Abstract
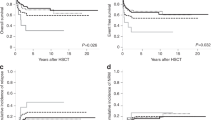
To examine whether the percentage of myeloperoxidase (MPO)-positive blast cells is useful as a prognostic factor for acute myeloid leukemia (AML), cytochemical analysis of MPO was performed in 491 patients who were registered to the Japan Adult Leukemia Study Group-AML92 study. Patients were divided into two using the percentage of MPO-positive blast (high [⩾50%] and low (<50%)). Complete remission rates were 85.4% in the former and 64.1% in the latter (P=0.001). The overall survival (OS) and the disease-free survival (DFS) were significantly better in the high MPO group (48.3 vs 18.7% for OS, and 36.3 vs 20.1% for DFS, P<0.001, respectively). Multivariate analysis showed that both karyotype and the percentage of MPO-positive blast cells were equally important prognostic factors. The high MPO group still showed a better survival even when restricted to the intermediate chromosomal risk group or the patients with normal karyotype (P<0.001). The OS of patients with normal karyotype in the high MPO group was almost equal with that of the favorable chromosomal risk group. The percentage of MPO-positive blast cells is a simple and highly significant prognostic factor for AML patients, and especially useful to stratify patients with normal karyotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zaki SR, Austin GE, Swan D, Srinivasan A, Ragab AH, Chan WC . Human myeloperoxidase gene expression in acute leukemia. Blood 1989; 74: 2096–2102.
Lubbert M, Oster W, Ludwig WD, Ganser A, Mertelsmann R, Herrmann F . A switch toward demethylation is associated with the expression of myeloperoxidase in acute myeloblastic and promyelocytic leukemias. Blood 1992; 80: 2066–2073.
Austin GE, Lam L, Zaki SR, Chan WC, Hodge T, Hou J et al. Sequence comparison of putative regulatory DNA of the 5′ flanking region of the myeloperoxidase gene in normal and leukemic bone marrow cells. Leukemia 1993; 7: 1445–1450.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposals for the classification of acute leukeamias. French–Amirican–British (FAB) co-operative group. Br J Haematol 1976; 33: 451–458.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. A variant form of hypergranular promyelocytic leukaemia (M3). Br J Haematol 1980; 44: 169–170.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. The morphological classification of acute lymphoblastic leukaemia. Concordance among observers and clinical correlations. Br J Haematol 1981; 47: 553–561.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposed revised criteria for the classification of acute myeloid leukemia. A report of the French–American–British Cooperative Group. Ann Intern Med 1985; 103: 620–625.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Criteria for the diagnosis of acute leukemia of megakaryocyte lineage (M7). Ann Intern Med 1985; 103: 460–462.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposal for the recognition of minimally differentiated acute myeloid leukaemia (AML-M0). Br J Haematol 1991; 78: 325–329.
Takubo T, Kubota Y, Oguma S, Ueda T, Shibata H, Nakamura H et al. Classification of acute non-lymphocytic leukemia according to the distribution picture of peroxidase activity and cell size: correlation between the classification and therapeutic response. Blood Cells 1983; 9: 501–514.
Matsuo T, Cox C, Bennett JM . Prognostic significance of myeloperoxidase positivity of blast cells in acute myeloblastic leukemia without maturation (FAB: M1): An ECOG study. Hematol Pathol 1989; 3: 153–158.
Suic M, Boban D, Markovic-Glamocak M, Petrovecki M, Marusic M, Labar B . Prognostic significance of cytochemical analysis of leukemic M2 blasts. Med Oncol Tumor Pharmacother 1992; 9: 41–45.
Kuriyama K, Tomonaga M, Kobayashi T, Takeuchi J, Ohshima T, Furusawa S et al. Morphological diagnoses of the Japan adult leukemia study group acute myeloid leukemia protocols: central review. Int J Hematol 2001; 73: 93–99.
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G et al. The importance of diagnostic cytogenetics on outcome in AML: analysis of 1612 patients entered into the MRC AML 10 trial. Blood 1998; 92: 2322–2333.
Miyawaki S, Tanimoto M, Kobayashi T, Minami S, Tamura J, Omoto E et al. No beneficial effect from addition of etoposide to daunorubicin, cytarabine, and 6-mercaptopurine in individualized induction therapy of adult acute myeloid leukemia: the JALSG-AML92 study. Int J Hematol 1999; 70: 97–104.
Kuriyama K, Tomonaga M, Matsuo T, Kobayashi T, Miwa H, Shirakawa S et al. Poor response to intensive chemotherapy in de novo acute myeloid leukaemia with trilineage myelodysplasia. Br J Haematol 1994; 86: 767–773.
