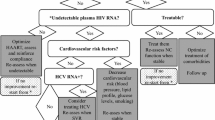
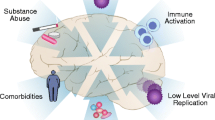
Abstract
In the era of highly active antiretroviral therapy (HAART), HIV-1-associated neurocognitive disorder (HAND) continues to be a common and significant morbidity among individuals infected with HIV. The term HAND encompasses a spectrum of progressively severe CNS involvement, ranging from asymptomatic neurocognitive impairment and minor neurocognitive disorder through to the most severe form of HIV-associated dementia (HAD). While the incidence of HAD has declined significantly with HAART, the milder forms of HAND persist. In addition, HAND now develops in individuals with less advanced immunosuppression. The reasons for the persistence of milder forms of HAND in individuals treated with HAART are not entirely known. There are several hypotheses to explain this phenomenon that include the legacy effect, a failure of antiretroviral agents to reverse neurological damage, poor access of antiretroviral agents to the CNS, chronic systemic immune activation associated with microbial translocation products, sustained CNS inflammation, the improved survival of HIV-seropositive individuals and the possible contribution from aging, amyloid deposition and other co-morbidities. In contrast, the incidence of HIV-associated CNS opportunistic processes including progressive multifocal leukoencephalopathy, tuberculosis, CNS toxo-plasmosis, cytomegalovirus encephalitis, cryptococcosis and primary CNS lymphoma has declined dramatically with the introduction of HAART. This review briefly summarizes our current understanding of HAND and the pathological mechanisms involved, namely direct injury from HIV-1 and viral proteins, indirect neurotoxicity from proinflammatory cytokines and chronic, sustained immune activation in the CNS. To date, only HAART has been shown to benefit HAND despite numerous controlled trials of adjunctive ‘anti-inflammatory’ agents. Although HAART has a profound impact on the incidence and severity of HAND, there exists a ‘therapeutic gap’ as even HAART that is effective at inducing durable virological suppression may only partially reverse HAND. In addition, there may be potential CNS adverse effects of antiretroviral agents. There is an ongoing multicentre clinical trial to investigate the role of the CNS Penetration-Effectiveness index, an indicator of drug permeability and availability in the CNS, to help guide the choice of antiretroviral agents in the treatment of HAND. With recent recommendations for earlier treatment intervention with HAART for HIV-1 infection, it remains to be seen the effects of this on HAND. There is an urgent need to better define the therapeutic guidelines for the prevention and treatment of HAND.



Similar content being viewed by others
References
The Antiretroviral Therapy Cohort Collaboration. Life expectancy of individuals on combination antiretroviral therapy in high-income countries: a collaborative analysis of 14 cohort studies. Lancet 2008 Jul 26; 372(9635): 293–9
Simioni S, Cavassini M, Annoni JM, et al. Cognitive dysfunction in HIV patients despite long-standing suppression of viremia. AIDS 2010 Jun 1; 24(9): 1243–50
Cysique LA, Maruff P, Brew BJ. Prevalence and pattern of neuropsychological impairment in human immunodeficiency virus-infected/acquired immunodeficiency syndrome (HIV/AIDS) patients across pre- and post-highly active antiretroviral therapy eras: a combined study of two cohorts. J Neurovirol 2004 Dec; 10(6): 350–7
McArthur JC, Brew BJ, Nath A. Neurological complications of HIV infection. Lancet Neurol 2005 Sep; 4(9): 543–55
McArthur JC, Steiner J, Sacktor N, et al. Human immunodeficiency virus-associated neurocognitive disorders: mind the gap. Ann Neurol 2010 Jun; 67(6): 699–714
