Abstract
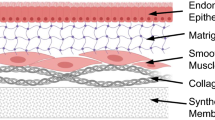
Endometrial stromal and epithelial cell cross talk is known to influence many of the dynamic changes that occur during the menstrual cycle. We modified our previous model and embedded telomerase-immortalized human endometrial stromal cells and Ishikawa adenocarcinoma epithelial cells in a collagen—Matrigel hydrogel to create a tissue-engineered model of the endometrium. Comparisons of single and cocultured cells examined communication between endometrial stromal and epithelial cells, which were cultured with 0 or 10 nmol/L 17β estradiol; conditioned medium was used to look at the production of paracrine factors. Using this model, we were able to identify the changes in interleukin 6 (IL-6) and active matrix metalloproteinase 2, which appear to be due to paracrine signaling and differences in transforming growth factor β1 (TGF-β1) that do not appear to be due to paracrine signaling. Moreover, IL-6, TGF-β1, and DNA content were also affected by the presence of estradiol in many of the tissues. These results indicate that paracrine and endocrine signaling are involved in human endometrial responses and support the use of coculture models to further investigate cell–cell and cell–matrix interactions.
Similar content being viewed by others
References
Pretto CM, Gaide Chevronnay HP, Cornet PB, et al. Production of interleukin-1alpha by human endometrial stromal cells is triggered during menses and dysfunctional bleeding and is induced in culture by epithelial interleukin-1 alpha released upon ovarian steroids withdrawal. J Clin Endocrinol Metab. 2008;93(10): 4126–4134.
Singer CF, Marbaix E, Kokorine I, et al. Paracrine stimulation of interstitial collagenase (MMP-1) in the human endometrium by interleukin 1alpha and its dual block by ovarian steroids. Proc Natl Acad Sci USA. 1997;94(19):10341–10345.
Wang H, Bocca S, Anderson S, et al. Sex steroids regulate epithelial–stromal cell cross talk and trophoblast attachment invasion in a three-dimensional human endometrial culture system. Tissue Eng Part C Methods. 2013;19(9):676–687.
Evron A, Goldman S, Shalev E. Effect of primary human endometrial stromal cells on epithelial cell receptivity and protein expression is dependent on menstrual cycle stage. Hum Reprod. 2011;26(1):176–190.
Kim MR, Park DW, Lee JH, et al. Progesterone-dependent release of transforming growth factor-beta1 from epithelial cells enhances the endometrial decidualization by turning on the Smad signalling in stromal cells. Mol Hum Reprod. 2005;11(11):801–808.
Osteen KG, Rodgers WH, Gaire M, Hargrove JT, Gorstein F, Matrisian LM. Stromal–epithelial interaction mediates steroidal regulation of metalloproteinase expression in human endometrium. Proc Natl Acad Sci USA. 1994;91(21):10129–10133.
Bruner KL, Rodgers WH, Gold LI, et al. Transforming growth factor beta mediates the progesterone suppression of an epithelial metalloproteinase by adjacent stroma in the human endometrium. Proc Natl Acad Sci USA. 1995;92(16):7362–7366.
Zhang H, Li M, Wang F, et al. Endometriotic epithelial cells induce MMPs expression in endometrial stromal cells via an NFkappaB-dependent pathway. Gynecol Endocrinol. 2010;26(6):456–467.
Zhang L, Patterson AL, Teixeira JM, Pru JK. Endometrial stromal beta-catenin is required for steroid-dependent mesenchymal–epithelial cross talk and decidualization. Reprod Biol Endocrinol. 2012;10:75.
Pierro E, Minici F, Alesiani O, et al. Stromal–epithelial interactions modulate estrogen responsiveness in normal human endometrium. Biol Reprod. 2001;64(3):831–838.
Rubel CA, Jeong JW, Tsai SY, Lydon JP, Demayo FJ. Epithelial–stromal interaction and progesterone receptors in the mouse uterus. Semin Reprod Med. 2010;28(1):27–35.
Qi XF, Nan ZC, Jin YP, Qu YY, Zhao XJ, Wang AH. Stromal–epithelial interactions modulate the effect of ovarian steroids on goat uterine epithelial cell interleukin-18 release. Domest Anim Endocrinol. 2012;42(4):210–219.
Mohtashami M, Zuniga-Pflucker JC. Three-dimensional architecture of the thymus is required to maintain delta-like expression necessary for inducing T cell development. J Immunol. 2006; 176(2):730–734.
