Abstract
SUMMARY: Trichothiodystrophy (TTD) is a rare group of autosomal recessive disorders of DNA repair unified by the presence of sulfur-deficient brittle hair. We report a 3-year-old boy with classic clinical features of TTD, including ichthyosis, alopecia, developmental delay, and tiger-tail banding of the hair shaft on polarizing microscopy. Brain MR imaging showed both diffuse dysmyelination and osteosclerosis, findings that, in combination, may be specific for TTD.
Trichothiodystrophy (TTD), a rare group of autosomal recessive disorders of DNA repair with a wide range of phenotypic expression, is unified by the presence of sulfur-deficient brittle hair.1,2 Many terms have been proposed to subcategorize patients by groups of clinical features. The acronyms PIBIDS, IBIDS, and BIDS stand for various combinations of (in order) Photosensitivity, Ichthyosis, Brittle hair, Intellectual impairment, Decreased fertility, and Short stature,1 whereas the term “Tay Syndrome” refers to TTD with ichthyosis.2 We present the case of a child with TTD and MR imaging findings of dysmyelination and central osteosclerosis.
Case Report
A 3-year-old African-American boy presented for evaluation of failure to thrive, global developmental delay, and ichthyosis. Birth history was significant for an induced vaginal delivery at 37 weeks’ gestational age for maternal pre-eclampsia and prolonged maternal morning sickness. At birth, a collodion membrane was noted, and his weight was 2268 g. He spent 4 days in the neonatal intensive care unit with hypothermia and severely cracked skin. His mother stated that his skin has improved with time, but peeling, dryness, and scaling persist. He also sunburns easily. MR imaging of the brain at birth was reported to be normal. He had no subsequent hospitalizations and no history of seizure. Developmental history was notable for sitting when placed by 1 year, pulling to stand by 2 years, and first words at 18 months. He had not yet taken any steps.
Physical examination was notable for microcephaly, low-set and posteriorly rotated ears, hypoplastic nails, sharpened teeth, and cryptorchidism. Ichthyosis primarily over the proximal upper extremities, chest, and back was noted. Neurologic examination was grossly normal with the exception of tight hip abductors. Ophthalmologic examination demonstrated horizontal conjugate nystagmus.
Focused dermatologic examination revealed thin, sparse hair and eyebrows, with hair loss over the frontal scalp. He had rounded fingernails and toenails and plaquelike scale over the abdomen, chest, and back, with minimal scaling of the arms, legs, and face. Hair samples demonstrated tiger-tail banding under polarizing microscopy, consistent with TTD (Fig 1).
Hair sample demonstrates an alternating light-and-dark “tiger-tail” banding pattern under polarizing microscopy, classic for TTD. Bar is 100 μm.
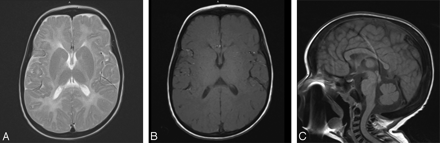
MR imaging of the brain showed diffuse, severely increased T2 signal intensity in the supratentorial white matter, with relative preservation of the cerebellar white matter, consistent with dysmyelination (Fig 2). There was diffuse hypointensity in the calvarial marrow on T1-weighted images, consistent with osteosclerosis. No diffusion or structural abnormalities were appreciated.
A, Axial T2-weighted image shows diffuse supratentorial white matter hyperintensity suggesting dysmyelination. B, Axial T1-weighted image shows minimally diminished gray-white matter contrast. C, Sagittal T1-weighted image shows, to better advantage, low calvarial and vertebral marrow signal intensity, consistent with osteosclerosis.