Antinori A, Arendt G, Becker JT, et al. Updated research nosology for HIV-associated neurocognitive disorders. Neurology 2007 Oct 30; 69(18): 1789–99
McArthur JC. HIV dementia: an evolving disease. J Neuroimmunol 2004 Dec; 157(1–2): 3–10
Luther VP, Wilkin AM. HIV infection in older adults. Clin Geriatr Med 2007 Aug; 23(3): 567–83, vii
Sevigny JJ, Albert SM, McDermott MP, et al. Evaluation of HIV RNA and markers of immune activation as predictors of HIV-associated dementia. Neurology 2004 Dec 14; 63(11): 2084–90
Cysique LA, Maruff P, Brew BJ. Variable benefit in neuropsychological function in HIV-infected HAART-treated patients. Neurology 2006 May 9; 66(9): 1447–50
Clifford DB. HIV-associated neurocognitive disease continues in the antiretroviral era. Top HIV Med 2008 Jun–Jul; 16(2): 94–8
Brew BJ. Benefit or toxicity from neurologically targeted antiretroviral therapy? Clin Infect Dis 2010 Mar 15; 50(6): 930–2
Dore GJ, McDonald A, Li Y, et al. Marked improvement in survival following AIDS dementia complex in the era of highly active antiretroviral therapy. AIDS 2003 Jul 4; 17(10): 1539–45
Cunningham AL, Naif H, Saksena N, et al. HIV infection of macrophages and pathogenesis of AIDS dementia complex: interaction of the host cell and viral genotype. J Leukoc Biol 1997 Jul; 62(1): 117–25
Rabkin JG, Chesney MA. Adhering to complex regimens for HIV. GMHC Treat Issues 1998 Apr; 12(4): 8–11
Hinkin CH, Castellon SA, Durvasula RS, et al. Medication adherence among HIV+ adults: effects of cognitive dysfunction and regimen complexity. Neurology 2002 Dec 24; 59(12): 1944–50
Heaton RK, Marcotte TD, Mindt MR, et al. The impact of HIV-associated neuropsychological impairment on everyday functioning. J Int Neuropsychol Soc 2004 May; 10(3): 317–31
Ellis RJ, Deutsch R, Heaton RK, et al. Neurocognitive impairment is an independent risk factor for death in HIV infection: San Diego HIV Neurobehavioral Research Center Group. Arch Neurol 1997 Apr; 54(4): 416–24
Tozzi V, Balestra P, Galgani S, et al. Neurocognitive performance and quality of life in patients with HIV infection. AIDS Res Hum Retroviruses 2003 Aug; 19(8): 643–52
Davis LE, Hjelle BL, Miller VE, et al. Early viral brain invasion in iatrogenic human immunodeficiency virus infection. Neurology 1992 Sep; 42(9): 1736–9
Rumbaugh JA, Nath A. Developments in HIV neuropathogenesis. Curr Pharm Des 2006; 12(9): 1023–44
Schouten J, Cinque P, Gisslen M, et al. HIV-1 infection and cognitive impairment in the cART era: a review. AIDS 2010 Mar 13; 25(5): 561–75
Gonzalez-Scarano F, Martin-Garcia J. The neuropathogenesis of AIDS. Nat Rev Immunol 2005 Jan; 5(1): 69–81
Price RW, Brew B, Sidtis J, et al. The brain in AIDS: central nervous system HIV-1 infection and AIDS dementia complex. Science 1988 Feb 5; 239(4840): 586–92
Churchill MJ, Wesselingh SL, Cowley D, et al. Extensive astrocyte infection is prominent in human immunodeficiency virus-associated dementia. Ann Neurol 2009 Aug; 66(2): 253–8
Kaul M, Lipton SA. Mechanisms of neuroimmunity and neurodegeneration associated with HIV-1 infection and AIDS. JNeuroimmune Pharmacol 2006 Jun; 1(2): 138–51
Wiley CA, Achim C. Human immunodeficiency virus encephalitis is the pathological correlate of dementia in acquired immunodeficiency syndrome. Ann Neurol 1994 Oct; 36(4): 673–6
Brew BJ, Rosenblum M, Cronin K, et al. AIDS dementia complex and HIV-1 brain infection: clinical-virological correlations. Ann Neurol 1995 Oct; 38(4): 563–70
Green DA, Masliah E, Vinters HV, et al. Brain deposition of beta-amyloid is a common pathologic feature in HIV positive patients. AIDS 2005 Mar 4; 19(4): 407–11