Schyschka L, Sanchez JJ, Wang Z, et al. Hepatic 3D cultures but not 2D cultures preserve specific transporter activity for acetaminophen-induced hepatotoxicity. Arch Toxicol. 2013; 87(8):1581–1593.
Ghosh S, Spagnoli GC, Martin I, et al. Three-dimensional culture of melanoma cells profoundly affects gene expression profile: a high density oligonucleotide array study. J Cell Physiol. 2005; 204(2):522–531.
Gomez-Lechon MJ, Jover R, Donato T, et al. Long-term expression of differentiated functions in hepatocytes cultured in three-dimensional collagen matrix. J Cell Physiol. 1998;177(4):553–562.
Delcommenne M, Streuli CH. Control of integrin expression by extracellular matrix. J Biol Chem. 1995;270(45):26794–26801.
Kenny PA, Lee GY, Myers CA, et al. The morphologies of breast cancer cell lines in three-dimensional assays correlate with their profiles of gene expression. Mol Oncol. 2007;1(1):84–96.
Schutte SC, Taylor RN. A tissue-engineered human endometrial stroma that responds to cues for secretory differentiation, decidualization, and menstruation. Fertil Steril. 2012;97(4):997–1003.
Martin RD. The evolution of human reproduction: a primatological perspective. Am J Phys Anthropol. 2007;(suppl 45):59–84.
Marbaix E, Kokorine I, Moulin P, Donnez J, Eeckhout Y, Courtoy PJ. Menstrual breakdown of human endometrium can be mimicked in vitro and is selectively and reversibly blocked by inhibitors of matrix metalloproteinases. Proc Natl Acad Sci USA. 1996;93(17):9120–9125.
Salamonsen LA, Woolley DE. Menstruation: induction by matrix metalloproteinases and inflammatory cells. J Reprod Immunol. 1999;44(1–2):1–27.
Gaide Chevronnay HP, Selvais C, Emonard H, Galant C, Marbaix E, Henriet P. Regulation of matrix metalloproteinases activity studied in human endometrium as a paradigm of cyclic tissue breakdown and regeneration. Biochim Biophys Acta. 2012; 1824(1):146–156.
Cooke PS, Buchanan DL, Young P, et al. Stromal estrogen receptors mediate mitogenic effects of estradiol on uterine epithelium. Proc Natl Acad Sci USA. 1997;94(12):6535–6540.
Salama SA, Mohammad MA, Diaz-Arrastia CR, et al. Estradiol-17beta upregulates Pyruvate kinase M2 expression to co-activate estrogen receptor-alpha and to integrate metabolic reprogramming with the mitogenic response in endometrial cells [published online January 28, 2014.]. J Clin Endocrinol Metab. 2014:jc20132639.
Wallace A, Cooney TE, Englund R, Lubahn JD. Effects of interleukin-6 ablation on fracture healing in mice. J Orthop Res. 2011;29(9):1437–1442.
Ebihara N, Matsuda A, Nakamura S, Matsuda H, Murakami A. Role of the IL-6 classic- and trans-signaling pathways in corneal sterile inflammation and wound healing. Invest Ophthalmol Vis Sci. 2011;52(9):8549–8557.
Jiang GX, Zhong XY, Cui YF, et al. IL-6/STAT3/TFF3 signaling regulates human biliary epithelial cell migration and wound healing in vitro. Mol Biol Rep. 2010;37(8):3813–3818.
McFarland-Mancini MM, Funk HM, Paluch AM, et al. Differences in wound healing in mice with deficiency of IL-6 versus IL-6 receptor. J Immunol. 2010;184(12):7219–7228.
Girasole G, Jilka RL, Passeri G, et al. 17 beta-estradiol inhibits interleukin-6 production by bone marrow-derived stromal cells and osteoblasts in vitro: a potential mechanism for the antiosteo-porotic effect of estrogens. J Clin Invest. 1992;89(3):883–891.
Kovacs EJ, Plackett TP, Witte PL. Estrogen replacement, aging, and cell-mediated immunity after injury. J Leukoc Biol. 2004; 76(1):36–41.
Papanicolaou DA, Wilder RL, Manolagas SC, Chrousos GP. The pathophysiologic roles of interleukin–6 in human disease. Ann Intern Med. 1998;128(2):127–137.
Pottratz ST, Bellido T, Mocharla H, Crabb D, Manolagas SC. 17 beta-Estradiol inhibits expression of human interleukin-6 promoter-reporter constructs by a receptor-dependent mechanism. J Clin Invest. 1994;93(3):944–950.