Discussion
Patients with TTD have abnormal production of transcription factor II H (TFIIH), a general transcription factor active in basal transcription and nucleotide excision repair, due to mutations in genes encoding 3 subunits of TFIIH—ERCC2 (XPD), ERCC3 (XPB), and GTF2H5 (TTDA)—most with mutations of XPD.3–5 Photosensitive TTD patients with (∼50%) have defective nucleotide excision repair mechanisms due to reduced amounts of TFIIH,3,4 without the increased incidence of skin cancer seen in xeroderma pigmentosum, another autosomal recessive disorder of DNA repair demonstrating mutations in XPB and XPD.1,4,6 The severity of symptoms ranges widely and correlates with the amount of damaged TFIIH and abnormal transcription product.3
The sulfur-deficient hair and dysmyelination seen in TTD and in Cockayne syndrome, another autosomal recessive disorder of DNA repair, are believed to be related to impaired basal transcription of genes encoding high-sulfur matrix proteins, rather than to defects of nucleotide excision repair.5,6 Deficiencies in the sulfur-containing amino acids cystine and methionine have been documented in patients with TTD.1,6 Cystine-cystine disulfide linkages stabilize keratins. Cystine deficiency likely causes the hair shaft fragility and resultant alopecia seen in TTD.5,6 Regular undulation of the hair shaft, possibly due to altered protein transcription, results in the characteristic “tiger-tail” banding pattern seen in all patients with TTD, but is also seen in a number of nutritional and metabolic deficiencies.5,6 Impaired transcription of structural components of myelin and of high-sulfur-content molecules important in central nervous system (CNS) development, such as neurocan and phosphacan, has been implicated in the dysmyelination present in TTD.2,6,7
Complications are common in pregnancies yielding affected infants who demonstrate abnormalities at birth. Most patients born with a collodion membrane develop ichthyosis, which is highly correlated with developmental delay in TTD.1 Other clinical features of TTD present in this case are low birth weight, growth deficiency, microcephaly, low-set and posteriorly rotated ears, nail abnormalities, dental abnormalities, and cryptorchidism. Horizontal nystagmus has also been previously observed,8 though cataracts are the most common ocular abnormality.1 Patients with TTD may also demonstrate spasticity, ataxia, and altered reflexes.1,2
As ectodermal derivatives, central nervous system and dermatologic abnormalities frequently occur in tandem. Compared with neurocutaneous disorders such as neurofibromatosis, Sturge-Weber syndrome, and tuberous sclerosis, the findings in TTD are more variable and less specific. Generalized dysmyelination is the most common neuroimaging abnormality in TTD. Similar dysmyelination in Cockayne syndrome is more likely to demonstrate cerebral calcifications, particularly in the basal ganglia.1,2,6,9
Other entities in the differential diagnosis for diffuse dysmyelination in the absence of gray matter abnormalities in children include Pelizaeus-Merzbacher disease, 18q-syndrome, and Salla disease.10 Although inborn errors of metabolism may result in delayed myelination mimicking the diffuse dysmyelination present in TTD, atrophy and distinct clinical syndromes generally allow differentiation.10 Axial osteosclerosis, though seen in a minority of patients with TTD, has not been reported in other diffuse dysmyelinating disorders and may be specific for the diagnosis in the presence of characteristic clinical findings.11–13
Other nonspecific structural abnormalities observed in TTD include cortical heterotopias, partial agenesis of the corpus callosum, perimedullary fibrosis of the spinal cord, and intracranial calcifications.6 MR spectroscopy has demonstrated increased myo-inositol and decreased choline in TTD, consistent with decreased membrane components in the absence of increased cell turnover.2 In practice, the clinical and pathologic findings remain the primary diagnostic criteria for TTD, whereas MR imaging is helpful for providing ancillary information in this heterogeneous set of disorders.
In conclusion, TTD should be included in the differential diagnosis for diffuse white matter abnormality in the pediatric population. The presence of diffuse dysmyelination in a child with documented hair and skin abnormalities strongly suggests the diagnosis. The presence of axial osteosclerosis in conjunction with diffuse dysmyelination may allow a specific diagnosis of TTD.
References
- Received March 31, 2009.
- Accepted after revision April 17, 2009.
- Copyright © American Society of Neuroradiology