Abstract
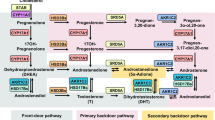
The metabolic pathways of androgens and processes by which androgens induce re-growth after androgen deprivation therapy in prostate cancer have not been fully elucidated. In this study, finasteride decreased PSA secretion in medium containing testosterone, androstenedione, androstenediol and dehydroepiandrosterone, whereas dihydrotestosterone (DHT)- and hydroxy-flutamide-induced PSA production was not inhibited by finasteride in LNCaP-FGC cells. The present data show that adrenal androgen precursors do not directly interact with androgen receptors (ARs) but are converted to DHT via the intraprostatic metabolic pathways, resulting in the induction of LNCaP activity. This is the first report confirming this mechanism experimentally and also suggest the use of combined therapies that target ARs and prevent the formation of DHT within prostate cancer cells to achieve optimal therapeutic efficacy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Huggins C, Hodges CV . Studies on prostate cancer. Effect of castration, estrogen and androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res 1941; 1: 293–297.
Navarro D, Luzardo OP, Fernandez L, Chesa N, Diaz-Chico BN . Transition to androgen-independence in prostate cancer. J Steroid Biochem Mol Biol 2002; 81: 191–201.
Gregory CW, Johnson Jr RT, Mohler JL, French FS, Wilson EM . Androgen receptor stabilization in recurrent prostate cancer is associated with hypersensitivity to low androgen. Cancer Res 2001; 61: 2892–2898.
Rau KM, Kang HY, Cha TL, Miller SA, Hung MC . The mechanisms and managements of hormone-therapy resistance in breast and prostate cancers. Endocr Relat Cancer 2005; 12: 511–532.
Gioeli D et al. Androgen receptor phosphorylation. Regulation and identification of the phosphorylation sites. J Biol Chem 2002; 277: 29304–29314.
Tremblay A, Tremblay GB, Labrie F, Giguère V . Ligand-independent recruitment of SRC-1 to estrogen receptor β through phosphorylation of activation function AF-1. Mol Cell 1999; 3: 513–519.
Kato S et al. Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase. Science 1995; 270: 1491–1494.
Vihko P et al. Control of cell proliferation by steroids: the role of 17HSDs. Mol Cell Endocrinol 2006; 241: 141–148.
Tomita Y, Bilim V, Hara N, Kasahara T, Takahashi K . Role of IRF-1 and caspase-7 in IFN-gamma enhancement of Fas-mediated apoptosis in ACHN renal cell carcinoma cells. Int J Cancer 2003; 104: 400–408.
Berns EM, de Boer W, Mulder E . Androgen-dependent growth regulation of and release of specific protein(s) by the androgen receptor containing human prostate tumor cell line LNCaP. Prostate 1986; 9: 247–259.
van Steenbrugge GJ, van Uffelen CJ, Bolt J, Schroder FH . The human prostatic cancer cell line LNCaP and its derived sublines: an in vitro model for the study of androgen sensitivity. J Steroid Biochem Mol Biol 1991; 40: 207–214.
Soronen P et al. Sex steroid hormone metabolism and prostate cancer. J Steroid Biochem Mol Biol 2004; 92: 281–286.
Pelletier G, Luu-The V, Li S, Labrie F . Localization of type 5 17beta-hydroxysteroid dehydrogenase mRNA in mouse tissues as studied by in situ hybridization. Cell Tissue Res 2005; 320: 393–398.
Elo JP et al. Characterization of 17beta-hydroxysteroid dehydrogenase isoenzyme expression in benign and malignant human prostate. Int J Cancer 1996; 66: 37–41.
Koh E, Noda T, Kanaya J, Namiki M . Differential expression of 17beta-hydroxysteroid dehydrogenase isozyme genes in prostate cancer and noncancer tissues. Prostate 2002; 53: 154–159.
Lazier CB, Thomas LN, Douglas RC, Vessey JP, Rittmaster RS . Dutasteride, the dual 5alpha-reductase inhibitor, inhibits androgen action and promotes cell death in the LNCaP prostate cancer cell line. Prostate 2004; 58: 130–144.
Bonkhoff H, Stein U, Aumuller G, Remberger K . Differential expression of 5 alpha-reductase isoenzymes in the human prostate and prostatic carcinomas. Prostate 1996; 29: 261–271.
