-
PDF
- Split View
-
Views
-
Cite
Cite
Helen Olofsson, Alicia M. Frame, Maria R. Servedio, CAN REINFORCEMENT OCCUR WITH A LEARNED TRAIT?, Evolution, Volume 65, Issue 7, 1 July 2011, Pages 1992–2003, https://doi.org/10.1111/j.1558-5646.2011.01286.x
Close - Share Icon Share
Abstract
We use birdsong as a case study to ask whether reinforcement can occur via the spread of a genetically determined female preference for a socially inherited (learned) male trait. We envision secondary contact between two neighboring populations with different song dialects. An individual's ability to learn song is confined by a genetic predisposition: if predispositions are strong, there will be no phenotypic overlap in song between populations, whereas weak predispositions allow phenotypic overlap, or “mixed” song. To determine if reinforcement has occurred, we consider if an allele for within‐population female mating preference, based on song, can spread, and whether population specific songs can concurrently be maintained at equilibrium. We model several scenarios, including costs to mating preferences, mating preferences in hybrids, and hybrids having the ability to learn pure songs. We find that when weak predispositions are fixed within a population reinforcement based on song cannot occur. However, when some individuals have strong predispositions, restricting phenotypic overlap between populations in the trait, reinforcement is only slightly inhibited from a purely genetic model. Generalizing beyond the example of song, we conclude that socially learned signals will tend to prohibit reinforcement, but it may still occur if some individuals acquire trait phenotypes genetically.
The prevalence of natural hybrids is evidence that premating isolation between species is often incomplete. As human activity increasingly alters species ranges, species pairs are coming into secondary contact, where they will subsequently undergo population fusion, extinction, or divergence. Mounting empirical evidence shows that secondary contact sometimes results in divergence through the process of reinforcement, the evolution of premating isolation after secondary contact due to selection against hybridization (reviews in Noor 1999; Servedio and Noor 2003; Coyne and Orr 2004). Ongoing research in this field addresses the important question of what biological factors influence whether this divergence will occur or not.
All reinforcement models to date have assumed that mate choice during premating isolation occurs based upon mating cues (the targets of mating preferences) that are completely genetically inherited (e.g., Liou and Price 1994; Kelly and Noor 1996; Servedio 2000). A recent model showed that reinforcement can occur via mating preferences that are learned by sexual imprinting (Servedio et al. 2009). Likewise, some male traits, such as song in birds, can also have a substantial learned component (e.g., Slater 1989; Catchpole and Slater 1995; Kroodsma and Miller 1996; Freeberg 2000). Signal learning, like learned preferences, has been shown both empirically (e.g., in flycatchers, Haavie et al. 2004), and theoretically (e.g., Lachlan and Servedio 2004) to affect the conditions for speciation. It has been suggested that song learning might inhibit speciation in its early stages but promote it in its later stages (Slabbekoorn and Smith 2002). In this study, we use a theoretical model to examine how learned signaling traits may affect the process of reinforcement. We concentrate on birdsong as a case study. To isolate the effects on reinforcement of trait (signal) learning specifically we assume that preferences are genetically inherited, and discuss at the end of the article the possible effects of violations of this assumption.
Many classic examples of learning are found in birds. Songbirds (oscines, order Passeriformes, suborder Passeri) are known to learn habitat preferences (Slagsvold and Wiebe 2007), mate preferences (ten Cate and Vos 1999), and song, which can be a sexually selected trait (e.g., Searcy and Yasukawa 1996; Nowicki and Searcy 2004). Some songbirds, such as song sparrows, have been shown to have predispositions to learn conspecific song over heterospecific song (e.g., Marler and Peters 1977; Podos et al. 2004). These predispositions may be neurological (e.g., Whaling et al 1997; Nelson 2000) or due to morphological constraints (e.g., Wallschläger 1980; Palacios and Tubaro 2000; Mahler and Tubaro 2001; Podos 2001; Ballentine 2006; Christensen et al. 2006; Derryberry 2009). Song thus also has a genetic component, in the form of a constraint on learning.
To use birdsong as a case study for reinforcement with a learned trait, we develop a single model that can be interpreted in two ways: one that is specific to birdsong, and the other that is generalizable to any learned signaling trait. First, considering birdsong specifically, we focus on alleles that control the genetic predispositions that underlie learned song, with different alleles characteristic of each population. We imagine the case in which there are nonoverlapping genetic predispositions that denote the ideal range of songs that could be acquired by each population. When these genetic predispositions are strong they exercise a great deal of constraint over the song learned; with such predispositions, it is possible that the songs acquired by individuals in the two species (or populations) would also be nonoverlapping. If instead predispositions were weak, the range of song that individuals in each population might acquire would be more likely to overlap, especially if the birds are sympatric and potentially using members of the opposite population as tutors for song learning. One of the parameters that we vary in this article is the proportion of individuals in a population with strong versus weak predispositions. The models in this article also have a more general interpretation outside of birdsong, which focuses on the probability that a trait (signal) used as a mating cue is socially learned. In this case, we consider alleles in each population that determine trait phenotypes that can be modified by learning. When the trait is entirely genetically controlled there may again be no overlap in phenotypes between populations. If instead the trait, which may be a vocal trait or a display trait, has a certain probability of being learned within the confines of genetic control, overlap could again be possible between two populations, depending on the tutor, in an identical fashion to the case of weak predispositions in birdsong. In this more general interpretation, the parameter determining the frequency of individuals with weak predispositions in the birdsong case changes to a parameter that determines the probability of learning. Our model thus applies to the situation of birdsong, but can also address the evolution of reinforcement for socially learned signals in general, provided learning leads to overlap in phenotype. When we wish to stress the generality of our results we turn to this latter interpretation.
