Abstract
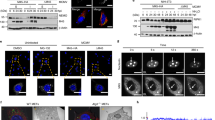
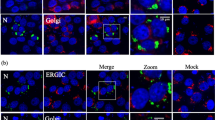
The interferon-induced promyelocytic leukaemia (PML) protein localizes both in the nucleoplasm and in matrix-associated multi-protein complexes known as nuclear bodies (NBs). NBs are disorganized in acute promyelocytic leukaemia or during some viral infections, suggesting that PML NBs could be a part of cellular defense mechanism. Rabies virus, a member of the rhabdoviridae family, replicates in the cytoplasm. Rabies phosphoprotein P and four other amino-terminally truncated products (P2, P3, P4, P5) are all translated from P mRNA. P and P2 are located in the cytoplasm, whereas P3, P4 and P5 are found mostly in the nucleus. Infection with rabies virus reorganized PML NBs. PML NBs became larger and appeared as dense aggregates when analysed by confocal or electron microscopy, respectively. The expression of P sequesters PML in the cytoplasm where both proteins colocalize, whereas that of P3 results in an increase in PML body size, as observed in infected cells. The P and P3 interacted directly in vivo and in vitro with PML. The C-terminal domain of P and the PML RING finger seem to be involved in this binding. Moreover, PML−/− primary mouse embryonic fibroblasts expressed viral proteins at a higher level and produced 20 times more virus than wild-type cells, suggesting that the absence of all PML isoforms resulted in an increase in rabies virus replication.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- NBs:
-
nuclear bodies
- PML:
-
promyelocytic leukaemia
- VSV:
-
vesicular stomatitis virus
- M.O.I.:
-
multiplicity of infection
- IFN:
-
interferon
- SUMO:
-
small ubiquitin modifier
References
Ahn J, Jang W, Hayward G . 1999 J. Virol. 73: 10458–10471
Bell P, Lieberman PM, Maul GG . 2000 J. Virol. 74: 11800–11810
Bonilla WV, Pinschewer DD, Klenerman P, Rousson V, Gaboli M, Pandolfi P, Zinkernagel RM, Salvato MS, Hengartner H . 2002 J. Virol. 76: 3810–3818
Borden KLB, Campbell-Dwyer EJ, Salvato MS . 1998 J. Virol. 72: 758–766
Bourhy H, Kissi B, Tordo N . 1993 Virology 194: 70–81
Carvalho T, Seeler J, Ohman K, Jordan P, Petterson U, Akusjarvi G, Carmo-Fonseca M, Dejean A . 1995 J. Biol. Cell. 131: 45–56
Chelbi-Alix MK, de Thé H . 1999 Oncogene 18: 935–941
Chelbi-Alix MK, Pelicano L, Quignon F, Koken M, Stadler M, Pavlovic J, Degos L, de Thé H . 1995 Leukemia 9: 2027–2033
Chelbi-Alix MK, Quignon F, Pelicano L, Koken MHM, de Thé H . 1998 J. Virol. 72: 1043–1051
Chenik M, Chebli K, Blondel D . 1995 J. Virol. 69: 707–712
Chenik M, Chebli K, Gaudin Y, Blondel D . 1994 J. Gen. Virol. 75: 2889–2896
Chenik M, Schnell M, Conzelmann KK, Blondel D . 1998 J. Virol. 72: 1925–1930
Daniel M-T, Koken M, Romagné O, Barbey S, Bazarbachi A, Stadler M, Guillemin MC, Degos L, Chomienne C, de Thé H . 1993 Blood 82: 1858–1867
de Thé H, Lavau C, Marchio A, Chomienne C, Degos L, Dejean A . 1991 Cell 66: 675–684
Djavani M, Rodas J, Lukashevich I, Horejsh D, Pandolfi P, Borden K, Salvato M . 2001 J. Virol. 75: 6204–6208
Duprez E, Saurin AJ, Desterro JM, Lallemand-Breitenbach V, Howe K, Boddy MN, Solomon E, de Thé H, Hay RT, Freemont PS . 1999 J. Cell Sci. 112: 381–393
Dyck JA, Maul GG, Miller WH, Chen JD, Kakizuka A, Evans RM . 1994 Cell 76: 333–343
Everett RD . 2001 Oncogene 20: 7266–7273
Everett RD, Freemont P, Saitoh H, Dasso M, Orr A, Kathoria M, Parkinson J . 1998 J. Virol. 72: 6581–6591
Gaudin Y, Moreira S, Bénéjean J, Flamand A, Tuffereau C . 1999 J. Gen. Virol. 80: 1647–1656
Gaudin Y, Ruigrok R, Tuffereau C, Knossow M, Flamand A . 1992 Virology 187: 627–732
Gupta A, Blondel D, Choudhary S, Banerjee A . 2000 J. Virol. 74: 91–98
Goddard AD, Borrow J, Freemont PS, Solomon E . 1991 Science 254: 1371–1374