Brenchley JM, Price DA, Schacker TW, et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat Med 2006 Dec; 12(12): 1365–71
Price RW, Spudich S. Antiretroviral therapy and central nervous system HIV type 1 infection. J Infect Dis 2008 May 15; 197 Suppl. 3: S294–306
Spudich SS, Nilsson AC, Lollo ND, et al. Cerebrospinal fluid HIV infection and pleocytosis: relation to systemic infection and antiretroviral treatment. BMC Infect Dis 2005; 5: 98
Hagberg L, Dotevall L, Norkrans G, et al. Cerebrospinal fluid neopterin concentrations in central nervous system infection. J Infect Dis 1993 Nov; 168(5): 1285–8
Brew BJ, Bhalla RB, Paul M, et al. Cerebrospinal fluid beta 2-microglobulin in patients with AIDS dementia complex: an expanded series including response to zidovudine treatment. AIDS 1992 May; 6(5): 461–5
Clifford DB, Fagan AM, Holtzman DM, et al. CSF biomarkers of Alzheimer disease in HIV-associated neurologic disease. Neurology 2009 Dec 8; 73(23): 1982–7
Ancuta P, Kamat A, Kunstman KJ, et al. Microbial translocation is associated with increased monocyte activation and dementia in AIDS patients. PLoS One 2008; 3(6):e2516
Brenchley JM, Price DA, Douek DC. HIV disease: fallout from a mucosal catastrophe? Nat Immunol 2006 Mar; 7(3): 235–9
Kaul M. HIV-1 associated dementia: update on pathological mechanisms and therapeutic approaches. Curr Opin Neurol 2009 Jun; 22(3): 315–20
Perry VH, Newman TA, Cunningham C. The impact of systemic infection on the progression of neurodegenerative disease. Nat Rev Neurosci 2003 Feb; 4(2): 103–12
Bashir RM, Harris NL, Hochberg FH, et al. Detection of Epstein-Barr virus in CNS lymphomas by in-situ hybridization. Neurology 1989 Jun; 39(6): 813–7
Tan IL, McArthur JC. HIV-associated central nervous system diseases in the era of combination antiretroviral therapy. Eur J Neurol 2011 Mar; 18(3): 371–2
Garvey L, Winston A, Walsh J, et al. HIV-associated central nervous system diseases in the recent combination antiretroviral therapy era. Eur J Neurol 2011 Mar; 18(3): 527–34
d’Arminio Monforte A, Cinque P, Mocroft A, et al. Changing incidence of central nervous system diseases in the EuroSIDA cohort. Ann Neurol 2004 Mar; 55(3): 320–8
Kirk O, Pedersen C, Cozzi-Lepri A, et al. Non-Hodgkin lymphoma in HIV-infected patients in the era of highly active antiretroviral therapy. Blood 2001 Dec 1; 98(12): 3406–12
Heaton RK, Grant I, Butters N, et al. The HNRC 500: neuropsychology of HIV infection at different disease stages. HIV Neurobehavioral Research Center. J Int Neuropsychol Soc 1995 May; 1(3): 231–51
Sacktor N. The epidemiology of human immunodeficiency virus-associated neurological disease in the era of highly active antiretroviral therapy. J Neurovirol 2002 Dec; 8 Suppl. 2: 115–21
Heaton RK, Clifford DB, Franklin Jr DR, et al. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER Study. Neurology 2010 Dec 7; 75(23): 2087–96
Janssen RS, Nwanyanwu OC, Selik RM, et al. Epidemiology of human immunodeficiency virus encephalopathy in the United States. Neurology 1992 Aug; 42(8): 1472–6
Bhaskaran K, Mussini C, Antinori A, et al. Changes in the incidence and predictors of human immunodeficiency virus-associated dementia in the era of highly active antiretroviral therapy. Ann Neurol 2008 Feb; 63(2): 213–21
Letendre SL, McCutchan JA, Childers ME, et al. Enhancing antiretroviral therapy for human immunodeficiency virus cognitive disorders. Ann Neurol 2004 Sep; 56(3): 416–23
Tozzi V, Balestra P, Galgani S, et al. Positive and sustained effects of highly active antiretroviral therapy on HIV-1-associated neurocognitive impairment. AIDS 1999 Oct 1; 13(14): 1889–97
Robertson KR, Robertson WT, Ford S, et al. Highly active antiretroviral therapy improves neurocognitive functioning. J Acquir Immune Defic Syndr 2004 May 1; 36(1): 562–6