The concept of traits that phenotypically “overlap” between two populations is an important one in our article. In the context of birdsong, overlap between songs of different species could occur in the form of “mixed” song. Mixed singers are individuals that extensively copy the vocal patterns of another species, either by alternating foreign song with their own song or by combining fragments of both songs into a single phrase (Helb et al. 1985). There is some evidence to suggest that mixed songs most likely arise when there are errors in copying song from the wrong species of tutor (Helb et al. 1985; Thielcke 1986).
We envision two initially allopatric habitats, where females mate randomly and males produce songs that are controlled by either strong or weak genetic predispositions. Over time in the two habitats, local dialects have evolved by the fixation of different allele combinations at loci affecting song; hybrid genotypes at these loci have low fitness. We consider secondary contact to occur in the form of gene flow via dispersal, and examine the effects on reinforcement of the frequency of strong versus weak predispositions in a population. We consider versions of the model with and without costs to choice, learning of song by hybrids, and hybrids expressing mating preferences. In order for reinforcement to occur in our model (1) an allele coding for within‐population female mating preference must spread upon secondary contact of the two populations and (2) population‐specific songs must be maintained at the final equilibrium. We find that when all individuals have weak predispositions, reinforcement is prohibited, but when at least some individuals in each population have strong predispositions reinforcement generally occurs only slightly less often than in a model with a genetically controlled trait. Under the more general interpretation of the model, we find that reinforcement cannot occur if all individuals learn their signaling trait by cultural transmission, but is generally only slightly inhibited when there is a moderate probability that some individuals will acquire their trait phenotype genetically.
Model
We use a haploid two‐island model, which assumes that there are two populations and two separate habitats, connected by juvenile dispersal. Our model can further be characterized as a “one allele” (sensu Felsenstein 1981) assortative mating model. We assume one female mate choice locus, A, and two loci determining species characteristics, M and N. The locus A has two alleles A1 and A2. The allele A1 represents random mating, whereas A2 causes females to mate assortatively with males singing songs characteristic of the female's genetic type. We assume that A1 is initially fixed across the system and we investigate if allele A2 can spread in the two populations. The M and N loci have two alleles each, M1 and M2, and N1 and N2, with each subscript characteristic of a different population. These two loci serve to partially determine which songs a male produces, and can be used as markers for whether an individual is a hybrid (i.e., M1N2 and M2N1) or not (i.e., M1N1 and M2N2, a “pure” individual).
We consider three distinct versions of the model. In the first (the “basic” model), we assume that hybrids, which are assumed to be in a poor condition in all versions of the model, mate randomly, and only sing mixed song, which overlaps between both populations (see Song Production). Random mating may be expected in hybrids if their poorer condition prohibits them from expressing a preference. Hybrids may only sing mixed song if their song production is limited to the range of overlap of parental songs (see also Lemaire 1977); this assumption further allows us to consider mixed song partly as an indicator of hybrid status. In the second version, we vary the basic model by assuming that hybrids, like purebreds, have a preference for the song that is characteristic of their genotype (in their case, mixed song). In the third version of the model, we instead assume that hybrids are able to learn parental song, and can thus sing either mixed song or pure song from either parental dialect. Finally the assumptions of the second and third versions of the model are also combined to test the robustness of our results.
SONG PRODUCTION
Males produce songs that are learned but are also based on an underlying genetic predisposition (based on the model of Lachlan and Feldman 2003; Lachlan and Servedio 2004; Olofsson and Servedio 2008). We assume that songs can be grouped into three different song “categories,” which can be thought of as any population‐specific collection of songs or song elements. Two of these categories (1 and 3) form local dialects in the two allopatric habitats. We assume that songs from category 1 are characteristic of M1N1 individuals whereas songs from category 3 are characteristic of M2N2 individuals, due in each case to the genotype at the M and N loci coding for a genetic predisposition to sing songs from each of these intervals. Category 2 contains songs derived from categories 1 and 3; the songs in this category can be thought of as either mixed song composed of parts of songs from each pure category or as a mixed repertoire of songs from each pure category (in this case we assume that females hear multiple songs from a male's repertoire while choosing a mate and that males cannot bias songs from a tutor's repertoire toward population‐specific song). For simplicity, we discuss primarily the former interpretation throughout the article. We assume throughout the article that purebred females discriminate against mixed song compared to song characteristic of their predisposition, but that females prefer mixed song to completely foreign song.
We assume that mixed songs originate because either (1) hybrid individuals (i.e., M1N2 or M2N1) produce songs that are blends of the pure songs from the allele‐specific song categories (or in the case of a mixed repertoire, do not sing exclusively from one pure category because of shifted or generalized predispositions), or (2) individuals make mistakes in the learning process. These mistakes happen when purebred individuals with weak predispositions try to produce songs from the category not coded for by their genotype at the M and N loci. That is, for example, M1N1 individuals trying to produce songs from category 3 end up singing the mixed songs; mixed song can thus be thought of as an area of “overlap” of song categories 1 and 3 that can occur with weak predispositions. Note that this model does not account for the possibility that songs in category 2 may arise spontaneously by cultural mutation; we believe that unless cultural mutation is very frequent this is unlikely to substantially affect the outcome of the model.
