaelin, W.G. Jr. et al. Expression cloning of a cDNA encoding a retinoblastoma-binding protein with E2F-like properties. Cell 70, 351–364 (1992).
Blackwood, E.M. & Eisenman, R.N. Max: a helix-loop-helix zipper protein that forms a sequence-specific DNA-binding complex with Myc. Science 251, 1211–1217 (1991).
Kaido, M. et al. Downregulation of the NbNACa1 gene encoding a movement-protein-interacting protein reduces cell-to-cell movement of Brome mosaic virus in Nicotiana benthamiana. Mol. Plant Microbe Interact. 20, 671–681 (2007).
Cantor, S.B. et al. BACH1, a novel helicase-like protein, interacts directly with BRCA1 and contributes to its DNA repair function. Cell 105, 149–160 (2001).
Gupta, R. et al. FANCJ (BACH1) helicase forms DNA damage inducible foci with replication protein A and interacts physically and functionally with the single-stranded DNA binding protein. Blood 110, 2390–2398 (2007).
Feldman, M., Zusman, T., Hagag, S. & Segal, G. Coevolution between nonhomologous but functionally similar proteins and their conserved partners in the Legionella pathogenesis system. Proc. Natl. Acad. Sci. USA 102, 12206–12211 (2005).
Wu, Y. et al. Kinesin-2 mediates physical and functional interactions between polycystin-2 and fibrocystin. Hum. Mol. Genet. 15, 3280–3292 (2006).
Cutler, P. Protein Purification Protocols. (Humana Press, Totowa, New Jersey, 2003).
Sambrook, J., Fritsch, E.F. & Maniatis, T. Detection and analysis of proteins expressed from cloned genes. In Molecular Cloning: A laboratory Manual 2nd edn. 18.19–18.74 (Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, 1989).
Acknowledgements
This work was supported by the Canadian Institutes of Health Research and the Kidney Foundation of Canada. X.-Z.C. is a Senior Scholar of the Alberta Heritage Foundation for Medical Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Y., Li, Q. & Chen, XZ. Detecting protein–protein interactions by far western blotting. Nat Protoc 2, 3278–3284 (2007). https://doi.org/10.1038/nprot.2007.459
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.459
This article is cited by
-
MGCG regulates glioblastoma tumorigenicity via hnRNPK/ATG2A and promotes autophagy
Cell Death & Disease (2023)
-
A recombinant Aspergillus oryzae fungus transmitted from larvae to adults of Anopheles stephensi mosquitoes inhibits malaria parasite oocyst development
Scientific Reports (2023)
-
Transglutaminase 2 associated with PI3K and PTEN in a membrane-bound signalosome platform blunts cell death
Cell Death & Disease (2023)
-
Bacillus subtilis biofilm matrix components target seed oil bodies to promote growth and anti-fungal resistance in melon
Nature Microbiology (2022)
-
Biochemical and cytological interactions between callose synthase and microtubules in the tobacco pollen tube
Plant Cell Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.