Goodbourn S, Didcock L, Randall R . 2000 J. Gen. Virol. 81: 2341–2364
Ishov A, Sotnikov A, Negorev D, Vladimirova O, Neff N, Kamitani T, Yeh E, Strauss III J, Maul G . 1999 J. Cell Biol. 147: 221–223
Jacob Y, Real E, Tordo N . 2001 J. Virol. 75: 9613–9622
Jensen K, Shiels C, Freemont PS . 2001 Oncogene 20: 7223–7233
Kakizuka A, Miller Jr W, Umesono K, Warrell Jr R, Frankel SR, Murty VV, Dmitrovsky E, Evans RM . 1991 Cell 66: 663–674
Koken MHM, Puvion-Dutilleul F, Guillemin MC, Viron A, Linares-Cruz G, Stuurman N, de Jong L, Szostecki C, Calvo F, Chomienne C, Degos L, Puvion E, de Thé H . 1994 EMBO J. 13: 1073–1083
Lallemand-Breitenbach V, Zhu J, Puvion F, Koken M, Honore N, Doubeikovsky A, Duprez E, Pandolfi PP, Puvion E, Freemont P, de Thé H . 2001 J. Exp. Med. 193: 1361–1371
Muller S, Dejean A . 1999 J. Virol. 73: 5137–5143
Muller S, Matunis MJ, Dejean A . 1998 EMBO J. 17: 61–70
Negorev D, Maul G . 2001 Oncogene 20: 7234–7242
Pandolfi PP, Grignani F, Alcalay M, Mencarelli A, Biondi A, LoCoco F, Grignani F, Pelicci PG . 1991 Oncogene 6: 1285–1292
Puvion-Dutilleul F, Chelbi-Alix M, Koken M, Quignon F, Puvion E, de Thé H . 1995 Exp. Cell. Res. 218: 9–16
Puvion-Dutilleul F, Legrand V, Mehtali M, Chelbi-Alix MK, de Thé H, Puvion E . 1999 Biol. Cell 91: 617–628
Raux H, Iseni F, Lafay F, Blondel D . 1997 J. Gen. Virol. 78: 119–124
Regad T, Chelbi-Alix MK . 2001 Oncogene 20: 7274–7286
Regad T, Saib A, Lallemand-Breitenbach V, Pandolfi PP, de Thé H, Chelbi-Alix MK . 2001 EMBO J. 20: 3495–3505
Rose JK, Whitt MA . 2001 Fields Virology 4th edn Knipe DM and Howley PM (eds) Philadelphia: Lippincott Williams and Wilkins pp 1221–1244
Seeler J, Dejean A . 2001 Oncogene 20: 7243–7249
Stadler M, Chelbi-Alix MK, Koken MHM, Venturini L, Lee C, Saïb A, Quignon F, Pelicano L, Guillemin M-C, Schindler C, de Thé H . 1995 Oncogene 11: 2565–2573
Stark GR, Kerr IM, William BRG, Silverman RH, Schreiber RD . 1998 Annu. Rev. Biochem. 67: 227–264
Szekely L, Pokrovskaja K, Jiang W, de Thé H, Ringertz N, Klein G . 1996 J. Virol. 70: 2562–2568
Wang Z-G, Ruggero D, Ronchetti S, Zhong S, Gaboli M, Rivi R, Pandolfi PP . 1998 Nature Genet. 20: 266–272
Weis K, Rambaud S, Lavau C, Jansen J, Carvalho T, Carmo-Fonseca M, Lamond A, Dejean A . 1994 Cell 76: 345–356
Acknowledgements
We acknowledge E Duprez and P Freemont for the RING finger PML Cys 57,60 mutant. The help of M Schmid and Z Mishal with the confocal microscopy analysis was greatly appreciated. We would like to thank Magalis Berges for technical assistance. This work was supported by grants from the Centre National pour la Recherche Scientifique, the Association pour la Recherche sur le Cancer (ARC) and the Fondation de la Recherche Médicale. T Regad was supported by a fellowship from ARC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blondel, D., Regad, T., Poisson, N. et al. Rabies virus P and small P products interact directly with PML and reorganize PML nuclear bodies. Oncogene 21, 7957–7970 (2002). https://doi.org/10.1038/sj.onc.1205931
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205931
Keywords
This article is cited by
-
Rabies virus phosphoprotein P5 binding to BECN1 regulates self-replication by BECN1-mediated autophagy signaling pathway
Cell Communication and Signaling (2020)
-
1H, 15N and 13C resonance assignments of the C-terminal domain of the P protein of the Nishigahara strain of rabies virus
Biomolecular NMR Assignments (2019)
-
Cholesterol 25-hydroxylase suppresses rabies virus infection by inhibiting viral entry
Archives of Virology (2019)
-
Proteomic analysis of purified turkey adenovirus 3 virions
Veterinary Research (2015)
-
Rabies virus phosphoprotein interacts with mitochondrial Complex I and induces mitochondrial dysfunction and oxidative stress
Journal of NeuroVirology (2015)