Once they have originated by one of the mechanisms above, songs in the mixed category can also be transmitted by learning from a male singing mixed song regardless of the learner's genotype at the M and N loci, again provided that predispositions are weak enough to allow overlap. We assume that learning is oblique, where the tutors are nonparental adult males. In males that have strong predispositions, which do not allow overlap between song categories 1 and 3, M1N1 males can only produce songs in category 1, and similarly M2N2 males can only produce songs in category 3. The frequency of males in a population with weak predispositions is denoted by parameter B (in the more general interpretation of the model B is the probability that the trait is learned). When B is higher, there will generally be a higher probability that learning is attempted from foreign or hybrid males and thus that song in category 2 is acquired. This parameter is given at the beginning of each simulation and is not allowed to evolve (see Olofsson and Servedio 2008 for a model of the evolution of this parameter).
LIFE CYCLE
We keep track of genotypes at the A, M, and N loci in females and phenogenotypes (genotype at the A, M, and N loci plus the song phenotype) in males. At the beginning of each simulation, population 1 females are fixed for the A1M1N1 genotype and population 1 males are fixed for the phenogenotype A1M1N11 (all males carry the allele combination A1M1N1 and are singing songs from category 1). Likewise, population 2 females are fixed for the A1M2N2 genotype and population 2 males are fixed for the phenogenotype A1M2N23. Migration, the first step in the life cycle, occurs as individuals move at a fixed rate m between the two populations after the commencement of secondary contact (see Appendix S1 for all equations).
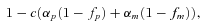
Following selection via costs of preference are mating and song learning. Before the introduction of A2, all females have the allele A1, which means that they mate randomly with respect to song (all females are always assumed to have equal mating success). We allow the two populations to reach an initial equilibrium after the commencement of secondary contact before assortative mating is introduced (see below for discussion of violations of this assumption). After this initial equilibrium is reached, we introduce the allele for assortative mating A2 at a low frequency (0.01 %) in both populations (A2 is introduced in linkage equilibrium with all of the other alleles in the system). Females who carry this allele have a preference for some male songs over other songs. Specifically, purebred females that carry A2 are 1 +αp times as likely to mate with a male that sings songs from the pure song category that corresponds with their population, compared to a female who is carrying the A1 allele and compared to males singing the opposite pure song. These females are also 1 +αm times as likely to mate with a male singing mixed song (we set αm= 1 < αp; recall that mixed song contains some elements of both pure songs, or following our second interpretation that mixed repertoires contain local as well as foreign song, so we assume that females prefer them over completely foreign song). Females thus mate assortatively based on the allele combination at the M and N loci; an A2 M1N1 female will prefer males singing songs from category 1 and A2M2N2 female will prefer songs from category 3. We assume, however, that females cannot perceive the difference between mixed songs sung by males of different genotypes. A female is therefore equally likely to mate with any genotype of male singing songs from category 2.
We assume in the basic model that hybrid females mate randomly. In the second version of the model, we vary this to assume that hybrid females are 1 +αp times as likely to mate with a male that sings a mixed song (which hybrids are generally more likely to sing themselves) than with a male singing a pure song. In this version of the model, the hybrid songs thus gain a mating advantage absent in the basic version of the model.
New allele frequencies are calculated after mating, followed by the acquisition of song by juvenile males. Pure males (with allele combination M1N1 or M2N2) can either produce songs constrained by strong predispositions (songs from category 1 or 3 respectively) with probability 1 −B, or they can produce song constrained by weak predispositions (which for both types of males could include song from category 2), with probability B. When a male learns, he listens to what adult males are singing in his population and acquires his songs in proportion to the frequency of each song category in the parental generation, given the confines of his genetic predisposition. For example, M1N1 males learning from other males singing songs from category 1 learn these songs, whereas those learning from males singing songs from categories 2 or 3 produce songs in category 2. In the “basic” version of the model, all hybrid males sing songs from category 2, regardless of the strength of their predisposition. This is later expanded, in version three of the model, to assume that hybrids with weak predispositions learn in proportion to the songs that they hear, including from categories 1 and 3, while hybrids with strong predispositions only produce mixed song.
The final step in the life cycle is viability selection acting on juvenile hybrids. The strength of selection against hybrids is measured by the selection coefficient sE, which reduces the fitness of individuals carrying the hybrid genotypes (M1N2 or M2N1). The assumption that the same loci (M and N) affect song and hybrid fitness renders this model a “magic trait” model (sensu Gavrilets 2004), in which the trait under divergent selection is the same as the mating cue (for review see Servedio 2009). Speciation generally occurs more easily in a magic trait model than a model in which divergent selection and mating occur at different loci, but this should not affect the comparisons between different features (weak vs. strong predispositions, hybrids having preferences, etc) within this model.
We created numerical simulations that iterated the exact equations of the life cycle, and let each simulation run, after the introduction of A2, until the changes in the frequency of each genotype between two generations is less than 10−10.