Thomas LN et al. Differential alterations in 5alpha-reductase type 1 and type 2 levels during development and progression of prostate cancer. Prostate 2005; 63: 231–239.
Negri-Cesi P et al. Presence of 5alpha-reductase isozymes and aromatase in human prostate cancer cells and in benign prostate hyperplastic tissue. Prostate 1998; 34: 283–291.
Tian G . In vivo time-dependent inhibition of human steroid 5 alpha-reductase by finasteride. J Pharm Sci 1996; 85: 106–111.
Nishiyama T, Hashimoto Y, Takahashi K . The influence of androgen deprivation therapy on dihydrotestosterone levels in the prostatic tissue of patients with prostate cancer. Clinical Cancer Research 2004; 10: 7121–7126.
Bartsch W, Klein H, Schiemann U, Bauer HW, Voigt KD . Enzymes of androgen formation and degradation in the human prostate. Ann NY Acad Sci 1990; 595: 53–66.
Culig Z et al. Mutant androgen receptor detected in an advanced-stage prostatic carcinoma is activated by adrenal androgens and progesterone. Mol Endocrinol 1993; 7: 1541–1550.
Palmberg C et al. Androgen receptor gene amplification at primary progression predicts response to combined androgen blockade as second line therapy for advanced prostate cancer. J Urol 2000; 164: 1992–1995.
Culig Z, Comuzzi B, Steiner H, Bartsch G, Hobisch A . Expression and function of androgen receptor coactivators in prostate cancer. J Steroid Biochem Mol Biol 2004; 92: 265–271.
Labrie F, Dupont A, Bélanger A . Complete androgen blockade for the treatment of prostate cancer. In: de Vita VT, Hellman S. et Rosenberg SA (eds). Important Advances in Oncology. J.B. Lippincott: Philadelphia, 1985; pp 193–217.
Prostate Cancer Triallists’ Collaborative Group. Maximum androgen blockade in advanced prostate cancer: an overview of the randomised trials. Lancet 2000; 355: 1491–1498.
Labrie F et al. GnRH agonists in the treatment of prostate cancer. Endocr Rev 2005; 26: 361–379.
Bennett CL et al. Maximum androgen-blockade with medical or surgical castration in advanced prostate cancer: a meta-analysis of nine published randomized controlled trials and 4128 patients using Flutamide. Prostate Cancer Prostatic Dis 1999; 2: 4–8.
Akaza H et al. Superior anti-tumor efficacy of bicalutamide 80 mg in combination with a luteinizing hormone-releasing hormone (LHRH) agonist versus LHRH agonist monotherapy as first-line treatment for advanced prostate cancer: interim results of a randomized study in Japanese patients. Jpn J Clin Oncol 2004; 34: 20–28.
McConnell JD et al. Finasteride, an inhibitor of 5 alpha-reductase, suppresses prostatic dihydrotestosterone in men with benign prostatic hyperplasia. J Clin Endocrinol Metab 1992; 74: 505–508.
Thompson IM et al. The influence of finasteride on the development of prostate cancer. New England J Med 2003; 349: 215–224.
Leibowitz RL, Tucker SJ . Treatment of localized prostate cancer with intermittent triple androgen blockade: preliminary results in 110 consecutive patients. Oncologist 2001; 6: 177–182.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, K., Nishiyama, T., Hara, N. et al. Importance of the intracrine metabolism of adrenal androgens in androgen-dependent prostate cancer. Prostate Cancer Prostatic Dis 10, 301–306 (2007). https://doi.org/10.1038/sj.pcan.4500956
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4500956
Keywords
This article is cited by
-
The role of ketoconazole in current prostate cancer care
Nature Reviews Urology (2018)
-
Intracrine androgen biosynthesis in renal cell carcinoma
British Journal of Cancer (2017)
-
Circulating sex steroids and prostate cancer: introducing the time-dependency theory
World Journal of Urology (2013)
-
Targeting the Adrenal Gland in Castration-Resistant Prostate Cancer: A Case for Orteronel, a Selective CYP-17 17,20-Lyase Inhibitor
Current Oncology Reports (2013)
-
Inhibition of androstenediol-dependent LNCaP tumour growth by 17α-ethynyl-5α-androstane-3α, 17β-diol (HE3235)
British Journal of Cancer (2009)