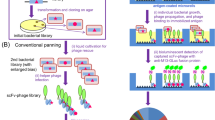
Abstract
The demand for monoclonal antibodies (mAbs) in biomedical research is significant, but the current methodologies used to discover them are both lengthy and costly. Consequently, the diversity of antibodies available for any particular antigen remains limited. Microengraving is a soft lithographic technique that provides a rapid and efficient alternative for discovering new mAbs. This protocol describes how to use microengraving to screen mouse hybridomas to establish new cell lines producing unique mAbs. Single cells from a polyclonal population are isolated into an array of microscale wells (∼105 cells per screen). The array is then used to print a protein microarray, where each element contains the antibodies captured from individual wells. The antibodies on the microarray are screened with antigens of interest, and mapped to the corresponding cells, which are then recovered from their microwells by micromanipulation. Screening and retrieval require approximately 1–3 d (9–12 d including the steps for preparing arrays of microwells).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Barrett, W. & Newsome, M.S.E. The clinical pharmacology of therapeutic monoclonal antibodies in the treatment of malignancy; have the magic bullets arrived? Br. J. Clin. Pharmacol. 66, 6–19 (2008).
Finn, O.J. Molecular origins of cancer—cancer immunology. N. Engl. J. Med. 358, 2704–2715 (2008).
Buttmann, M. & Rieckmann, P. Treating multiple sclerosis with monoclonal antibodies. Exp. Rev. Neurotherapeutics 8, 433–455 (2008).
Jennifer, M.D. & Stuart, M.L. Prospects for development of vaccines against fungal diseases. Drug Resist. Updat. 9, 105–110 (2006).
Keller, M.A. & Stiehm, E.R. Passive immunity in prevention and treatment of infectious diseases. Clin. Microbiol. Rev. 13, 602–614 (2000).
Editorial. Elective affinities. Nat. Methods 5, 851 (2008).
Uhlen, M., Gräslund, S. & Sundström, M. A pilot project to generate affinity reagents to human proteins. Nat. Methods 5, 854–855 (2008).
Dessain, S.K. et al. High efficiency creation of human monoclonal antibody-producing hybridomas. J. Immunol. Methods 291, 109–122 (2004).
Kohler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975).
Shulman, M., Wilde, C.D. & Kohler, G. A better cell line for making hybridomas secreting specific antibodies. Nature 276, 269–270 (1978).
Clackson, T., Hoogenboom, H.R., Griffiths, A.D. & Winter, G. Making antibody fragments using phage display libraries. Nature 352, 624–628 (1991).
Boder, E.T. & Wittrup, K.D. Yeast surface display for screening combinatorial polypeptide libraries. Nat. Biotech. 15, 553–557 (1997).
Fuchs, P., Breitling, F., Dubel, S., Seehaus, T. & Little, M. Targeting recombinant antibodies to the surface of Escherichia-coli—fusion to a peptidoglycan associated lipoprotein. Biotechnology 9, 1369–1372 (1991).
Lee, C.M.Y., Iorno, N., Sierro, F. & Christ, D. Selection of human antibody fragments by phage display. Nat. Protoc. 2, 3001–3008 (2007).
Chao, G. et al. Isolating and engineering human antibodies using yeast surface display. Nat. Protoc. 1, 755–768 (2006).
Love, J.C., Ronan, J.L., Grotenbreg, G.M., van der Veen, A.G. & Ploegh, H.L. A microengraving method for rapid selection of single cells producing antigen-specific antibodies. Nat. Biotechnol. 24, 703–707 (2006).
Bradshaw, E.M. et al. Concurrent detection of secreted products from human lymphocytes by microengraving: antigen-reactive antibodies and cytokines. Clin. Immunol. 129, 10–18 (2008).
Story, C.M. et al. Profiling antibody responses by multiparametric analysis of primary B cells. Proc. Natl Acad. Sci. USA 105, 17902–17907 (2008).