Marra CM, Lockhart D, Zunt JR, et al. Changes in CSF and plasma HIV-1 RNA and cognition after starting potent antiretroviral therapy. Neurology 2003 Apr 22; 60(8): 1388–90
Kitahata MM, Gange SJ, Abraham AG, et al. Effect of early versus deferred antiretroviral therapy for HIV on survival. N Engl J Med 2009 Apr 30; 360(18): 1815–26
Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1 infected adults and adolescents. Bethesda (MD): Department of Health and Human Services, 2011 Jan 10: 1–166
Rumbaugh JA, Steiner J, Sacktor N, et al. Developing neuroprotective strategies for treatment of HIV-associated neurocognitive dysfunction. Futur HIV Ther 2008; 2(3): 271–80
Gavegnano C, Schinazi RF. Antiretroviral therapy in macrophages: implication for HIV eradication. Antivir Chem Chemother 2009; 20(2): 63–78
Sidtis JJ, Gatsonis C, Price RW, et al. Zidovudine treatment of the AIDS dementia complex: results of a placebocontrolled trial. AIDS Clinical Trials Group. Ann Neurol 1993 Apr; 33(4): 343–9
Portegies P, de Gans J, Lange JM, et al. Declining incidence of AIDS dementia complex after introduction of zidovudine treatment. BMJ 1989 Sep 30; 299(6703): 819–21
Brew BJ, Halman M, Catalan J, et al. Factors in AIDS dementia complex trial design: results and lessons from the abacavir trial. PLoS Clin Trials 2007; 2(3): e13
Foudraine NA, Hoetelmans RM, Lange JM, et al. Cerebrospinal-fluid HIV-1 RNA and drug concentrations after treatment with lamivudine plus zidovudine or stavudine.Lancet 1998 May 23; 351(9115): 1547–51
Martin C, Sonnerborg A, Svensson JO, et al. Indinavirbased treatment of HIV-1 infected patients: efficacy in the central nervous system. AIDS 1999 Jul 9; 13(10): 1227–32
Letendre SL, van den Brande G, Hermes A, et al. Lopinavir with ritonavir reduces the HIV RNA level in cerebrospinal fluid. Clin Infect Dis 2007 Dec 1; 45(11): 1511–7
Yilmaz A, Fuchs D, Hagberg L, et al. Cerebrospinal fluid HIV-1 RNA, intrathecal immunoactivation, and drug concentrations after treatment with a combination of saquinavir, nelfinavir, and two nucleoside analogues: the M61022 study. BMC Infect Dis 2006; 6: 63
Price RW, Yiannoutsos CT, Clifford DB, et al. Neurological outcomes in late HIV infection: adverse impact of neurological impairment on survival and protective effect of antiviral therapy. AIDS Clinical Trial Group and Neurological AIDS Research Consortium study team. AIDS 1999 Sep 10; 13(13): 1677–85
Ferrando S, van Gorp W, McElhiney M, et al. Highly active antiretroviral treatment in HIV infection: benefits for neuropsychological function. AIDS 1998 May 28; 12(8): F65–70
Letendre SL, Cherner M, Ellis RJ, et al. The effects of hepatitis C, HIV, and methamphetamine dependence on neuropsychological performance: biological correlates of disease. AIDS 2005 Oct; 19Suppl. 3: S72–8
Morgello S, Estanislao L, Ryan E, et al. Effects of hepatic function and hepatitis C virus on the nervous system assessment of advanced-stage HIV-infected individuals. AIDS 2005 Oct; 19Suppl. 3: S116–22
Perry W, Carlson MD, Barakat F, et al. Neuropsychological test performance in patients co-infected with hepatitis C virus and HIV. AIDS 2005 Oct; 19 Suppl. 3: S79–84
McArthur JC, Hoover DR, Bacellar H, et al. Dementia in AIDS patients: incidence and risk factors. Multicenter AIDS Cohort Study. Neurology 1993 Nov; 43(11): 2245–52
Valcour V, Yee P, Williams AE, et al. Lowest ever CD4 lymphocyte count (CD4 nadir) as a predictor of current cognitive and neurological status in human immunodeficiency virus type 1 infection: the Hawaii Aging with HIV Cohort. J Neurovirol 2006 Oct; 12(5): 387–91
Valcour V, Sithinamsuwan P, Letendre S, et al. Pathogenesis of HIV in the central nervous system. Curr HIV/AIDS Rep 2010 Mar; 8(1): 54–61