Results
In all three versions of the model we found broad regions of parameter space in which reinforcement occurred. We assess reinforcement by a combination of two results: (1) the spread of the allele for assortative mating, A2, and (2) the maintenance of a high frequency of the population‐specific song categories 1 and 3 in their respective populations. These results must co‐occur in any region of the parameter space in order for reinforcement to have occurred resulting in the maintenance of distinct song dialects.
BASIC MODEL
We first consider the assumptions of the basic model where hybrids mate at random and learn only songs from category 2, to assess the importance of selection against hybrids, migration rate, strength of preference, frequency of weak and strong predispositions, and costs of preference. It is well established from previous models that strong selection against hybrids makes reinforcement more likely, and that stronger selection against hybrids is necessary to obtain reinforcement with higher migration rates in a two‐island model (e.g., Servedio and Kirkpatrick 1997). The results of our basic model with no costs of preference (e.g., Fig. 1A) are entirely consistent with these generalities. We therefore use the strength of selection against hybrids necessary for the establishment of the assortative mating allele as a measure of the “ease” of reinforcement in the rest of our analyses, and consider a range of migration rates.
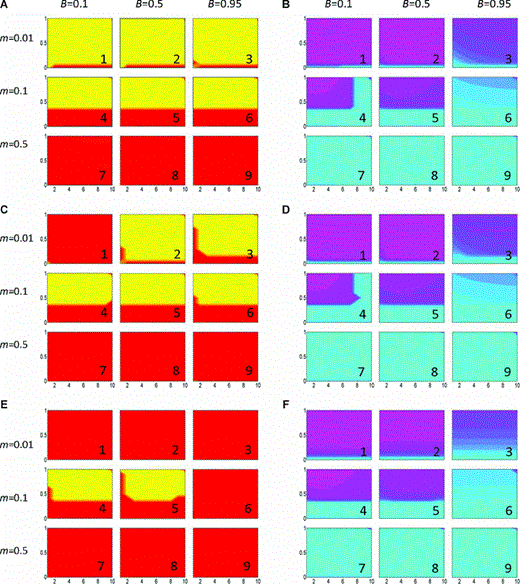
Evolution of assortative mating and prevalence of local dialects in the basic model. For each individual plot, the x‐axis represents the strength of female preference (αp), and the y‐axis is the strength of selection against hybrids (sE). For each of the six 3 × 3 panels of plots (A through E), we vary the migration rate (m) across the rows and vary the frequency of weak predispositions (B, defined as the probability of learning in the general interpretation of the model) by column. The panels in the left column (A, C, and E) show the frequency of the assortative mating allele A2 in the total population; yellow indicates A2 is fixed, red indicates that A2 has been lost, and intermediate shades represent polymorphisms where more yellow = higher frequency of A2. The panels in the right hand column (B, D, and F) display the prevalence of mixed versus pure song types; light purple indicates that population specific songs (categories 1 and 3) are fixed in their respective populations, whereas light blue indicates that they are not (either all individuals are singing mixed song (category 2), or one of the pure dialects (category 1 or 3) is fixed across both populations). Intermediate shades between blue and purple represent decreasing frequencies of local dialects in their respective habitats. We consider reinforcement to have occurred if the allele A2 has spread and local dialects are maintained at reasonably high frequencies (yellow on the left‐hand panels coinciding with purple on the right‐hand panels). Panels A and B: no cost c= 0; panels C and D: c= 0.001; panels E and F: c= 0.01.
We find that when the migration rate is low and costs are absent, reinforcement generally occurs: assortative mating evolves and local dialects are maintained (Fig. 1A1,2,3, B1,2,3). Given strong enough selection against hybrids, the assortative mating allele A2 also fixes for higher migration rates (e.g., 1 %, Fig. 1A). Populations also maintain a significant proportion of local dialects over most of this parameter space, although as the percentage of individuals with weak predispositions increases, local dialects generally become increasingly replaced by mixed song (e.g., Figs. 1B and 2), making the differences between the habitats less distinct. Local dialects still persist at some frequency, however, even with quite a high frequency of weak predispositions. Thus despite the fact that individuals migrate between the two habitats, and weak predispositions are present in the populations, local dialects are generally preserved.
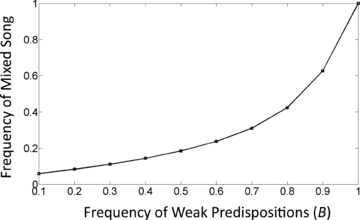
Effect of weak predispositions on the prevalence of mixed song. The frequency of weak predispositions (B) is on the x‐axis and the frequency of mixed song is on the y‐axis. Parameter values are αp= 5, sE= 0.5, c= 0, and m= 0.05.
In some cases, we find that the spread of assortative mating can be accompanied by the subsequent rapid loss of variation in the trait. Specifically, when strong predispositions are common, migration was relatively high (e.g., 1 %), selection was high, and preferences were strong (e.g., Fig. 1B4, blue area above sE≈ 0.4 on right), we found that allele A2 spread, but subsequently songs from either category 1 or 3 would suddenly take over across both populations (note that in the figures purple indicates both dialects are present in their respective populations, so the presence of blue can indicate either mixed song, or in this case, one dialect fixed across both populations). These cases are due to unstable dynamics (see Fig S1), as was confirmed by examining the eigenvalues of the Jacobian matrices for the system of equations (the Jacobian matrices were also examined for all cases with c= 0 and lower migration rates that did not exhibit this instability to confirm the stability of those apparent equilibria).