Shibata A, Bennett JM, Castoldi GL, Catovsky D, Flandrin G, Jaffe ES et al. Recommended methods for cytological procedures in haematology. International Committee for Standardization in Haematology (ICSH). Clin Lab Heamatol 1985; 7: 55–74.
SAS Institute. SAS/Stat User's Guide, Version 6, 4th edn. Cary, NC: SAS Institute, 1990.
Hoyle CF, Gray RG, Wheatley K, Swirsky D, de Bastos M, Sherrington P et al. Prognostic importance of Sudan Black positivity: a study of bone marrow slides from 1386 patients with de novo acute myeloid leukaemia. Br J Haematol 1991; 79: 398–407.
Venditti A, Del Poeta G, Buccisano F, Tamburini A, Cox MC, Stasi R et al. Minimally differentiated acute myeloid leukemia (AML-M0): comparison of 25 cases with other French–American–British subtypes. Blood 1997; 89: 621–629.
Campos L, Guyotat D, Archimbaud E, Calmard-Oriol P, Tsuruo T, Troncy J et al. Clinical significance of multidrug resistance P-glycoprotein expression on acute nonlymphoblastic leukemia cells at diagnosis. Blood 1992; 79: 473–476.
Legrand O, Perrot JY, Tang R, Simonin G, Gurbuxani S, Zittoun R et al. Pgp and MRP activities using calcein-AM are prognostic factors in adult acute myeloid leukemia patients. Blood 1998; 91: 4480–4488.
Leith CP, Kopecky KJ, Chen IM, Eijdems L, Slovak ML, McConnell TS et al. Frequency and clinical significance of the expression of the multidrug resistance proteins MDR1/P-glycoprotein, MRP1, and LRP in acute myeloid leukemia. A Southwest Oncology Group Study. Blood 1999; 94: 1086–1099.
Leith CP, Kopecky KJ, Godwin J, McConnell T, Slovak ML, Chen IM et al. Acute myeloid leukemia in the elderly: assessment of multidrug resistance (MDR1) and cytogenetics distinguishes biological subgroups with remarkably distinct responses to standard chemotherapy. Blood 1997; 89: 3323–3329.
Senent L, Jarque I, Martin G, Sempere A, Gonzalez-Garcia Y, Gomis F et al. P-glycoprotein expression and prognostic value in acute myeloid leukemia. Haematologica 1998; 83: 783–787.
Look AT . Oncogenic transcription factors in the human acute leukemias. Science 1997; 278: 1059–1064.
Senent L, Jarque I, Martin G, Sempere A, Gonzalez-Garcia Y, Gomis F et al. An allelic association implicates myeloperoxidase in the etiology of acute promyelocytic leukemia. Blood 1997; 90: 2730–2737.
Bruno JG, Herman TS, Cano VL, Stribling L, Kiel JL . Selective cytotoxicity of 3-amino-L-tyrosine correlates with peroxidase activity. In Vitro Cell Dev Biol Anim 1999; 35: 376–382.
Wagner BA, Buettner GR, Oberley LW, Darby CJ, Burns CP . Myeloperoxidase is involved in H2O2-induced apoptosis of HL-60 human leukemia cells. J Biol Chem 2000; 275: 22461–22469.
Acknowledgements
We appreciate all physicians who participated in this trial. We thank Drs J Taguchi and T Fukushima for providing assistance. This work was supported in part by the grant from the Ministry of Health, Labour and Welfare of Japan.
Author information
Authors and Affiliations
Consortia
Rights and permissions
About this article
Cite this article
Matsuo, T., Kuriyama, K., Miyazaki, Y. et al. The percentage of myeloperoxidase-positive blast cells is a strong independent prognostic factor in acute myeloid leukemia, even in the patients with normal karyotype. Leukemia 17, 1538–1543 (2003). https://doi.org/10.1038/sj.leu.2403010
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403010
Keywords
This article is cited by
-
Circulating cytokines and small molecules follow distinct expression patterns in acute myeloid leukaemia
British Journal of Cancer (2017)
-
Association of −463G/A MPO gene polymorphism and risk of cervical intraepithelial neoplasia
Archives of Gynecology and Obstetrics (2016)
-
Expression of myeloperoxidase in acute myeloid leukemia blasts mirrors the distinct DNA methylation pattern involving the downregulation of DNA methyltransferase DNMT3B
Leukemia (2014)
-
Expression of myeloperoxidase and gene mutations in AML patients with normal karyotype: double CEBPA mutations are associated with high percentage of MPO positivity in leukemic blasts
International Journal of Hematology (2011)
-
Expression of myeloperoxidase enhances the chemosensitivity of leukemia cells through the generation of reactive oxygen species and the nitration of protein
Leukemia (2008)