Letendre S, Marquie-Beck J, Capparelli E, et al. Validation of the CNS Penetration-Effectiveness rank for quantifying antiretroviral penetration into the central nervous system. Arch Neurol 2008 Jan; 65(1): 65–70
Letendre SL, Ellis RJ, Ances BM, et al. Neurologic complications of HIV disease and their treatment. Top HIV Med 2010 Apr–May; 18(2): 45–55
Lanoy E, Guiguet M, Bentata M, et al. Survival after neuroAIDS: association with antiretroviral CNS Penetration-Effectiveness score. Neurology 2011 Feb 15; 76(7): 644–51
Marra CM, Zhao Y, Clifford DB, et al. Impact of combination antiretroviral therapy on cerebrospinal fluid HIV RNA and neurocognitive performance. AIDS 2009 Jul 17; 23(11): 1359–66
Letendre SL, FitSimons C, Ellis R, et al. Correlates of CSF viral loads in 1,221 volunteers of the CHARTER cohort [abstract no. 172]. 17th Conference on Retroviruses and Opportunistic Infections (CROI); 2010 Feb 16–19; San Francisco (CA)
McGee B, Smith N, Aweeka F. HIV pharmacology: barriers to the eradication of HIV from the CNS. HIV Clin Trials 2006 May–Jun; 7(3): 142–53
Yilmaz A, Gisslen M, Spudich S, et al. Raltegravir cerebrospinal fluid concentrations in HIV-1 infection. PLoS One 2009; 4(9): e6877
Yilmaz A, Izadkhashti A, Price RW, et al. Darunavir concentrations in cerebrospinal fluid and blood in HIV-1-infected individuals. AIDS Res Hum Retroviruses 2009 Apr; 25(4): 457–61
Yilmaz A, Watson V, Else L, et al. Cerebrospinal fluid maraviroc concentrations in HIV-1 infected patients. AIDS 2009 Nov 27; 23(18): 2537–40
Smurzynski M, Wu K, Letendre S, et al. Effects of central nervous system antiretroviral penetration on cognitive functioning in the ALLRT cohort. AIDS 2011 Jan 28; 25(3): 357–65
Sacktor N, Tarwater PM, Skolasky RL, et al. CSF antiretroviral drug penetrance and the treatment of HIV-associated psychomotor slowing. Neurology 2001 Aug 14; 57(3): 542–4
Cysique LA, Maruff P, Brew BJ. Antiretroviral therapy in HIV infection: are neurologically active drugs important? Arch Neurol 2004 Nov; 61(11): 1699–704
Giancola ML, Lorenzini P, Balestra P, et al. Neuroactive antiretroviral drugs do not influence neurocognitive performance in less advanced HIV-infected patients responding to highly active antiretroviral therapy. J Acquir Immune Defic Syndr 2006 Mar; 41(3): 332–7
University of California, San Diego. Clinical trial of CNS-targeted HAART [Clinicaltrials.gov identifier NCT00624195]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Nov 7]
Cunningham R, Silbergleit R. Viral myocarditis presenting with seizure and electrocardiographic findings of acute myocardial infarction in a 14-month-old child. Ann Emerg Med 2000 Jun; 35(6): 618–22
Cinque P, Presi S, Bestetti A, et al. Effect of genotypic resistance on the virological response to highly active antiretroviral therapy in cerebrospinal fluid. AIDS Res Hum Retroviruses 2001 Mar 20; 17(5): 377–83
Canestri A, Lescure FX, Jaureguiberry S, et al. Discordance between cerebral spinal fluid and plasma HIV replication in patients with neurological symptoms who are receiving suppressive antiretroviral therapy. Clin Infect Dis 2010 Mar 1; 50(5): 773–8
Kraft-Terry SD, Buch SJ, Fox HS, et al. A coat of many colors: neuroimmune crosstalk in human immunodeficiency virus infection. Neuron 2009 Oct 15; 64(1): 133–45
Nowacek A, Gendelman HE. NanoART, neuroAIDS and CNS drug delivery. Nanomedicine (Lond) 2009 Jul; 4(5): 557–74
Cysique LA, Brew BJ. Neuropsychological functioning and antiretroviral treatment in HIV/AIDS: a review. Neuropsychol Rev 2009 Jun; 19(2): 169–85
Dalakas MC, Semino-Mora C, Leon-Monzon M. Mitochondrial alterations with mitochondrial DNA depletion in the nerves of AIDS patients with peripheral neuropathy induced by 2′3′-dideoxycytidine (ddC). Lab Invest 2001 Nov; 81(11): 1537–44