There are two fundamental situations in which the spread of A2 is prevented entirely. First, when all males have weak predispositions, mixed song spreads in both habitats, until they are the only songs present. If this happens before the introduction of the allele for assortative mating, A2 will never spread. In this case, all males are identical when it comes to which songs they produce, hence there will be no selection for increasing the frequency of A2. Second, as the migration rate increases and selection becomes very low (e.g., 1 % migration and sE < 0.05), there is an increasing chance that one phenogenotype will take over and become the only one present in both habitats before variation in assortative mating is introduced (in simulations that started with symmetrical frequencies of local dialects we see this as a sudden departure from symmetry due to numerical error). In these cases, again, there is no variation upon which the spread of A2 can be based.
When there are no costs to preference, increasing the strength of preference often has no effect on the eventual spread of an allele for assortative mating in the parameter ranges tested. Stronger preferences can however alter the amount of song differentiation between populations in the directions of both increasing and decreasing the proportion of local dialects (e.g., Fig. 1B3–B6). In the context of assortative mating, stronger preferences can act as a greater force promoting population divergence, causing the initial increase in the maintenance of local dialects. When preferences causing assortative mating are very strong, however, they can actually reduce the force of sexual selection that drives populations to diverge (Servedio 2011), potentially leading to loss of dialect variation.
Increasing the costs of preference tends to cause a restriction in the parameter space in which assortative mating can evolve. This is seen by the prevention of the evolution of assortment with low preferences (e.g., Fig. 1C1,2,3,6, E4,5) and with low migration (Fig. 1C1, E1,2,3). The former case most likely indicates that with weak preferences the forces driving the evolution of assortative mating are not strong enough to overcome the costs. The latter case contrasts with the general finding that higher, not lower, migration tends to prevent reinforcement. Reinforcement may not be occurring with low migration in this case because the force of selection from rare hybridization, which would be the driving force for the evolution of assortment, is too weak to counter the stronger costs against preferences. With intermediate migration rates, there are more hybrids being formed and hence stronger selection against them to counter the preference costs, whereas with very strong migration variation in song is generally lost at the initial equilibrium, as described above. Finally, assortative mating also does not evolve with a very high frequency of weak predispositions when costs are high (Fig. 1E6). This most likely indicates that here costs of preference are strong enough to overcome evolution of assortative mating when it is based upon song differences that are fairly weak between populations.
HYBRIDS EXPRESS A MATING PREFERENCE
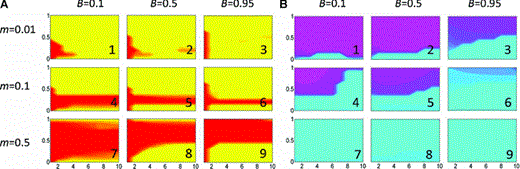
The assumption that hybrid females express a preference for males singing the mixed song often leads to a restriction of the parameter space in which reinforcement can occur. We tested this assumption assuming no costs to preference for simplicity; Figure 3 should thus be compared to Figure 1A,B. It can be seen that with low migration rates, the spread of the assortative mating allele is somewhat restricted with low preference strengths even when selection against hybrids is substantial (close to sE= 0.5, Fig. 3A1,2,3). Although the assortative mating allele can now spread with strong preferences even when selection against hybrids is weak, variation in local song dialects are generally not maintained at significant levels under these parameter combinations (Fig. 3B1,2,3). As the migration rate increases there is a shrinking of the parameter space in which reinforcement can occur and maintain local dialects, as in the basic model: song variation is generally low or lost with weak selection against hybrids, producing a band at the bottom of the figures where assortative mating evolves but has little effect on the songs present in the population.
Evolution of assortative mating and prevalence of local dialects when hybrids express mating preferences. Specifics of the figure are the same as in Figure 1. In this figure c= 0.
HYBRIDS LEARN PARENTAL SONG
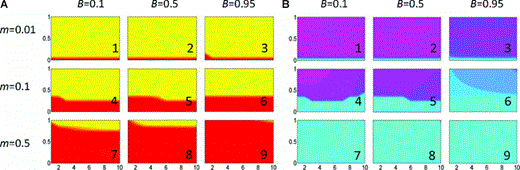
When we assume that hybrid males can express weak predispositions, allowing them to learn song from categories 1 and 3, we find that the evolution of assortative mating generally occurs over a little more of the parameter space than when hybrids all sing mixed song (again this assumption was implemented with no costs, so direct comparisons can be drawn between Figure 4 with Figures 3 and 1A,B). In particular assortative mating evolves more easily with high migration and strong selection against hybrids (Fig. 4A7,8). This assumption does not have a substantial effect on the likelihood of reinforcement as we define it, however, because song differentiation is only maintained in roughly the same area of parameter space as when hybrids do not learn song (Fig. 4B; with the exception of more maintenance of pure song with high preference in Fig. 4B4,5). We note, however, that the fixation of an allele for assortative mating occurs more slowly when hybrids are able to learn parental song than when they cannot (not shown), so that when reinforcement occurs, it takes longer.
