Abstract
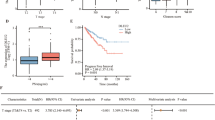
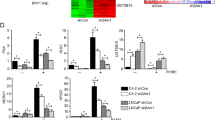
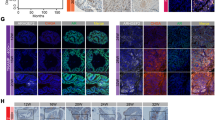
Glucocorticoids are extensively used in combination chemotherapy of advanced prostate cancer (PC). Little is known, however, about the status of the glucocorticoid receptor (GR) in PC. We evaluated over 200 prostate samples and determined that GR expression was strongly decreased or absent in 70–85% of PC. Similar to PC tumors, some PC cell lines, including LNCaP, also lack GR. To understand the role of GR, we reconstituted its expression in LNCaP cells using lentiviral approach. Treatment of LNCaP-GR cells with the glucocorticoids strongly inhibited proliferation in the monolayer cultures and blocked anchorage-independent growth. This was accompanied by upregulation of p21 and p27, down-regulation of cyclin D1 expression and c-Myc phosphorylation. Importantly, the activation of GR resulted in normalized expression of PC markers hepsin, AMACR, and maspin. On the signaling level, GR decreased expression and inhibited activity of the MAP-kinases (MAPKs) including p38, JNK/SAPK, Mek1/2 and Erk1/2. We also found that activation of GR inhibited activity of numerous transcription factors (TF) including AP-1, SRF, NF-κB, p53, ATF-2, CEBPα, Ets-1, Elk-1, STAT1 and others, many of which are regulated via MAPK cascade. The structural analysis of hepsin and AMACR promoters provided the mechanistic rationale for PC marker downregulation by glucocorticoids via inhibition of specific TFs. Our data suggest that GR functions as a tumor suppressor in prostate, and inhibits multiple signaling pathways and transcriptional factors involved in proliferation and transformation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- AMACR:
-
alpha-methylacyl-CoA racemase
- AP-1:
-
activator protein 1
- BPH:
-
benign prostatic hyperplasia
- Erk1/2:
-
extracellular signal-regulated kinase 1 and 2
- FA:
-
fluocinolone acetonide
- HGPIN:
-
high-grade prostatic intraepithelial neoplasia
- HRPC:
-
hormone refractory prostate carcinoma
- MAPKs:
-
mitogen-activated protein kinases
- Mek1/2:
-
dual specificity mitogen-activated protein kinase 1 and 2
- NF-κB:
-
nuclear factor kappa-B
- PC:
-
prostate carcinoma
- PIN:
-
prostatic intraepithelial neoplasia
- SAPK/JNK:
-
stress-activated protein kinase/c-Jun-N-terminal kinase
- TF:
-
transcription factor
- YFP:
-
yellow fluorescent protein
References
Ananthanarayanan V, Deaton RJ, Yang XJ, Pins MR, Gann PH . (2005). Alpha-methylacyl-CoA Racemase (AMACR) expression in normal prostatic glands and high-grade prostatic intraepithelial neoplasia (HGPIN): association with diagnosis of prostate cancer. Prostate 63: 341–346.
Bourcier T, Forgez P, Borderie V, Scheer S, Rostene W, Laroche L . (2000). Regulation of human corneal epithelial cell proliferation and apoptosis by dexamethasone. Invest Ophthalmol Vis Sci 41: 4133–4141.
Bruna A, Nicolas M, Munoz A, Kyriakis JM, Caelles C . (2003). Glucocorticoid receptor–JNK interaction mediates inhibition of the JNK pathway by glucocorticoids. EMBO J 22: 6035–6044.
Budunova IV, Carbajal S, Kang H, Viaje A, Slaga TJ . (1997). Altered glucocorticoid receptor expression and function during mouse skin carcinogenesis. Mol Carcinogen 18: 177–185.
Chen Z, Fan Z, McNeal JE, Nolley R, Caldwell MC, Mahadevappa M et al. (2003). Hepsin and maspin are inversely expressed in laser capture microdissectioned prostate cancer. J Urol 169: 1316–1319.