Ronan, J.L., Story, C.M., Papa, E. & Love, J.C. Optimization of the surfaces used to capture antibodies from single hybridomas reduces the time required for microengraving. J. Immunol. Methods 340, 164–169 (2009).
Coligan, J.E. (Greene Pub. Associates and Wiley-Interscience, New York, 1992).
Love, J.C., Wolfe, D.B., Gates, B.D. & Whitesides, G.M. McGraw Hill Yearbook of Science and Technology 323–326 (McGraw-Hill, New York, 2004).
Kane, R.S., Takayama, S., Ostuni, E., Ingber, D.E. & Whitesides, G.M. Patterning proteins and cells using soft lithography. Biomaterials 20, 2363–2376 (1999).
Park, J.W., Vahidi, B., Taylor, A.M., Rhee, S.W. & Jeon, N.L. Microfluidic culture platform for neuroscience research. Nat. Protoc. 1, 2128–2136 (2006).
Tourovskaia, A., Figueroa-Masot, X. & Folch, A. Long-term microfluidic cultures of myotube microarrays for high-throughput focal stimulation. Nat. Protoc. 1, 1092–1104 (2006).
Harlow, E. & Lane, D. Antibodies: A Laboratory Manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, USA, 1988).
Espina, V. et al. Protein microarrays: molecular profiling technologies for clinical specimens. Proteomics 3, 2091–2100 (2003).
Saff, D. & Sacilotto, D. Printmaking: History and Process (Holt, Rinehart, Winston, New York, USA, 1978).
MacBeath, G. & Schreiber, S.L. Printing proteins as microarrays for high-throughput function determination. Science 289, 1760–1763 (2000).
Kramer, S., Joos, T.O. & Templin, M.F. Protein Microarrays. In Current Protocols in Protein Science (eds. J.E. Coligan, B.M. Dunn, D.W. Speicher & P.T. Wingfield) (John Wiley & Sons, Inc., New York, USA, 2005).
Engvall, E. & Perlmann, P. Enzyme-linked immunosorbent assay, Elisa: III. quantitation of specific antibodies by enzyme-labeled anti-immunoglobulin in antigen-coated tubes. J. Immunol. 109, 129–135 (1972).
Kovac, J.R. & Voldman, J. Intuitive, image-based cell sorting using optofluidic cell sorting. Anal. Chem. 79, 9321–9330 (2007).
Simon, K., Lingappa, V.R. & Ganem, D. Secreted hepatitis-B surface-antigen polypeptides are derived from a transmembrane precursor. J. Cell Biol. 107, 2163–2168 (1988).
Acknowledgements
This research was supported by the Eli and Edythe L. Broad Institute, and used facilities at the Center for Nanoscale Systems at Harvard University supported by the NSF under the National Nanotechnology Infrastructure Network, and services at the Stanford Microfluidics Foundry. E.P. was an NSERC postgraduate fellow. C.M.S. thanks Gordon College for sabbatical leave. The authors thank J. Ronan and J.H. Choi for helpful discussions on this protocol.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ogunniyi, A., Story, C., Papa, E. et al. Screening individual hybridomas by microengraving to discover monoclonal antibodies. Nat Protoc 4, 767–782 (2009). https://doi.org/10.1038/nprot.2009.40
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2009.40
This article is cited by
-
Predicting unseen antibodies’ neutralizability via adaptive graph neural networks
Nature Machine Intelligence (2022)
-
Single-cell sorting based on secreted products for functionally defined cell therapies
Microsystems & Nanoengineering (2022)
-
Microfluidic Single-cell Trapping and Cultivation for the Analysis of Host-viral Interactions
Biotechnology and Bioprocess Engineering (2021)
-
Development of therapeutic antibodies for the treatment of diseases
Journal of Biomedical Science (2020)
-
A digital microfluidic system with 3D microstructures for single-cell culture
Microsystems & Nanoengineering (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.