Evolution of assortative mating and prevalence of local dialects when hybrids can learn parental song. Specifics of the figure are the same as in Figure 1. In this figure c= 0.
When both the assumptions of hybrids expressing weak predispositions by learning parental song and hybrids having mating preferences are implemented simultaneously, the results are very similar to those when only the assumption of hybrids having preferences is implemented (not shown).
Discussion
We use a model of mating based on birdsong to show how a learned trait affects speciation via reinforcement. We test a variety of scenarios, including the effects of costs to preference, hybrids expressing versus not expressing preferences, and hybrids being able to learn and produce parental song. We find that under most conditions the ability to learn the songs of another population (which in our model occurs with weak predispositions) has an inhibitory effect on two components necessary for song maintenance by reinforcement: (1) the spread of an allele for assortative mating based on song, and (2) the maintenance of differences in song between the two populations. However, this inhibitory effect is generally not drastic unless weak predispositions are widespread in the population. To generalize this to the broader case of a signaling trait that is acquired genetically, allowing no overlap in phenotype between populations, or by social learning, allowing overlap, reinforcement would be impossible if all individuals learned their trait. If, on the other hand, there were an intermediate probability that the trait was learned, we would expect reinforcement to be inhibited by the presence of learning in the population, but perhaps not to a significant extent.
We first consider the situation in which costs to preferences are absent, to obtain a basic picture of the evolution of preferences and song in the model. We find that in the absence of costs, conditions of relatively low migration and strong selection against hybrids are the most conducive to reinforcement, matching expectations from previous studies (e.g., Kelly and Noor 1996; Servedio and Kirkpatrick 1997; Servedio 2000). With low migration rates the evolution of assortative mating can occur even when weak predispositions are very common in the population, but when weak predispositions are fixed, the spread of an allele for assortative mating is prevented. The strength of predispositions also affects the degree to which population‐specific songs (local dialects) can be maintained after assortative mating is established, with less population specificity when weak predispositions are common than when strong predispositions are common. When costs are absent, weak predispositions thus only inhibit the spread of assortative mating in extreme cases, but do affect the maintenance of population‐specific songs.
Analysis of the presence or absence of local dialects helps in the understanding of when assortative mating will evolve. Our model assumes that a mutation introducing assortative mating occurs after secondary contact has been established long enough for an initial equilibrium in gene frequencies to be reached. We find that as long as population‐specific dialects are maintained at any frequency at the initial equilibrium with random mating, an allele for assortative mating will spread if introduced. This finding parallels that of Servedio (2000), who found that an allele for assortative mating would always spread when it targeted a locally adapted mating cue that was able to maintain population differentiation after secondary contact (this assortative mating model, and the current model, are both “one‐allele” models sensu Felsenstein 1981, and are thus the most favorable models for reinforcement). In both cases, the maintenance of variation in the mating cue is enough to cause the uninhibited spread of an assortative mating allele that targets it. In Servedio (2000) this evolution occurs because in both of two incipient species the assortative mating allele becomes genetically associated (in linkage disequilbrium) with locally adapted trait alleles. The selection that derives from local adaptation in Servedio (2000) is replaced in the current model by the frequency‐dependent advantage that forms under assortative mating from the more common song predisposition allele combination being less likely to be involved in a mating that produces low‐fitness hybrids. If weak predispositions are fixed within a population, mixed song becomes the only song present at the initial equilibrium; mixed song gains an advantage during song learning because it is sung by individuals of both populations, and is therefore overrepresented, per population, among the field of acceptable tutors. When mixed song is fixed in both populations, the alleles in the system are no longer associated with a pure song interval, and the assortative mating allele will thus no longer spread. It should still be possible, however, for the assortative mating allele to spread before the time at which pure songs have been completely lost, if an initial equilibrium in song frequencies is not reached before the allele for assortative mating is introduced (not tested directly in our models). Note that when some individuals have strong predispositions it is guaranteed that some individuals will always sing songs from categories 1 and 3, preventing the fixation of mixed song; in our model we specify that strong predispositions do not allow overlap, which is a critical assumption to allow the maintenance of categories 1 and 3 if there is learning. The degree of overlap of the predispositions of different populations in phenotype space should not affect these conclusions. Finally, note that when migration is too high relative to selection against hybrids local song differentiation is lost through the universal spread of one dialect before the introduction of assortative mating; in this case the selection for the evolution of assortative mating is also removed.
Even when assortative mating can evolve, problems with dialect maintenance can persist. Dialect variation can sometimes disappear completely when weak predispositions are common, leading to solely mixed song. Under other conditions one of the population‐specific songs ranges can fix across both populations after the evolution of assortative mating, removing song variation in the system. There are also ranges of conditions in which population‐specific dialects can be maintained at fairly low frequencies, sometimes as low as a few percentage points—this is more likely to occur when the frequency of weak predispositions is very high. In this case mixed song becomes the most common song in both populations. Although reinforcement may be said to occur with these latter parameter values, very little differentiation is maintained between populations so it does not have a notable effect.