Puder JJ, Freda PU, Goland RS, Wardlaw SL. Estrogen modulates the hypothalamic-pituitary-adrenal and inflammatory cytokine responses to endotoxin in women. J Clin Endocrinol Metab. 2001;86(6):2403–2408.
Ikushima H, Miyazono K. TGFbeta signalling: a complex web in cancer progression. Nat Rev Cancer. 2010;10(6):415–424.
Han G, Li F, Singh TP, Wolf P, Wang XJ. The pro-inflammatory role of TGFbeta1: a paradox? Int J Biol Sci. 2012;8(2):228–235.
Roberts AB, Heine UI, Flanders KC, Sporn MB. Transforming growth factor-beta. Major role in regulation of extracellular matrix. Ann N Y Acad Sci. 1990;580:225–232.
Li MO, Wan YY, Sanjabi S, Robertson AK, Flavell RA. Transforming growth factor-beta regulation of immune responses. Annu Rev Immunol. 2006;24:99–146.
Goffin F, Munaut C, Frankenne F, et al. Expression pattern of metalloproteinases and tissue inhibitors of matrix-metallopro teinases in cycling human endometrium. Biol Reprod. 2003; 69(3):976–984.
Rigot V, Marbaix E, Lemoine P, Courtoy PJ, Eeckhout Y. In vivo perimenstrual activation of progelatinase B (proMMP-9) in the human endometrium and its dependence on stromelysin 1 (MMP-3) ex vivo. Biochem J. 2001;358(pt 1):275–280.
Irwin JC, Kirk D, Gwatkin RB, Navre M, Cannon P, Giudice LC. Human endometrial matrix metalloproteinase-2, a putative menstrual proteinase. Hormonal regulation in cultured stromal cells and messenger RNA expression during the menstrual cycle. J Clin Invest. 1996;97(2):438–447.
Krikun G, Mor G, Alvero A, et al. A novel immortalized human endometrial stromal cell line with normal progestational response. Endocrinology. 2004;145(5):2291–2296.
Li WA, Barry ZT, Cohen JD, et al. Detection of femtomole quantities of mature cathepsin K with zymography. Anal Biochem. 2010;401(1):91–98.
Wenger SL, Senft JR, Sargent LM, Bamezai R, Bairwa N, Grant SG. Comparison of established cell lines at different passages by karyotype and comparative genomic hybridization. Biosci Rep. 2004;24(6):631–639.
Korch C, Spillman MA, Jackson TA, et al. DNA profiling analysis of endometrial and ovarian cell lines reveals misidentification, redundancy and contamination. Gynecol Oncol. 2012;127(1):241–248.
Laws MJ, Taylor RN, Sidell N, et al. Gap junction communication between uterine stromal cells plays a critical role in pregnancy-associated neovascularization and embryo survival. Development. 2008;135:2659–2668.
Lu Y, Bocca S, Anderson S, et al. Modulation of the expression of the transcription factors T-bet and GATA-3 in immortalized human endometrial stromal cells (HESCs) by sex steroid hormones and cAMP. Reprod Sci. 2013;20(6):699–709.
Logan PC, Ponnampalam AP, Steiner, Mitchell MD. Effect of cyclic AMP and estrogen/progesterone on the transcription of DNA methyltransferases during the decidualization of human endometrial stromal cells. Mol Hum Reprod. 2013;19(5):302–312.
Klinkova O, Hansen KA, Winterton E, Mark CJ, Eyster KM. Two-way communication between endometrial stromal cells and monocytes. Reprod Sci. 2010;17(2):125–136.
Koyama H, Raines EW, Bornfeldt KE, Roberts JM, Ross R. Fibrillar collagen inhibits arterial smooth muscle proliferation through regulation of Cdk2 inhibitors. Cell. 1996;87(6):1069–1078.
Sawatsri S, Desai N, Rock JA, Sidell N. Retinoic acid suppresses interleukin-6 production in human endometrial cells. Fertil Steril. 2000;73(5):1012–1019.
Chen JC, Erikson DW, Piltonen TT, et al. Coculturing human endometrial epithelial cells and stromal fibroblasts alters cell-specific gene expression and cytokine production. Fertil Steril. 2013;100(4):1132–1143.
Mai KT, Yazdi HM, Perkins DG, Parks W. Pathogenetic role of the stromal cells in endometriosis and adenomyosis. Histopathology. 1997;30(5):430–442.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schutte, S.C., James, C.O., Sidell, N. et al. Tissue-Engineered Endometrial Model for the Study of Cell—Cell Interactions. Reprod. Sci. 22, 308–315 (2015). https://doi.org/10.1177/1933719114542008
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719114542008