De Bosscher K, Vanden Berghe W, Haegeman G . (2003). The interplay between the glucocorticoid receptor and nuclear factor-kappaB or activator protein-1: molecular mechanisms for gene repression. Endocr Rev 24: 488–522.
Dhanasekaran SM, Barrette TR, Ghosh D, Shah R, Varambally S, Kurachi K et al. (2001). Delineation of prognostic biomarkers in prostate cancer. Nature 412: 822–826.
Fakih M, Johnson CS, Trump DL . (2002). Glucocorticoids and treatment of prostate cancer: a preclinical and clinical review. Urology 60: 553–561.
Fixemer T, Remberger K, Bonkhoff H . (2003). Differential expression of the estrogen receptor beta (ERbeta) in human prostate tissue, premalignant changes, and in primary, metastatic, and recurrent prostatic adenocarcinoma. Prostate 54: 79–87.
Gioeli D, Black BE, Gordon V, Spencer A, Kesler CT, Eblen ST et al. (2006). Stress kinase signaling regulates androgen receptor phosphorylation, transcription, and localization. Mol Endocrinol 20: 503–515.
Greenberg AK, Hu J, Basu S, Hay J, Reibman J, Yie TA et al. (2002). Glucocorticoids inhibit lung cancer cell growth through both the extracellular signal-related kinase pathway and cell cycle regulators. Am J Respir Cell Mol Biol 27: 320–328.
Greenstein S, Ghias K, Krett NL, Rosen ST . (2002). Mechanisms of glucocorticoid-mediated apoptosis in hematological malignancies. Clin Cancer Res 8: 1681–1694.
Grossmann ME, Huang H, Tindall DJ . (2001). Androgen receptor signaling in androgen-refractory prostate cancer. J Natl Cancer Inst 93: 1687–1697.
Jiang X, Norman M, Roth L, Li X . (2004). Protein-DNA array-based identification of transcription factor activities regulated by interaction with the glucocorticoid receptor. J Biol Chem 279: 38480–38485.
Jiang Z, Woda BA . (2004). Diagnostic utility of alpha-methylacyl CoA racemase (P504S) on prostate needle biopsy. Adv Anat Pathol 11: 316–321.
Kassel O, Sancono A, Kratzschmar J, Kreft B, Stassen M, Cato AC . (2001). Glucocorticoids inhibit MAP kinase via increased expression and decreased degradation of MKP-1. EMBO J 20: 7108–7116.
Klezovitch O, Chevillet J, Mirosevich J, Roberts RL, Matusik RJ, Vasioukhin V . (2004). Hepsin promotes prostate cancer progression and metastasis. Cancer Cell 6: 185–195.
Koutsilieris M, Mitsiades C, Dimopoulos T, Vacalicos J, Lambou T, Tsintavis A et al. (2002). Combination of dexamethasone and a somatostatin analogue in the treatment of advanced prostate cancer. Expert Opin Invest Drugs 11: 283–293.
Li CL, Johnson GR . (1998). Clonal cultures in vitro for hematopoietic cells using semisolid agar medium. In: Celis JS (ed). Cell Biology: A Laboratory Handbook 2nd edn, vol. 1. Academic Press: San-Diego (CA), pp 172–177.
Maroni PD, Koul S, Meacham RB, Koul HK . (2004). Mitogen activated protein kinase signal transduction pathways in the prostate. Cell Commun Signal 2: 5–12.
Mimeault M, Pommery N, Henichart JP . (2003). New advances on prostate carcinogenesis and therapies: involvement of EGF–EGFR transduction system. Growth Factors 21: 1–14.
Mohler JL, Chen Y, Hamil K, Hall SH, Cidlowski JA, Wilson EM et al. (1996). Androgen and glucocorticoid receptors in the stroma and epithelium of prostatic hyperplasia and carcinoma. Clin Cancer Res 2: 889–895.
Necela BM, Cidlowski JA . (2004). Mechanisms of glucocorticoid receptor action in noninflammatory and inflammatory cells. Proc Am Thorac Soc 1: 239–246.
Nishimura K, Nonomura N, Satoh E, Harada Y, Nakayama M, Tokizane T et al. (2001). Potential mechanism for the effects of dexamethasone on growth of androgen-independent prostate cancer. J Natl Cancer Inst 93: 1739–1746.