When costs of preference are added to the model, the conditions under which reinforcement occurs are more restricted, depending upon the strength of these costs. Interestingly, we find that the addition of costs can lead to the evolution of assortative mating only with intermediate migration rates. When migration rates are too low, there are very few hybrids produced, leading to very weak selection driving reinforcement—seemingly too weak to overcome a sufficiently strong direct cost to preferences. As in the case without costs, migration rates that are too high lead to the loss of variation in song at the initial equilibrium, before the allele for assortative mating is introduced. Reinforcement may thus only evolve when migration occurs at an intermediate rate. We also find that when costs are high, a high frequency of weak predispositions can also inhibit the evolution of assortment; the selective force driving the evolution of assortment is weaker with a high frequency of weak predispositions and cannot overcome strong costs.
In the basic version of our model, we assume that hybrids mate randomly, but we also consider a version of the model in which hybrids, like purebred individuals, mate assortatively. The evolution of assortative mating in this situation may thus increase the spread of mixed song, which hybrids would prefer, making reinforcement less likely. We did observe this effect in our simulations but it was not drastic; distinct dialects were still maintained in most of the parameter space where they occurred when hybrids were assumed to mate at random. The assumption of assortative mating of hybrids also affected the evolution of assortative mating itself in two ways: by restricting it when preferences were very weak, and by increasing it when selection against hybrids was low. When hybrids mate assortatively, it increases the frequency of individuals that prefer mixed song; under certain parameter values this seems to provide more of a target for the evolution of assortment. Assortative mating thus evolved in areas in which it was lost in the basic model. Note, however, that we would not consider reinforcement to have occurred in most of these cases because the evolution of assortment did not lead to differences between the populations (song variation was often lost).
Finally, we considered a version of the model in which hybrids with weak predispositions can learn pure parental songs. This assumption might initially also be expected to restrict the conditions for reinforcement, because it would cause purebred females that mate according to song to sometimes be choosing hybrid mates. Several complicated effects are interacting in this situation however. First note that because purebred females mating with hybrids can produce backcrossed offspring that reconstitute the parental genotype (50 % of the offspring in haploids regardless of the recombination rate), mating with hybrids is not as detrimental as mating with a purebred of the opposite species. Second, whenever more hybrids are produced selection against them creates more of a driving force for reinforcement; in this case hybrids learning parental song will lead to stronger effective selection for a given preference strength than when hybrids only sing mixed song. Third, when hybrids learn pure parental song this increases the frequency of pure (not mixed) song in the interacting populations; because learning is frequency dependent, this increases the frequency of pure song that will in turn be learned by pure individuals of each species. Finally, when mating is assortative, the increased chance of mating with a hybrid must also be balanced against the lessened chance of mating with the opposite species when compared to random mating; this will also be a frequency‐dependent effect. The predictions for hybrids being able to learn parental song are therefore not entirely straightforward. When we analyze this version of the model explicitly, we find that reinforcement occurs over a very similar area of parameter space regardless of whether hybrids can learn parental song. We do, however, find that when assortative mating evolves, it does so more slowly when hybrids can learn pure parental song than when they cannot.
Thus in all versions of our model, reinforcement was found to occur in a significant proportion of the parameter space when costs to preferences were absent, and in all versions this area was found to be smaller when the frequency of weak predispositions rose too high. Because of the simple nature of costs, we expect that the addition of costs to the variants of the model where hybrids can learn parental song or where hybrids mate assortatively would have a similar effect to that which they had in the basic model. Our different analyses of the effects of hybrid behavior stem from the fact that there are little empirical data on these issues. Although we found that different assumptions about hybrid behavior did not critically change our results, uncovering the actual behavioral properties of hybrids seems to us an interesting empirical challenge.
Note that because of the assumption that the same loci (M and N) control the species marker (song) and affect hybrid fitness, our models are “magic trait” models, which should make it easier for reinforcement to occur (Gavrilets 2004). Previous work with a very similar model to the current one, however, has shown that reinforcement can occur even if separate loci controlled these two critical traits (e.g., Servedio et al. 2009). The separation of these loci should restrict the parameter space under which reinforcement would occur, but would not be expected to affect the comparisons between the versions of the model that we analyze. Furthermore, it is possible that bill morphology may sometimes constrain both song and feeding ecology in a way that would match with magic trait assumption made here (but note that the evidence to date suggests uniform preferences for high performance song that is constrained by bill morphology, rather than preference for matching genotypes as we modeled here, e.g., Podos 2001; Ballentine 2006).
It is interesting to consider the potential effects of other simplifying assumptions used in our model. We assume that individuals are haploid, whereas birds of course are diploid. Diploid versions of reinforcement models have not thus far exhibited any unexpected behavior when compared to similar haploid models (e.g., Kirkpatrick and Servedio 1999; Servedio and Sætre 2003; Servedio et al. 2009), and there is no a priori reason to believe that this assumption would alter the conclusions of the present model. Additionally, our model assumes that all mixed song falls into one intermediate category, whereas in reality it is possible that mixed song may be more or less like one of the parental songs, which may affect how likely it is to be learned by different parental genotypes. It is difficult to speculate about the exact consequences of varying this assumption, although it seems likely that the conclusions we make about the impossibility of reinforcement when traits are completely learned and about the comparisons between the three versions of our model would still stand. We also assume that there are only two populations, and that migration between them is restricted. It would also be interesting to consider the evolution of song across a spatial grid or when secondary contact results in complete sympatry.