Nolan D, Mallal S. Complications associated with NRTI therapy: update on clinical features and possible pathogenic mechanisms. Antivir Ther 2004 Dec; 9(6): 849–63
Cespedes MS, Aberg JA. Neuropsychiatric complications of antiretroviral therapy. Drug Saf 2006; 29(10): 865–74
Rihs TA, Begley K, Smith DE, et al. Efavirenz and chronic neuropsychiatric symptoms: a cross-sectional case control study. HIV Med 2006 Nov; 7(8): 544–8
Robertson KR, Su Z, Margolis DM, et al. Neurocognitive effects of treatment interruption in stable HIV-positive patients in an observational cohort. Neurology 2010 Apr 20; 74(16): 1260–6
Childer ME, Woods SP, Letendre S, et al. Cognitive functioning during highly active antiretroviral therapy interruption in human immunodeficiency virus type 1 infection. J Neurovirol 2008 Nov; 14(6): 550–7
Kirk JB, Goetz MB. Human immunodeficiency virus in an aging population, a complication of success. J Am Geriatr Soc 2009 Nov; 57(11): 2129–38
Valcour V, Shikuma C, Shiramizu B, et al. Higher frequency of dementia in older HIV-1 individuals: the Hawaii Aging with HIV-1 Cohort. Neurology 2004 Sep 14; 63(5): 822–7
Shelburne 3rd SA, Hamill RJ, Rodriguez-Barradas MC, et al. Immune reconstitution inflammatory syndrome: emergence of a unique syndrome during highly active antiretroviral therapy. Medicine (Baltimore) 2002 May; 81(3): 213–27
Gray F, Bazille C, Adle-Biassette H, et al. Central nervous system immune reconstitution disease in acquired immunodeficiency syndrome patients receiving highly active antiretroviral treatment. J Neurovirol 2005; 11 Suppl. 3: 16–22
Venkataramana A, Pardo CA, McArthur JC, et al. Immune reconstitution inflammatory syndrome in the CNS of HIV-infected patients. Neurology 2006 Aug 8; 67(3): 383–8
Johnson T, Nath A. Immune reconstitution inflammatory syndrome and the central nervous system. Curr Opin Neurol 2011 Jun; 24(3): 284–90
Zhao Y, Navia BA, Marra CM, et al. Memantine for AIDS dementia complex: open-label report of ACTG 301. HIV Clin Trials 2010 Jan–Feb; 11(1): 59–67
Schifitto G, Navia BA, Yiannoutsos CT, et al. Memantine and HIV-associated cognitive impairment: a neuropsychological and proton magnetic resonance spectroscopy study. AIDS 2007 Sep 12; 21(14): 1877–86
Navia BA, Dafni U, Simpson D, et al. A phase I/II trial of nimodipine for HIV-related neurologic complications. Neurology 1998 Jul; 51(1): 221–8
Sacktor N, Schifitto G, McDermott MP, et al. Transdermal selegiline in HIV-associated cognitive impairment: pilot, placebo-controlled study. Neurology 2000 Jan 11; 54(1): 233–5
The Dana Consortium on the Therapy of HIV Ddementia and Related Cognitive Disorders. A randomized, double-blind, placebo-controlled trial of deprenyl and thioctic acid in human immunodeficiency virus-associated cognitive impairment: Dana Consortium on the Therapy of HIV Dementia and Related Cognitive Disorders. Neurology 1998 Mar; 50(3): 645–51
Schifitto G, Yiannoutsos CT, Ernst T, et al. Selegiline and oxidative stress in HIV-associated cognitive impairment. Neurology 2009 Dec 8; 73(23): 1975–81
Zink CF, Pagnoni G, Chappelow J, et al. Human striatal activation reflects degree of stimulus saliency. Neuroimage 2006 Feb 1; 29(3): 977–83
Szeto GL, Brice AK, Yang HC, et al. Minocycline attenuates HIV infection and reactivation by suppressing cellular activation in human CD4+ T cells. J Infect Dis 2010 Apr 15; 201(8): 1132–40
Probasco JC, Spudich SS, Critchfield J, et al. Failure of atorvastatin to modulate CSF HIV-1 infection: results of a pilot study. Neurology 2008 Aug 12; 71(7): 521–4