Peterziel H, Mink S, Schonert A, Becker M, Klocker H, Cato AC . (1999). Rapid signalling by androgen receptor in prostate cancer cells. Oncogene 18: 6322–6329.
Quinn DI, Henshall SM, Sutherland RL . (2005). Molecular markers of prostate cancer outcome. Eur J Cancer 41: 858–887.
Ray DW . (1996). Molecular mechanisms of glucocorticoid resistance. J Endocrinol 149: 1–5.
Ricote M, Garcia-Tunon I, Bethencourt F, Fraile B, Onsurbe P, Paniagua R et al. (2006). The p38 transduction pathway in prostatic neoplasia. J Pathol 208: 401–407.
Rosenberg I . (1996). Protein Analysis and Purification: Benchtop Techniques. Birkhauser: Boston, MA, pp 103–109.
Schacke H, Docke WD, Asadullah K . (2002). Mechanisms involved in the side effects of glucocorticoids. Pharmacol Ther 96: 23–43.
Schaefer JS, Zhang M . (2003). Role of maspin in tumor metastasis and angiogenesis. Curr Mol Med 3: 653–658.
Schug J, Overton CG . (2005). TESS: Transcription Element Search Software on the WWW. Eur J Cancer 41: 858–887 (http://www.cbil.upenn.edu/cgi-bin/tess/tess).
Torlakovic E, Lilleby W, Berner A, Torlakovic G, Chibbar R, Furre T et al. (2005). Differential expression of steroid receptors in prostate tissues before and after radiation therapy for prostatic adenocarcinoma. Int J Cancer 117: 381–386.
Weaver I, Champagne F, Brown S, Dymov S, Sharma S, Meaney M et al. (2005). Reversal of maternal programming of stress responses in adult offspring through methyl supplementation: altering epigenetic marking later in life. J Neurosci 25: 11045–11054.
Wu W, Chaudhuri S, Brickley DR, Pang D, Karrison T, Conzen SD . (2004). Microarray analysis reveals glucocorticoid-regulated survival genes that are associated with inhibition of apoptosis in breast epithelial cells. Cancer Res 64: 1757–1764.
Zha S, Ferdinandusse S, Denis S, Wanders RJ, Ewing CM, Luo J et al. (2003). Alpha-methylacyl-CoA racemase as an androgen-independent growth modifier in prostate cancer. Cancer Res 63: 7365–7376.
Acknowledgements
This work was supported by the DOD prostate grant DAMD17-03-1-0522 and the Northwestern University Prostate SPORE Developmental Project (to IB). We thank Drs KR Yamamoto (University of California, San Francisco, CA, USA), WC Greene (Gladstone Institute for Virology and Immunology, University of California, San Francisco, CA, USA), DJ Klumpp, (Northwestern University, Chicago IL, USA), M Beato, (Philipps-Universitat, Marburg, Germany) for their generous gift of plasmids. We are grateful to Drs P Stern, J Pelling and O Volpert (Northwestern University, Chicago, IL, USA) for fruitful discussions of our work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Yemelyanov, A., Czwornog, J., Chebotaev, D. et al. Tumor suppressor activity of glucocorticoid receptor in the prostate. Oncogene 26, 1885–1896 (2007). https://doi.org/10.1038/sj.onc.1209991
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209991
Keywords
This article is cited by
-
Glucocorticoid receptor triggers a reversible drug-tolerant dormancy state with acquired therapeutic vulnerabilities in lung cancer
Nature Communications (2021)
-
SPOP promotes ATF2 ubiquitination and degradation to suppress prostate cancer progression
Journal of Experimental & Clinical Cancer Research (2018)
-
Comparing the rules of engagement of androgen and glucocorticoid receptors
Cellular and Molecular Life Sciences (2017)
-
The role of glucocorticoid receptor in prostate cancer progression: from bench to bedside
International Urology and Nephrology (2017)
-
Differential regulation of bladder cancer growth by various glucocorticoids: corticosterone and prednisone inhibit cell invasion without promoting cell proliferation or reducing cisplatin cytotoxicity
Cancer Chemotherapy and Pharmacology (2014)