Although in birds song learning is often accompanied by female mating preferences that are also at least partially learned (e.g., Riebel 2003), in this article we wished to isolate the effect of trait learning in particular on reinforcement. To accomplish this, we assumed that preferences are genetically inherited. In contrast to learned traits, the effects of learned preferences on reinforcement have been addressed in previous models and are fairly well understood. Specifically using a reinforcement model, Servedio et al. (2009) showed that preferences learned by paternal imprinting closely mimic the effects of self‐referent phenotype matching (identical to the assumption of assortative mating used in this article), primarily because these are both one‐allele models (sensu Felsenstein 1981). Verzijden et al. (2005), using toy models of sympatric speciation, showed that parental imprinting closely mimics self‐referent phenotype matching, although maternal imprinting is a better match than paternal imprinting. Tramm and Servedio (2008) developed the explanation of this latter effect by pointing out that when there is sexual selection on male traits, paternal imprinting causes the acquisition of preferences that at the population level are disproportionally skewed towards the more preferred male trait, precisely because this trait is over‐represented among fathers (successfully mated males). Verzijden et al. (2005) also showed that oblique imprinting can have very different properties that maternal and paternal imprinting because it does not allow gene‐culture disequilbrium to be established between preferences and traits. From this discussion, we conclude that the results of our model would be extremely similar to the current findings if preferences were learned by maternal or paternal imprinting instead of being genetically acquired, but that preferences learned by oblique transmission may be likely to have different effects.
One conclusion drawn from our models is that variation in song should be lost across both populations with high migration. It is possible, however, that song dialects may be maintained or originate in parapatry or even sympatry (e.g., Nottebohm 1969, 1975; Handford 1981). Migration may somehow be low even in these situations, but this may not be likely. This discrepancy may be due to other mechanisms for dialect maintenance, such as song learning later in life (e.g., Bell et al 1998; Wright et al. 2005; Koetz et al. 2007), lowered fitness of immigrants (e.g., Jenkins 1978; Baker 1982; Beecher et al. 2000; Wilson et al. 2000), or potentially the constant formation of new songs in different areas, that are not considered in the current model.
Although several previous studies have examined the evolution of genetically inherited preferences for learned traits (e.g., Aoki 1989), this evolution has not been previously considered in the context of reinforcement. Previous studies have, however, examined song divergence outside of a reinforcement context and analyzed whether this divergence was promoted or inhibited by learning. Ellers and Slabbekoorn (2003) found that learning promoted song divergence along a spatial grid, allowing more divergence in phenotype than was found in genotype. Lachlan and Servedio (2004) also found that learning promoted speciation, but considered an allopatric scenario where drift was a key factor in allowing divergence to occur. In contrast to these models, we find that learning can be considered to inhibit reinforcement when it occurs via the evolution of a preference based upon learned song, due to the fact that it allows overlap of trait phenotypes. Thus if speciation often proceeds in allopatry or parapatry followed by secondary sympatry, learning may be expected to promote divergence in the early stages of speciation and inhibit it later on (cf. the predictions of Slabbekoorn and Smith, 2002, made assuming significant dispersal early in the speciation process).
Our model generates several testable predictions. In the case of birdsong, unless dialects have diverged enough to be close to the point where they cannot be recognized by the other incipient species, it may be likely that population‐level variation in the category of predispositions will not occur; predispositions will either be weak or strong throughout a population (we feel, however, that cases on this border, where there may be variation in our categories of predispositions, may be some of the most interesting from the standpoint of speciation research). If predispositions are strong, learning of mixed song would not occur, and reinforcement should be as easy as with a trait that is genetically inherited. If predispositions are weak, allowing for the development of mixed song, we would predict that reinforcement should not occur unless it is temporary, if the spread of mixed song throughout the populations was very slow. The rate of spread of mixed song is likely to be slowed by factors we did not consider in our model, such as learning that is partly paternally based or is from immediate neighbors (rather than from the population at large), potentially allowing for some evolution of assortative mating and temporary maintenance of song differences even with uniformly weak predispositions. If factors that we did not consider stabilize the production of mixed versus pure song these effects may be longer lasting. Parallels to these predictions can also be made for the generalized case of a socially learned signaling trait. Only one empirical study that we are aware of has studied reinforcement based on a trait affected by learning: Haavie et al. (2004) found evidence for reinforcement based on song in collared flycatchers, in a case in which the other species involved, pied flycatchers, sometimes learn mixed song. Interestingly, in this case mixed song is only learned by one species, making comparisons with our model difficult. Reinforcement in this study also occurred by song divergence, not through the de novo evolution of assortative mating based on such song.
We conclude that when song is learned based upon uniformly weak predispositions, the evolution of assortment leading to the maintenance of song differences is impossible, or may occur as a transitory phenomenon accompanying the slow spread of mixed song. If population‐specific songs are given a refuge because of some individuals having strong predispositions, however, assortment involving mating based upon song can often occur almost as easily as when it is based on morphological differences. To generalize beyond song, whether learned traits may prove a fruitful avenue for evidence of reinforcement in the future is thus dependent upon whether all individuals learn, and whether learning allows overlap in trait phenotypes to occur.
Associate Editor: M. Doebeli
ACKNOWLEDGMENTS
The authors thank J. Adamson, S. Dhole, M. Doebeli, D. Irwin, J. Rowell, K. Sewall, H. Wiley, and two anonymous reviewers for comments. MRS and AMF were funded by the NSF grant DEB‐0614166 to MRS