Ances BM, Letendre SL, Alexander T, et al. Role of psychiatric medications as adjunct therapy in the treatment of HIV associated neurocognitive disorders. Int Rev Psychiatry 2008 Feb; 20(1): 89–93
University of Pennsylvania. Modulation of monocyte activation by atorvastatin in HIV infection [Clinicaltrials.gov identifier NCT01263938]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Nov 7]
University of Hawaii. Maraviroc intensification and peripheral blood monocyte HIV DNA levels [Clinical-Trials.gov identifier NCT00987948]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Nov 7]
Johns Hopkins University. Study of paroxetine and fluconazole for the treatment of HIV associated neurocognitive disorder (Paraflu) [ClinicalTrials.gov identifier NCT01354314]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Nov 7]
De Luca A, Ammassari A, Pezzotti P, et al. Cidofovir in addition to antiretroviral treatment is not effective for AIDS-associated progressive multifocal leukoencephalopathy: a multicohort analysis. AIDS 2008 Sep 12; 22(14): 1759–67
Huang SS, Skolasky RL, Dal Pan GJ, et al. Survival prolongation in HIV-associated progressive multifocal leukoencephalopathy treated with alpha-interferon: an observational study. J Neurovirol 1998 Jun; 4(3): 324–32
Vollmer-Haase J, Young P, Ringelstein EB. Efficacy of camptothecin in progressive multifocal leucoencephalopathy. Lancet 1997 May 10; 349(9062): 1366
Royal 3rd W, Dupont B, McGuire D, et al. Topotecan in the treatment of acquired immunodeficiency syndrome-related progressive multifocal leukoencephalopathy. J Neurovirol 2003 Jun; 9(3): 411–9
De Luca A, Giancola ML, Cingolani A, et al. Clinical and virological monitoring during treatment with intrathecal cytarabine in patients with AIDS-associated progressive multifocal leukoencephalopathy. Clin Infect Dis 1999 Mar; 28(3): 624–8
Hall CD, Dafni U, Simpson D, et al. Failure of cytarabine in progressive multifocal leukoencephalopathy associated with human immunodeficiency virus infection: AIDS Clinical Trials Group 243 Team. N Engl J Med 1998 May 7; 338(19): 1345–51
Marzocchetti A, Tompkins T, Clifford DB, et al. Determinants of survival in progressive multifocal leukoencephalopathy. Neurology 2009 Nov 10; 73(19): 1551–8
Cettomai D, McArthur JC. Mirtazapine use in human immunodeficiency virus-infected patients with progressive multifocal leukoencephalopathy. Arch Neurol 2009 Feb; 66(2): 255–8
Biogen Idec. Study to explore the effect of mefloquine in subjects with progressive multifocal leukoencephalopathy (PML) [ClinicalTrials.gov identifier NCT00746941]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Nov 7]]
Berger JR, Pall L, Lanska D, et al. Progressive multifocal leukoencephalopathy in patients with HIV infection. J Neurovirol 1998 Feb; 4(1): 59–68
Antinori A, Cingolani A, Lorenzini P, et al. Clinical epidemiology and survival of progressive multifocal leukoencephalopathy in the era of highly active antiretroviral therapy: data from the Italian Registry Investigative Neuro AIDS (IRINA). J Neurovirol 2003; 9 Suppl. 1: 47–53
Tan CS, Koralnik IJ. Progressive multifocal leukoencephalopathy and other disorders caused by JC virus: clinical features and pathogenesis. Lancet Neurol 2010 Apr; 9(4): 425–37
Acknowledgements
This work was supported by the JHU NIMH Center for novel therapeutics for HIV cognitive disorders (1P30MH075673). Dr Justin C. McArthur has recently received research grants from Biogen Idec, and book royalties from Oxford University Press. Dr Ik Lin Tan has no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tan, I.L., McArthur, J.C. HIV-Associated Neurological Disorders. CNS Drugs 26, 123–134 (2012). https://doi.org/10.2165/11597770-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11597770-000000000-00000